Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD) ranges from simple steatosis, metabolic dysfunction-associated steatohepatitis (MASH), or cirrhosis to even hepatocellular carcinoma1.
Hepatic fibrosis is the main determinant of MASLD progression. Advanced fibrosis (stage ≥ 3) is associated with a higher risk of developing hepatic decompensation (ascites, variceal bleeding, and hepatic encephalopathy), need for liver transplantation, and increased mortality. Furthermore, the relevance of hepatic fibrosis transcends the hepatic sphere, as it is also related to a higher risk of developing cardiovascular disease2–4.
Recent studies suggest that clinically significant hepatic fibrosis (stage ≥ 2) also carries relevant consequences, including a higher risk of progressing to cirrhosis. It is estimated that up to 20% of patients with fibrosis in early stages will develop advanced fibrosis in approximately 5 years5.
Hepatic fibrosis in early stages constitutes a critical phase of the disease, as it is potentially treatable and even reversible with appropriate interventions. In this context, precise evaluation and correct staging of fibrosis acquire a central role in monitoring MASLD progression, to identify patients with advanced or clinically significant fibrosis (stage ≥ 2) in a timely manner6.
The therapeutic approach, therefore, should not be limited to improving hepatic steatosis but should also be directed toward preventing and slowing the progression of fibrosis. This narrative review highlights the main pharmacological strategies currently approved for the treatment of MASLD.
Method
The following narrative evidence synthesis was developed in the following steps:
- – First step: topics related to the pharmacological treatment of MASLD were selected.
- – Second step: two of the authors (DLP and JAVRV) evaluated, synthesized, and coded the information into the various topics.
- – Third step: after coding, targeted questions were developed and a systematic review was conducted in the DynaMed, Google Scholar, and PubMed databases. The hierarchy of the pre-appraised evidence pyramid was used to obtain information. It started with evidence summaries (clinical practice guidelines, UpToDate and DynaMed), followed by evidence syntheses or synopses (systematic reviews), and finally original studies (randomized controlled trials and observational studies). The information search was conducted in Spanish and English, and the following keywords were used: “steatosis,” “non-alcoholic fatty liver disease,” “drugs,” “treatment,” “fibrosis,” “MASH,” “NASH,” “MASLD,” “resmetirom,” and “GLP-1.” Publications from 2010 to July 2025 were included.
The results obtained were reviewed by the team of authors, who, using the standardized format, extracted relevant information for inclusion in this narrative evidence synthesis. The most relevant aspects are presented below.
Results
Criteria for initiating pharmacological treatment
The purpose of pharmacological treatment is to provide tangible clinical benefits to patients. In chronic liver disease, this could be expressed through significant reduction of clinical outcomes such as the development of cirrhosis and its complications (ascites, hepatic encephalopathy, gastrointestinal hemorrhage), as well as all-cause mortality7.
Lifestyle modification remains the fundamental basis of MASLD treatment, and pharmacological treatment is increasingly recognized as fundamental support, mainly in patients with biopsy-confirmed MASH, significant fibrosis (≥ F2), or cardiometabolic comorbidity8. It has been demonstrated that, although MASH drives disease progression, fibrosis is the most critical predictor of prognosis, in terms of both liver disease and overall mortality. Treatment for MASH is considered indicated when there is a score ≥ 4 in the Non-Alcoholic Steatohepatitis (NASH) Clinical Research Network (NASH CRN) histological scoring system, or an activity score ≥ 2 according to the Steatosis, Activity, and Fibrosis (SAF) scoring system, together with significant fibrosis9,10.
Drugs with indication and recent clinical evidence
The pathophysiology of MASLD and MASH integrates insulin resistance, lipotoxicity, inflammation, and fibrogenic remodeling. Current and emerging therapies act on key metabolic pathways: 1) hepatospecific activation of thyroid hormone receptor (THR) β to increase β-oxidation and reduce lipogenesis (resmetirom); 2) incretins (glucagon-like peptide-1 [GLP-1] receptor agonists and glucose-dependent insulinotropic peptide [GIP]), which induce weight loss and improve metabolic homeostasis with direct hepatic effects; 3) fibroblast growth factor 21 (FGF21) agonism, which restores lipid metabolism and inflammation; and 4) modulation via peroxisome proliferator-activated receptor (PPAR) agonists of steatosis, inflammation, and fibrosis. Fig. 1 shows the pathways and mechanisms of drugs used in MASLD.
