Introduction
Diet as a non-pharmacological therapeutic intervention is defined as the structured and personalized modification (based on physical characteristics and nutritional needs according to pathologies) of eating habits with the aim of improving the clinical and metabolic status of patients. Focusing on metabolic-associated steatotic liver disease (MASLD), nutritional intervention is the fundamental pillar of clinical management, as it has been shown to achieve improvement in metabolic parameters, as well as a decrease in insulin resistance and systemic inflammation, in addition to the fact that the associated weight loss manages to reverse intrahepatic lipid accumulation, a key factor in the progression of liver disease; in those patients with mild fibrosis who achieve weight loss > 10%, reversal of fibrosis can be achieved1.
Furthermore, nutritional intervention combined with regular physical activity potentiates metabolic benefits, favoring sustained weight loss and improving the inflammatory profile. This multidisciplinary approach is essential given that MASLD is a complex disease with multiple etiological factors related to metabolic syndrome. Therefore, diet personalization and long-term adherence are crucial to achieving significant and sustainable clinical outcomes2.
Importance of diet in the treatment of the metabolic-associated steatotic liver disease spectrum
Diet plays a crucial role in the management of MASLD in conjunction with pharmacological therapy. Weight reduction achieved through caloric restriction and a healthy diet can achieve improvement in liver enzymes, intrahepatic fat, liver inflammation, and fibrosis, in a manner dependent on adherence. The clinical practice guidelines of the European Association for the Study of the Liver (EASL), the European Association for the Study of Diabetes (EASD) and the European Association for the Study of Obesity (EASO), and the European Society for Clinical Nutrition and Metabolism (ESPEN) guideline recommend weight loss of 7% to 10% to improve steatosis and liver biochemistry, and > 10% to reverse fibrosis. Normal-weight patients can benefit from visceral fat reduction through diet and physical activity; steatosis remission can be achieved without requiring weight loss > 5%. Normal-weight patients usually present with genetic determinants that can benefit from dietary modifications, especially those with the G allele rs738409 of PNPLA3, a genotype that amplifies the influence of dietary intake. Moreover, the relationship between some specific dietary nutrients and fibrosis risk is stronger for individuals with this genetic modification3,4.
For a nutritional intervention to be optimal, it must perfectly combine the qualitative and quantitative aspects of adequate nutrition, considering the role of individual macro- and micronutrients in the onset and development of the disease, as well as the importance of modulating their daily intake to promote weight loss and counteract collateral pathological conditions5. Evidence demonstrates that, regardless of total caloric intake, certain specific macronutrients are crucial in the development and progression of MASLD; among them, carbohydrates, particularly fructose, have a close relationship with the onset and progression of MASLD. In this context, it is essential to regulate the total dietary intake of carbohydrates, especially those with a high glycemic index, as excessive intake can contribute to obesity and metabolic alterations. Reducing carbohydrate intake can significantly improve MASLD-related outcomes, including hepatic triglyceride content, triglyceridemia, lipid oxidation, and insulin resistance3.
Is it time for individualized treatment in diet?
The clinical spectrum of MASLD encompasses a variety of phenotypes ranging from simple steatosis to steatohepatitis, conditions that differ by the variable presence of biochemical inflammation (elevation of transaminases or serum inflammatory markers, such as interleukins) and liver damage with evidence of inflammatory infiltrate in liver tissue, in addition to the presence of hepatic fibrosis and its severity6.
In addition to these clinical phenotypes, MASLD coexists with cardiometabolic comorbidities, such as overweight and obesity, insulin resistance, type 2 diabetes, atherogenic dyslipidemia (characterized by increased low-density lipoprotein cholesterol and decreased high-density lipoprotein cholesterol), and arterial hypertension. The coexistence of comorbidity not only worsens the hepatic prognosis but also increases the patient’s overall cardiovascular risk1.
Therefore, before establishing a nutritional intervention in MASLD, the heterogeneity of comorbidity present in each patient must be considered and analyzed, in addition to the presence or absence of inflammation (steatohepatitis) and hepatic fibrosis. For example, in patients with obesity who meet metabolic syndrome criteria, it has been shown that caloric restriction with emphasis on macronutrient quality, such as the adoption of Mediterranean-style dietary patterns rich in monounsaturated and polyunsaturated fats, improves insulin sensitivity, reduces hepatic lipid load, and modulates systemic inflammation2. In patients with type 2 diabetes, strict control of glucose intake and foods with a high glycemic index is essential to improve hepatic metabolism and prevent disease progression7. Therefore, each comorbidity situation imposes specific nutritional requirements that must be addressed to optimize therapeutic outcomes.