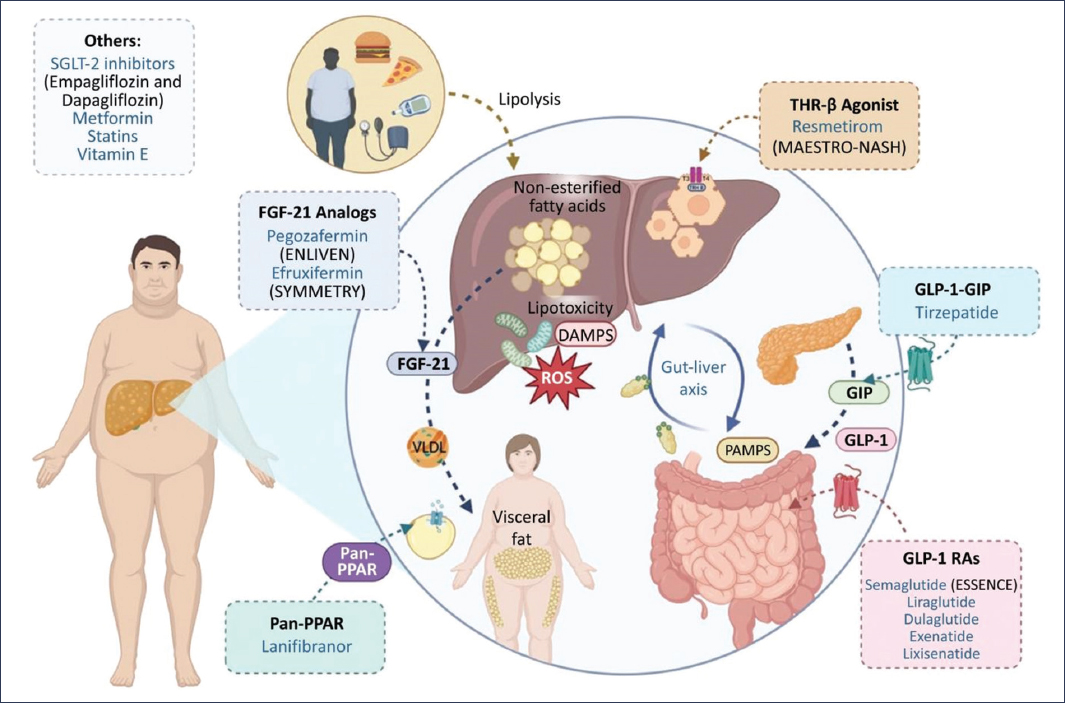
Figure 1. Pharmacological approach to MASLD. Schematic representation of the main pathophysiological mechanisms (lipotoxicity, oxidative stress, inflammation, and gut-liver axis) and therapies under evaluation directed at these processes, including THR-β agonists, FGF-21 analogs, pan-PPAR modulators, GLP-1 RAs, and GLP-1/GIP combinations, in addition to conventional treatments such as SGLT-2 inhibitors, metformin, statins, and vitamin E.
RESMETIROM
Thyroid hormones perform their functions by binding to two receptor isoforms, THR-α and THR-β. THR-β is expressed primarily in hepatocytes and plays a fundamental role in various metabolic processes. In particular, THR-β activation is linked to important metabolic benefits in the liver, as it can reduce cholesterol and triglyceride levels, stimulate bile acid synthesis, and promote fat oxidation. Resmetirom, a THR agonist, acts on hepatic fat accumulation to reduce MASH and advanced hepatic fibrosis11. When THR-β is stimulated by an agonist (e.g., resmetirom), it improves liver health by initiating a cascade of processes that are often dysregulated in patients with MASH and that are associated with disease progression and the development of hepatic fibrosis. To date, it is the first drug approved by the Food and Drug Administration (FDA) developed specifically for the treatment of MASH. The FDA’s determination to approve resmetirom was based on the findings of a phase 3 clinical trial (MAESTRO-NASH) that included a cohort of 966 patients with NASH12. Resolution of NASH without worsening of fibrosis was achieved in 25.9% of patients in the 80 mg resmetirom group and in 29.9% of those with 100 mg resmetirom, compared with 9.7% of those who received placebo (p < 0.001). Improvement in fibrosis by at least one stage, without worsening of the non-alcoholic fatty liver disease (NAFLD) activity score, was achieved in 24.2% of patients in the 80 mg resmetirom group and in 25.9% in the 100 mg resmetirom group, compared with 14.2% in the placebo group (p < 0.001). This study also documented a significant reduction in low-density lipoprotein cholesterol levels from baseline to week 24, compared with the placebo group (p < 0.001). Diarrhea and nausea were the most frequent adverse events; however, these were similar in all groups, confirming the general safety of resmetirom13.
These findings were confirmed in a meta-analysis of three randomized clinical trials (n = 2234) in which resmetirom significantly reduced hepatic fat by magnetic resonance imaging-proton density fat fraction (MRI-PDFF) with 80 mg (–27.74% at 36-52 weeks and −30.92% at 12-16 weeks) and with 100 mg (–36.05% and –36.89%; all p < 0.00001). Regarding safety, there were no overall differences in adverse events emerging compared with placebo (for 80 mg, odds ratio [OR]: 1.55, 95% confidence interval [95% CI]: 0.84-2.87; for 100 mg, OR: 1.13, 95% CI: 0.78-1.63), although diarrhea and nausea were more frequent in < 12 weeks14.
SEMAGLUTIDE
GLP-1 is released by enteroendocrine cells in the gastrointestinal tract and plays an essential role in the regulation of postprandial blood glucose levels by increasing insulin secretion and suppressing glucagon release15. GLP-1 also delays gastric emptying and attenuates food intake, achieving a balance between nutrient absorption and prevention of excessive weight gain. Due to its importance in energy metabolism, several studies have evaluated the effects and possible benefits of using GLP-1 receptor agonists in the context of liver health16. Among the various GLP-1 receptor agonists, semaglutide has demonstrated promising results in the resolution of MASH. The phase 3 ESSENCE trial, a multicenter, randomized, double-blind, placebo-controlled study, included 1197 patients with biopsy-confirmed MASH and F2-F3 fibrosis, of whom 800 were analyzed interim after 72 weeks of treatment with semaglutide at a dose of 2.4 mg subcutaneously weekly or placebo (2:1). Semaglutide achieved resolution of steatohepatitis without worsening of fibrosis in 62.9% versus 34.3% (Δ 28.7 pp; p < 0.001) and a reduction in fibrosis ≥ 1 stage without worsening of steatohepatitis in 36.8% versus 22.4% (Δ 14.4 pp; p < 0.001), with a mean weight loss of 10.5% versus 2.0% and improvements in enhanced liver fibrosis (ELF) index, hepatic stiffness, and cardiometabolic parameters. Regarding the safety profile, it was consistent with GLP-1 agonists, with mild to moderate gastrointestinal events predominating17. Recently, the FDA issued its approval for the treatment of MASH in adults with moderate to advanced fibrosis18.
PEGOZAFERMIN
Pegozafermin is a long-acting recombinant FGF21 analog, which regulates energy metabolism, mainly in situations of metabolic stress19. Preclinical studies have demonstrated that FGF21 could enhance AMP-activated protein kinase (AMPK) signaling, favoring fatty acid oxidation and reducing de novo lipogenesis, which contributes to suppression of fibrosis and improvement of metabolic status20. The phase 2b ENLIVEN trial, a multicenter, randomized, double-blind, placebo-controlled study, included 222 patients with biopsy-confirmed NASH and F2-F3 fibrosis, assigned to receive pegozafermin at doses of 15 mg weekly, 30 mg weekly, 44 mg every 2 weeks, or placebo for 24 weeks. Improvement in fibrosis ≥ 1 stage without worsening of NASH was achieved in 26% with 30 mg and in 27% with 44 mg, versus 7% with placebo (p = 0.009 and p = 0.008), and resolution of NASH without worsening of fibrosis in 23% with 30 mg, 26% with 44 mg, and 37% with 15 mg, versus 2% with placebo. In addition, a significant reduction in hepatic fat by MRI-PDFF of up to −48.2%, normalization of alanine aminotransferase (ALT) up to 65%, and improvements in ELF index, hepatic stiffness, Pro-C3, and lipid profile were observed. Regarding safety, mild to moderate gastrointestinal events were observed and one case of acute pancreatitis21. In another unique multicenter cohort, which included 20 patients with biopsy-confirmed NASH and F2-F3 fibrosis, after treatment with pegozafermin at a dose of 27 mg weekly for 20 weeks, an improvement ≥ 2 points in the NAFLD activity score (NAS) with ≥ 1 point in ballooning or lobular inflammation, and without worsening of fibrosis, was observed in 63% of patients, with a marked reduction in hepatic fat by MRI-PDFF (−64.7%; 100% with ≥ 30% reduction), normalization of ALT in 79% of those who had it elevated, and improvements in non-invasive markers of fibrosis22.
EFRUXIFERMIN
Efruxifermin is a bivalent FGF21 analog with direct antifibrotic actions and indirect effects on glucose and lipid metabolism, administered subcutaneously weekly. The phase 2b SYMMETRY trial, a multicenter, randomized, double-blind, placebo-controlled study, included 181 patients with compensated cirrhosis (Child-Pugh A) due to MASH (78% with histological diagnosis and 22% with cryptogenic cirrhosis attributed to MASH) assigned to receive efruxifermin at doses of 28 or 50 mg, or placebo (1:1:1), for 96 weeks. The primary endpoint (reduction in fibrosis ≥ 1 stage without worsening of MASH at 36 weeks) showed no significant differences: 13% with placebo, 18% with 28 mg, and 19% with 50 mg. At 96 weeks, the rates were 11%, 21%, and 29%, respectively (Δ vs. placebo: +16 pp for 50 mg; 95% CI: 2-30), with resolution of MASH in 42% of those treated versus 13% of those who received placebo, and with improvements in ELF index, hepatic stiffness, liver enzymes, and cardiometabolic profile. Adverse events were mainly gastrointestinal and local reactions, mostly mild or moderate23. In another previous study, 89% of patients experienced at least one treatment-related adverse event, the most common being grade 1-224. Finally, a meta-analysis that included four studies with a total of 325 patients with MASH and fibrosis in stages F1-F4 showed that efruxifermin has a relative risk (RR) of 1.97 (95% CI: 1.21-3.19; p = 0.006) for improving fibrosis ≥ 1 stage without worsening MASH. In addition, efruxifermin improved non-invasive biomarkers related to fibrosis, such as the ELF score and Pro-C325.