The need to individualize dietary strategies in MASLD lies in the complex interaction of clinical phenotypes, associated comorbidity, and individual patient characteristics, including social, genetic, biochemical, and environmental factors. Studies emphasize that a “one size fits all” approach to nutritional recommendations is insufficient and ineffective for achieving metabolic improvement and long-term follow-up. Personalization of the diet, integrating cultural preferences, metabolic profile, and specific biomarkers, allows for an efficient and sustainable long-term approach, promoting adherence and achieving better clinical outcomes8. In general, nutritional recommendations should minimize ultra-processed foods, sugary foods and beverages, and saturated fats4.
Some dietary recommendations that, from a practical point of view, can be very useful for first-contact medical personnel are based on:
- – Reduce total energy intake: moderate caloric restriction (500-1000 kcal/day) can be achieved with different dietary patterns; lose 5-10% of body weight over a 6-month period.
- – Follow a healthy and balanced eating pattern, appropriate to the patient’s context: rich in fruits, vegetables, whole grains, legumes, nuts, fish, and olive oil, and low in red and processed meats, refined carbohydrates, and saturated fats.
- – Limit consumption of added sugars and refined carbohydrates: avoid or limit consumption of sugar-sweetened beverages, sweets, and energy-dense snacks.
- – Avoid alcohol intake: alcohol can worsen liver damage, so its consumption should be avoided or limited.
- – Reduce saturated fat intake.
We must remember that long-term objectives focus on reducing intrahepatic fat, improving fibrosis, and preventing metabolic and hepatic complications. In conclusion, and in response to the question of whether it is time for individualized treatment in diet, the answer is yes: diet individualization is not only a clinical imperative but also a fundamental strategy to improve quality of life and prognosis of patients with MASLD and their comorbidity (Fig. 1).
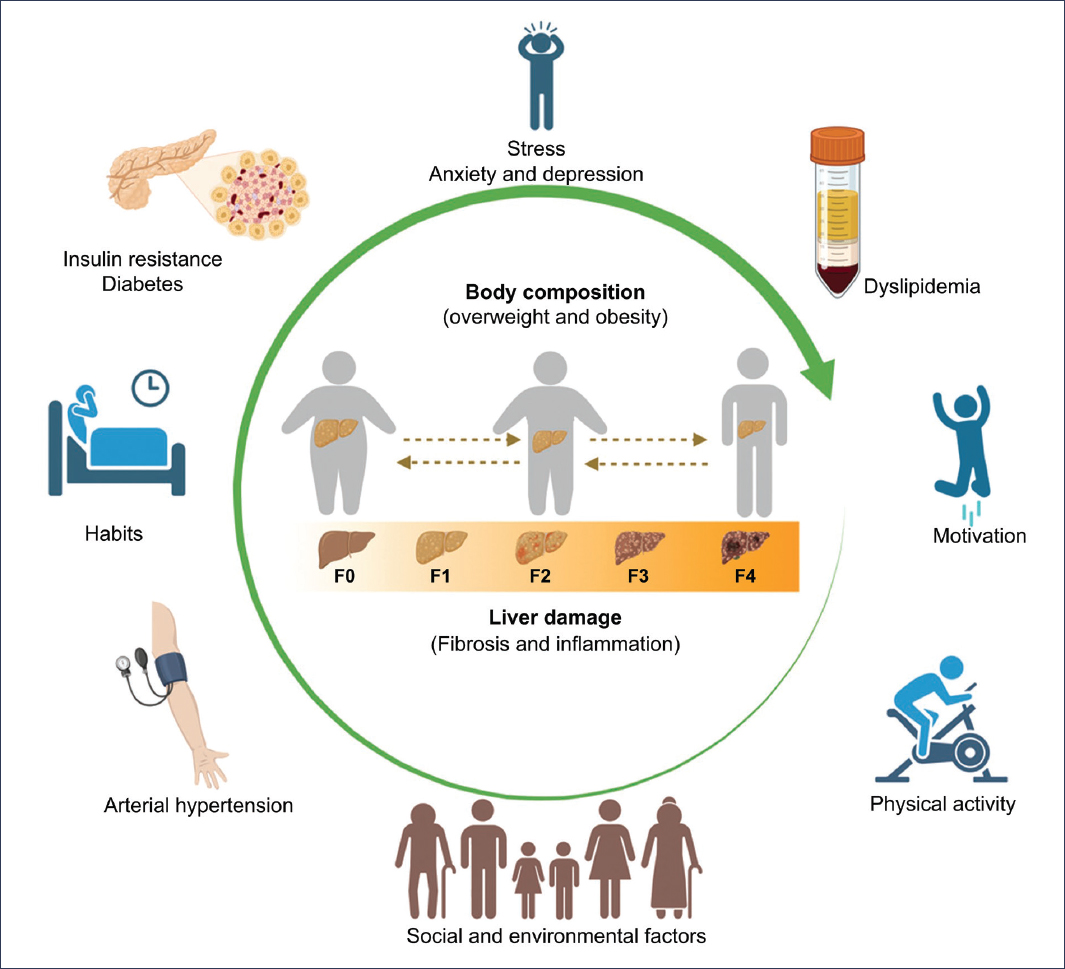
Figure 1. Components that should be considered for personalization of nutritional plans in patients with MASLD (created in BioRender).