LANIFIBRANOR
Lanifibranor is a pan-PPAR agonist that regulates metabolic, inflammatory, and fibrotic pathways. It specifically favors fatty acid oxidation and inhibits nuclear factor kappa B signaling26. In a phase IIb clinical trial, 288 patients were randomly assigned to receive 1200 or 800 mg of lanifibranor or placebo once daily for 24 weeks. At the end of treatment, a significant improvement in histological steatosis was observed compared with placebo: more than 35% of patients treated with lanifibranor showed an improvement of at least 2 grades in steatosis, versus 5% in the placebo group. The controlled attenuation parameter (CAP) decreased significantly: with 1200 mg, the estimated adjusted mean difference was –23 dB/m (95% CI: –41 to –5; p = 0.012), and with 800 mg it was –16 dB/m (95% CI: –33 to 1; p = 0.070), compared with placebo, with which there were no changes. At the end of treatment, a significantly higher proportion of patients achieved a CAP ≤ 302 dB/m (equivalent to mild or absent steatosis, ≤ S1): 50% with 1200 mg (95% CI: 36-64; p = 0.009), 46% with 800 mg (95% CI: 33-60; p = 0.019), and only 25% with placebo (95% CI: 13-37). A significant correlation was observed between CAP and the degree of histological steatosis, both at baseline and at the end of treatment (Spearman coefficient Rs = 0.37; p < 0.001). Finally, the reduction in CAP was associated with decreases in triglyceride and glycosylated hemoglobin levels at the end of treatment27.
TIRZEPATIDE
Tirzepatide is a co-agonist of the incretin hormones GLP-1 and GIP approved by the FDA for the treatment of type 2 diabetes mellitus (T2DM) and weight loss28. It is important to note that increased incretin signaling not only benefits patients with prolonged weight loss but also improves glycemic regulation, suggesting its clinical relevance in the treatment of MASH. To determine the efficacy and safety of tirzepatide, a phase 2 clinical trial was conducted with 190 patients, who were randomly assigned to receive weekly doses of 5, 10, or 15 mg or placebo, for 52 weeks. The percentage of participants who met the criteria for resolution of MASH without worsening of fibrosis was 10% in the placebo group, 44% in the 5 mg tirzepatide group (difference versus placebo 34%; 95% CI: 17-50), 56% in the 10 mg tirzepatide group (difference 46%; 95% CI: 29-62), and 62% in the 15 mg tirzepatide group (difference 53%; 95% CI: 37-69) (p < 0.001 for all three comparisons). The percentage of participants who presented an improvement in fibrosis of ≥ 1 stage without worsening of MASH was 30% in the placebo group, 55% in the 5 mg tirzepatide group (difference versus placebo 25%; 95% CI: 5-46), 51% in the 10 mg tirzepatide group (difference 22%; 95% CI: 1-42), and 51% in the 15 mg tirzepatide group (difference 21%; 95% CI: 1-42). The most frequent adverse events in the tirzepatide groups were gastrointestinal, and most were mild or moderate29.
SURVODUTIDE
Survodutide is a dual GLP-1 and GIP agonist. To evaluate its performance, a phase II clinical trial was conducted with 293 patients with a weekly dose of 2.4, 4.8, or 6.0 mg, versus placebo. An improvement in MASH without worsening of fibrosis was observed in 47% of participants in the 2.4 mg survodutide group, 62% of those in the 4.8 mg group, and 43% of those in the 6.0 mg group, compared with 14% of those in the placebo group (p < 0.001). A decrease of at least 30% in content was observed in 63% of participants in the 2.4 mg survodutide group, 67% of those in the 4.8 mg group, 57% of those in the 6.0 mg group, and 14% of those in the placebo group. There was an improvement in fibrosis of ≥ 1 stage in 34%, 36%, 34%, and 22%, respectively. The most frequent adverse events with survodutide versus placebo were nausea (66% versus 23%), diarrhea (49% versus 23%), and vomiting (41% versus 4%)30.
Other drugs
VITAMIN E
Vitamin E is an essential micronutrient and fat-soluble antioxidant with protein functions and gene modulation effects. Its metabolism depends on hepatic binding proteins that selectively retain alpha-tocopherol from food for its incorporation into very low-density lipoproteins and its subsequent tissue distribution, together with esterified cholesterol and triglycerides31. Several studies have demonstrated that vitamin E supplementation can significantly improve liver enzyme levels, aspartate aminotransferase (AST) and ALT, reduce hepatic steatosis, and improve hepatic inflammation32. In a systematic review, the efficacy of vitamin E supplementation in adult patients with MASLD was evaluated, with doses ranging from 200 IU to 1000 mg daily, and treatment duration ranged from 12 to 96 weeks. Higher doses consistently showed significant reductions in liver enzymes (AST and ALT), hepatic steatosis, and inflammation, although the effect on fibrosis was limited. This finding was reinforced in the study by Pervez et al.33, in which the administration of δ-tocotrienol and α-tocopherol achieved significant decreases (p < 0.001) in ALT and AST, as well as total cholesterol and low-density lipoprotein cholesterol (p < 0.05), and a significant increase in high-density lipoprotein cholesterol, compared with baseline values, in both study groups. With respect to histological improvements, this same study demonstrated significant improvements in hepatic steatosis and liver-spleen attenuation ratios in the tocopherol and tocotrienol groups, which further supports the role of vitamin E in improving hepatic fat content33. Abera et al.34 also found improvements in steatosis, indicating that higher doses of vitamin E (400-800 IU/day) are more effective in addressing histological characteristics.
The effect of vitamin E on fibrosis is less conclusive; although several studies found improvements in steatosis and inflammation, they did not show significant changes in fibrosis35. The overall evidence of the impact of vitamin E on fibrosis remains limited, suggesting that its main role may be in addressing early-stage liver damage rather than reversing advanced fibrosis.
STATINS
In addition to their lipid-lowering effect, statins have anti-inflammatory, antioxidant, pro-apoptotic, and antifibrotic properties that could benefit patients with MASLD and MASH36. In a study conducted in individuals with metabolic syndrome without diabetes or cerebrovascular event, liver enzymes and ultrasound improved during the study with the use of statins37. A meta-analysis highlighted the effect of statins in significantly reducing hepatic biochemical markers in MASLD38 and their association with a lower risk of hepatocellular carcinoma39. Ayada et al.40 conducted a study that included a cross-sectional investigation in an ongoing general population cohort (Rotterdam study) and a cohort of patients with MASLD (PERSONS cohort), a meta-analysis, and an experimental exploration. In the analysis of the 4576 participants in the Rotterdam study, statin use in patients with dyslipidemia was inversely associated with MASLD compared with participants with untreated dyslipidemia (OR: 0.72; 95% CI: 0.59-0.86). In the PERSONS cohort, analysis of 569 patients with biopsy-proven MASH showed that statin use was inversely associated with MASH (OR: 0.55; 95% CI: 0.32-0.95), but not with fibrosis (OR: 0.86; 95% CI: 0.44-1.68)40. Currently, the STAT MASH trial (NCT04679376) is evaluating the efficacy and safety of atorvastatin in MASH with histological criteria. Overall, statins are safe in patients with MASLD, do not increase the risk of hepatotoxicity, and are indicated to prevent cardiovascular events. However, their specific efficacy for the treatment of MASH still requires confirmation through large-scale clinical trials41.
SODIUM-GLUCOSE COTRANSPORTER TYPE 2 (SGLT2) INHIBITORS
They are antihyperglycemic agents that act on SGLT-2 proteins, expressed in the proximal convoluted tubules. These drugs exert their effect by preventing the reabsorption of filtered glucose in the tubular lumen42. In addition, through stimulation of the SIRT1/AMPK signaling pathway and inhibition of Akt/mTOR, they exert effects that include reduction of oxidative stress, inflammation, and microvascular damage, benefiting cardiac and renal functions43. Clinically, SGLT2 inhibitors have demonstrated cardiovascular benefit in patients with atherosclerotic cardiovascular disease and T2DM, making them a first-line therapeutic approach for this group of patients44. In particular, the EMPEROR-PRESERVED trial demonstrated that empagliflozin significantly reduces the combined risk of cardiovascular death or hospitalization for heart failure in patients with heart failure and preserved ejection fraction, regardless of the presence or absence of diabetes45. In the case of canagliflozin, randomized clinical trials have demonstrated that it improves hepatic insulin sensitivity, as well as insulin secretion and clearance in patients with T2DM. The reduction in hepatic triglyceride content is proportionally correlated with the magnitude of weight loss, which was greater and more frequent in patients treated with canagliflozin46. Notable are the EMPA-REG OUTCOME trial, which showed a significant reduction in aminotransferases with empagliflozin47, and a meta-analysis of ten randomized controlled trials with 573 participants that confirmed the superiority of SGLT2 inhibitors over other antihyperglycemic agents in terms of improvement in hepatic and metabolic parameters48. Evidence in non-diabetic patients is scarce; a small study compared dapagliflozin and teneligliptin in patients with MASLD and found improvements in transaminases and a reduction in body fat only in the group treated with dapagliflozin49. Despite this, there is no conclusive evidence to justify its use in the treatment of MASH41.