What is the best diet for patients with metabolic-associated steatotic liver disease?
The link between unhealthy eating habits and the development and progression of MASLD has been clearly demonstrated. Therefore, various nutritional interventions for the treatment of MASLD have been explored, with the Mediterranean diet being the most studied and recommended. Other plant-based dietary approaches, including the DASH (Dietary Approaches to Stop Hypertension) diet and vegetarian diet, have also shown potential benefits8,9. Optimal dietary intervention strategies for MASLD are not currently standardized.
Considering existing evidence, the approach of a diet based on healthy eating patterns with minimally processed or unprocessed foods, low in sugar and saturated fats, and high in polyphenols, omega-3 polyunsaturated fatty acids, and monounsaturated fatty acids, leads to the conclusion that, ideally, the Mediterranean diet, or other overlapping dietary patterns, should serve as a base and be restructured into different types of diets. The exact type of diet, in terms of macronutrient composition, food choice, and meal timing, can be adapted to patient preferences to increase long-term adherence, provided these principles are respected4.
The nutritional intervention plan should address problems with lean body mass that may appear in older subjects. Feng et al.10 demonstrated that patients with sarcopenia (both young and middle-aged) are more prone to MASLD (12.75%) than those without it (3.73%). The current approach to MASLD emphasizes exploration of the disease together with patient comorbidity, such as arterial hypertension, type 2 diabetes, or metabolic syndrome, among others, and the hygienic-dietary plan must be adaptable and flexible to different situations and needs of each patient, considering the various metabolic conditions that are involved. Therefore, there is no diet better than another, but nutritional intervention should be personalized depending on each patient’s characteristics.
Relevant clinical outcomes
It is undeniable that the Mediterranean diet has consolidated as the fundamental pillar in the management and reversal of MASLD, given its significant impact on hepatic and clinical parameters. This type of dietary regimen stands out for its sustained long-term benefits. Jurek et al.1 demonstrated, in a 2-year controlled clinical trial conducted in patients with MASLD, that greater adherence to the Mediterranean diet, combined with the promotion of physical activity, led to significant improvements in reducing intrahepatic fat content, fatty liver index (FLI), and plasma cytokeratin-18 levels, in addition to achieving progressive reduction of the dietary inflammatory index. This study evidenced improvement in liver enzymes, particularly in sustained decrease of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and gamma-glutamyl transferase (GGT), key indicators of liver function and cellular damage, reflecting hepatocellular recovery and reduction of liver inflammation1.
The benefits of the Mediterranean diet are mediated by its composition rich in monounsaturated fats, polyphenols, and omega-3 fatty acids, being considered a balanced diet, as its elements contribute to the modulation of oxidative stress and chronic inflammation, in addition to improving lipid and glucose metabolism. Yamazaki et al.11 conducted an analysis of diet diversity and color vividness in patients with MASLD compared to a balanced diet, and demonstrated that lower variation in chromaticity and lower average chromatism (indicating less colorful and less varied diets) are associated with elevated ALT levels and hepatic fibrosis. From these observations, it is suggested that monotonous eating patterns, with few variations, are associated with deficient nutrient and dietary fiber intake, and therefore with progression and severity of liver disease11.
It is important to highlight that food-based interventions have achieved significant reductions in anthropometric parameters such as body weight, body mass index (BMI), fat mass, and waist circumference, fundamental in controlling obesity and its metabolic complications1.