METFORMIN
Metformin is a hypoglycemic agent that acts by activating AMPK in cells and reducing glucose production in the liver50. In a meta-analysis, a significant association with the reduction of biochemical parameters in patients with MASLD was demonstrated. With respect to ALT, there was a mean reduction that reached the threshold of statistical significance (mean deviation [MD] = −10.84; 95% CI: −21.85 to 0.16; p = 0.05). For AST, the analysis showed a statistically significant decrease (MD = −4.82; 95% CI: −9.33 to −0.30; p = 0.04). Regarding body mass index, no significant effect of metformin was identified (MD = −0.65; 95% CI: −1.46 to 0.16; p = 0.12). On the contrary, a significant reduction was observed in serum triglyceride levels (MD = −0.17 mmol/l; 95% CI: −0.26 to −0.08; p = 0.0002) and total cholesterol (MD = −0.29 mmol/l; 95% CI: −0.47 to −0.10; p = 0.003). Additionally, insulin resistance, evaluated using the HOMA-IR index, showed a statistically significant reduction in the group treated with metformin (MD = −0.42; 95% CI: −0.82 to −0.02; p = 0.04). These findings suggest that metformin could contribute to improving hepatic and lipid profile, as well as insulin sensitivity51. However, there is currently no evidence that metformin alone can improve histology in liver disease associated with T2DM. Regarding clinical outcomes, there is evidence, derived from observational and case-control studies, suggesting that in patients with T2DM and advanced fibrosis or cirrhosis associated with MASLD, metformin may improve transplant-free survival without affecting the risk of hepatic decompensation, in addition to reducing the risk of hepatocellular carcinoma and extrahepatic neoplasms. In a cohort of patients with advanced cirrhosis, 85% of those treated with metformin and 88% of those not treated had this pathology. After a follow-up of 6.9 years, metformin use was associated with lower risk of all-cause mortality and transplantation (hazard ratio [HR]: 0.42; 95% CI: 0.24-0.74; p = 0.003), and hepatocellular carcinoma (HR: 0.25; 95% CI: 0.11-0.58; p = 0.001)52. Therefore, it is suggested that in individuals with cirrhosis, metformin should not be discontinued, unless necessary, as this could increase mortality53. There is insufficient evidence to recommend the use of metformin as targeted therapy for MASH, but it is safe in patients with MASLD and should be used for its respective indications, namely T2DM, heart failure, and chronic kidney disease54.
COMBINATION THERAPIES
In MASH, combination therapy directed at different pathways represents an attractive alternative to improve clinical efficacy and reduce adverse effects through lower doses or drugs with complementary actions55. Combination therapy constitutes an active area of research in MASH. The main rationale is that targeting multiple pathways would improve efficacy and therapeutic index56. In addition, the large number of pathways and agents considered for MASH generates difficulties in choosing two optimal pathways. Other considerations for combination therapy trials are the greater complexity of regulatory requirements, the higher number of patients required, the selection of the optimal dose, the possible increase in side effects, and cost57. Despite these challenges, several phase 2 combination trials have been completed. In a phase 2b trial, 392 patients with bridging fibrosis or compensated cirrhosis (F3-F4) were randomized to receive placebo, selonsertib 18 mg, cilofexor 30 mg, or firsocostat 20 mg, alone or in two-drug combinations, once daily, for 48 weeks. Most had cirrhosis (56%) and an NAFLD activity score ≥ 5 (83%). The combination of cilofexor and firsocostat achieved a significant decrease in the ML NASH CRN fibrosis score (p = 0.040) and a change in biopsy area from F3-F4 fibrosis patterns to ≤ F2. Compared with placebo, significantly higher proportions of patients with cilofexor/firsocostat had a reduction in NAS ≥ 2 points; reductions in steatosis, lobular inflammation, and ballooning; and significant improvements in ALT, AST, bilirubin, bile acids, cytokeratin-18, insulin, estimated glomerular filtration rate, ELF score, and hepatic stiffness by transient elastography (p ≤ 0.05 in all cases). Pruritus was observed in 20-29% of patients treated with cilofexor, versus 15% of those who received placebo58. In a trial with 108 patients, semaglutide with cilofexor (farnesoid X receptor [FXR] agonist), semaglutide with the acetyl-CoA carboxylase (ACC) enzyme inhibitor firsocostat, and all three drugs were compared. The treatments were well tolerated: the incidence of adverse events was similar between groups (73-90%), and most events were gastrointestinal in nature. Despite similar weight loss (7-10%), compared with semaglutide monotherapy, the combinations resulted in greater improvements in hepatic steatosis measured by MRI-PDFF (least squares mean of absolute changes: –9.8 to –11.0% versus –8.0%), hepatic biochemistry, and non-invasive tests of fibrosis. In addition, a uniform and statistically significant reduction in ALT, as a marker of hepatocyte damage, was observed in all combination groups59.
It is possible that combination therapy offers advantages over monotherapy, opening the debate on whether it should always be adopted as first-line treatment, reserved only for a brief initial phase followed by maintenance monotherapy, or applied only in patients who do not respond to monotherapy. This decision will depend largely on the type of benefits provided by the combination therapy, which could include a greater degree of response, an increase in the proportion of patients who respond, a faster or more durable response, as well as a better tolerability and adverse event profile57. Within the combination options, incretin-based therapies will play a relevant role, given that most patients with MASH have T2DM or obesity, conditions in which these are already commonly used. In clinical practice, the most frequent question will be to determine which drug to add to the incretin that a patient with MASH is already receiving. For example, a phase IIb, double-blind, placebo-controlled study was conducted in adults with T2DM and MASH with fibrosis (F1-F3) on stable treatment with GLP-1 receptor agonists to receive efruxifermin 50 mg or placebo once weekly for 12 weeks. In total, 31 adults with T2DM and fibrosis due to MASH (F1-F3) on treatment with a stable GLP-1RA (semaglutide 48.4%, dulaglutide 45.2%, and liraglutide 6.5%) received efruxifermin 50 mg (n = 21) or placebo (n = 10) for 12 weeks. The most frequent adverse events related to efruxifermin were gastrointestinal and mild to moderate. After 12 weeks, efruxifermin reduced hepatic fat fraction (HFF) by 65% (p < 0.0001 versus placebo) compared with a 10% reduction with placebo (GLP-1RA alone). Efruxifermin also improved non-invasive markers of liver injury, fibrosis, glucose, and lipid metabolism, while maintaining GLP-1RA-mediated weight loss60.
Table 1 shows the most relevant studies on drugs used in the treatment of MALSD.
Table 1. Pharmacological treatments of MASLD
| Drug | Clinical trials | Dosage | Study population | Results | Potential adverse effects |
|---|---|---|---|---|---|
| Vitamin E* | Sanyal et al.67 | 800 IU/day, 96 weeks | MASH without T2DM or cirrhosis | Improvement in steatosis, inflammation and ballooning, improvement in transaminases, MASH resolution? | Hemorrhagic cerebrovascular event, prostate cancer risk? |
| Semaglutide |
Newsome et al.68, Flint et al.69 | 0.4 mg/day, subcutaneous, 72 weeks | MASH without cirrhosis and MASLD without significant fibrosis | Resolution of MASH, decrease in HFC, decrease in weight, CVR and insulin resistance, decrease in fibrosis progression? | Nausea, constipation, vomiting, cholelithiasis, pancreatitis |
| Dulaglutide | Seko et al.70 Kuchay et al.71 | 0.75-1.5 mg/week, 12 to 24 weeks | MASLD with T2DM | Decrease in steatosis, weight loss, improvement in transaminases and GGT | Epigastralgia and diarrhea |
| Empagliflozin |
Kuchay et al.72 Kahl et al.73 | 10-25 mg/day, 20-24 weeks | MASLD with T2DM | Decrease in HFC and ALT | Polyuria and fungal genitourinary infections |
| Dapagliflozin |
Latva-Rasku et al.74 Tobita et al.75 | 5-10 mg/day, 8-12 weeks | MASLD with T2DM and MASLD alone | Decrease in HFC, weight loss and improvement in transaminases | |
| Ipragliflozin | Ito et al.76, Takahashi et al.77 | 50 mg/day, 24-72 weeks | MASLD with T2DM | Decrease in weight, abdominal fat, improvement in fibrosis and glycemic control | |
| Canagliflozin |
Akuta et al.78 | 100 mg/day, 24 weeks | MASLD with T2DM | Improvement in histological parameters and TE, BMI and visceral fat | |
| Tirzepatide | Gastaldelli et al.79 | 10-15 mg/week, subcutaneous, 52 weeks | T2DM with overweight and MASLD | Improvement in HFC, weight loss and improvement in insulin resistance | Cholelithiasis and pancreatitis |
| Retatrutide | Jastreboff et al.80 | 4-12 mg/week, 48 weeks | Obesity | Substantial weight reduction | Nausea, diarrhea, vomiting and constipation |
| Lanifibranor | Francque et al.26 | 800-1200 mg/day, 24 weeks | MASH without cirrhosis | Resolution of MASH, decrease in transaminases | Diarrhea, nausea, peripheral edema, anemia, weight gain |
| Elafibranor | Ratziu et al.81 | 80-120 mg/day, 52 weeks | MASH without cirrhosis | Resolution of MASH without worsening of fibrosis | Reversible creatinine elevation |
| Resmetirom | Harrison et al.12 | 80 or 100 mg/day, 52 weeks | MASH without cirrhosis | Resolution of MASH and improvement in fibrosis | Diarrhea and nausea |
* Main studies published with clinical results demonstrated in MASLD according to population characteristics and adverse effects observed. The first approved drug (resmetirom) is included. ALT: alanine aminotransferase; T2DM: type 2 diabetes mellitus; TE: transient elastography; GGT: gamma-glutamyl transpeptidase; BMI: body mass index; HFC: hepatic fat content; CVR: cardiovascular risk.
Pharmacogenomics
I148M VARIANT OF THE PNPLA3 GENE
The I148M mutation of the PNPLA3 gene has been considered the main genetic factor related to susceptibility to MASH61. PNPLA3 has hydrolase activity on triglycerides, and the I148M genetic variant is associated with a functional loss of this activity, which promotes the accumulation of triglycerides in hepatocytes and favors the development of hepatic steatosis62. The PNPLA3 gene mutation plays a fundamental role in the progression of MASLD. Therefore, targeting treatment toward the PNPLA3 protein that carries the I148M mutation represents a promising therapeutic strategy. In fact, several studies have demonstrated that specific silencing of PNPLA3 in the liver, through antisense oligonucleotides (ASO), significantly improves various aspects of MASLD, including decreased hepatic steatosis, inflammation, and fibrosis63. Considering these preclinical studies, an ASO directed at PNPLA3, called ION839 (AZD 2693), is currently in phase I clinical investigation in patients with MASH carrying the I148M risk alleles of PNPLA3 (NCT04483947). In addition to the ASO, the efficacy of an RNAi directed at PNPLA3, known as LY3849891, is also being evaluated in clinical trials for patients with MASH with PNPLA3 variants (NCT05395481)64. Specific modulation of PNPLA3 activity through RNAi integrates a potentially effective therapeutic approach for the treatment of MASH. However, these RNAi-based interventions are in a preliminary stage, and exhaustive research is required to clarify the therapeutic benefits and safety of these innovative approaches before their clinical application20.
RS72613567 VARIANT IN HSD17B13
The HSD17B13 protein is a hepatic retinol dehydrogenase that is located primarily on the surface of lipid droplets in hepatocytes. Inhibition of HSD17B13 expression suppresses hepatic fibrosis by reducing pyrimidine catabolism65. Although the precise mechanisms of HSD17B13 variants in MASH remain unclear, studies have identified that protein kinase A-mediated phosphorylation of HSD17B13 interacts with adipose triglyceride lipase to promote lipolysis and suppress MASH66. Therefore, these findings indicate that HSD17B13 is a possible pharmacological target against MASH.
A phase I/II proof-of-concept clinical trial was conducted to evaluate the efficacy of a 71-day intervention with ARO-HSD. Treatment was well tolerated, and the mean changes in hepatic HSD17β13 mRNA from baseline to day 71 were –56.9% with 25 mg, –85.5% with 100 mg, and –93.4% with 200 mg. Hepatic HSD17β13 protein levels were reduced similarly with all doses. In patients, changes in ALT from baseline to day 71 were –7.7% with 25 mg, –39.3% with 100 mg, and –42.3% with 200 mg (p < 0.001 for pooled cohorts). There were no serious adverse events related to treatment or drug discontinuations. The most frequently reported treatment-emergent adverse events were mild injection site reactions of short duration. ARO-HSD was well tolerated at doses ≤ 200 mg. This study demonstrated that short-term treatment with ARO-HSD reduces hepatic expression of HSD17β13 mRNA and protein, which is accompanied by reductions in ALT.
Conclusions
Intensive lifestyle modification remains the cornerstone of MASLD management. However, in MASH with significant fibrosis (≥ F2), pharmacological therapy provides measurable clinical benefits and should be considered with priority, especially if obesity or T2DM coexist (Fig. 2). Fibrosis is the strongest predictor of hepatic outcomes and overall mortality, above inflammatory activity, so the therapeutic objective should focus on halting and reversing fibrosis.
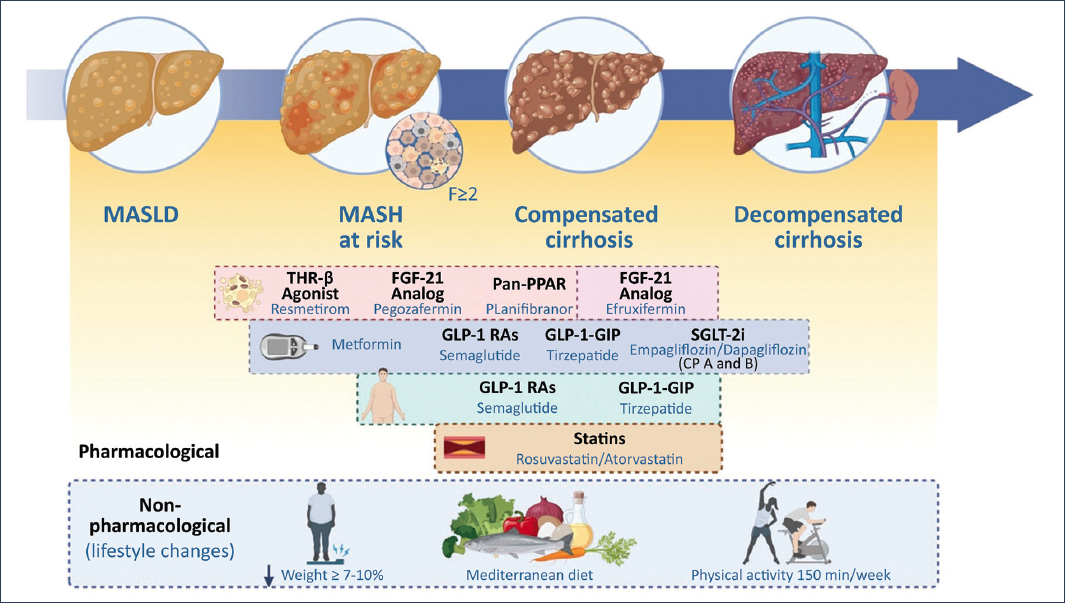
Figure 2. Treatment of MASLD throughout disease progression, from the initial stage to decompensated cirrhosis. Non-pharmacological interventions (weight loss ≥ 7-10%, Mediterranean diet, and physical activity) are represented as the therapeutic basis, together with pharmacological strategies under investigation or clinical use (THR-β agonists, FGF-21 analogs, pan-PPAR modulators, GLP-1 RAs, GLP-1/GIP, SGLT-2 inhibitors, metformin, and statins).
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
J.A. Velarde-Ruiz Velasco has participated in an advisory board for Novo Nordisk México.
Ethical considerations
Protection of people and animals. The authors declare that no experiments were performed on human beings or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal data of patients nor requires ethical approval. The SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.