Other dietary strategies, including time-restricted feeding and low-calorie diets or those with specific approaches, such as the DASH diet or vegetarian diet, have also shown positive effects on metabolic parameters, although with less robust evidence and shorter follow-up duration. However, long-term sustainability and impact appear to be superior with the Mediterranean diet, positioning it as the nutritional intervention of choice in MASLD1,6.
In summary, Mediterranean-style dietary intervention has demonstrated that, in the long term, it achieves relevant clinical improvements in patients with MASLD, evidenced by reduction of hepatic steatosis, decrease of biochemical liver markers of inflammation, and control of metabolic comorbidity. Dietary intervention is a non-pharmacological therapeutic strategy that constitutes a fundamental pillar in the comprehensive management of MASLD1,6.
Types of diet in metabolic-associated steatotic liver disease
In this review, we focus on dietary models with the most evidence, as well as models currently under study: the Mediterranean diet, modified Mediterranean diets, the DASH diet, low-fat diets, low-carbohydrate diets, the ketogenic diet, time-restricted feeding, and plant-based diets. Figure 2 shows the characteristics of the main diets most studied in MASLD.
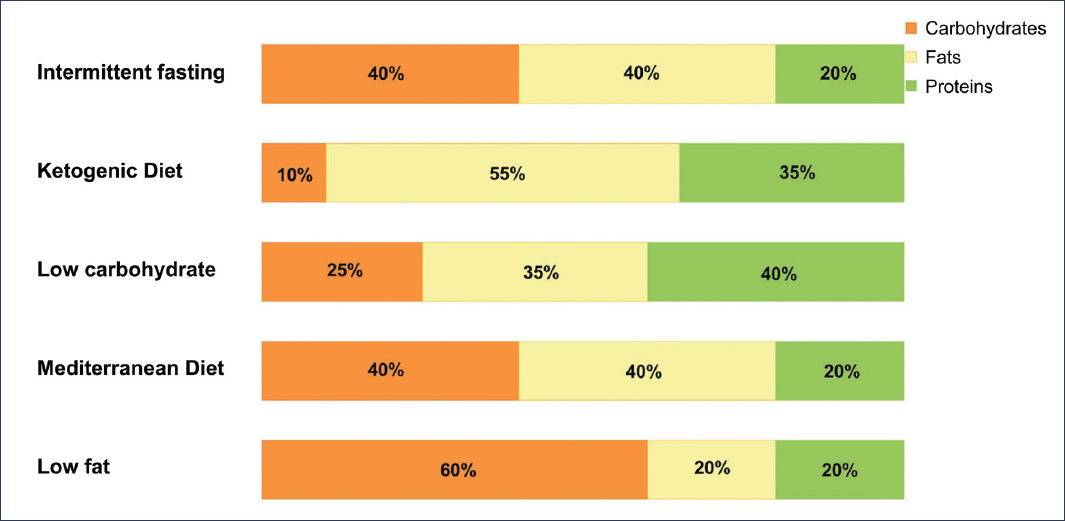
Figure 2. Distribution of macronutrients in different types of diets for MASLD. Macronutrients are presented as relative percentage of total energy. It is recommended that the distribution of macronutrients in the time-restricted diet adjust to the Mediterranean diet, although it may vary.
Mediterranean diet and modified Mediterranean diets
Dietary interventions within the framework of the Mediterranean diet have demonstrated significant efficacy in the management of MASLD. Being among the most effective and most studied approaches, it is characterized by a higher lipid intake than other dietary approaches, emphasizing unsaturated fatty acids and regular fiber intake, which contributes to its capacity to improve clinical conditions related to MASLD, nutritional status, body composition, and inflammation, including significant reduction of ALT; it also has the potential to block or slow the progression of MASLD to more severe stages1,3,12,13. It is characterized by high consumption of vegetables (fruits, vegetables, breads and other minimally refined cereals, potatoes, beans, nuts, and seeds); minimally processed, fresh seasonal and locally grown foods; fresh fruits as typical dessert, with sweets containing sugars or honey a few times per week; high consumption of olive oil (especially virgin and extra virgin olive oil) used as the main fat source; moderate intake of dairy products (mainly cheese and yogurt); eggs, fish, and poultry consumed in low to moderate amounts; red meats consumed in low amounts; and wine in moderation, consumed with meals1,14. Prospective observational studies support an inverse association of MASLD with the Mediterranean diet or overlapping dietary patterns. Clinical trials testing the effect of classic or modified Mediterranean diets (low-carbohydrate or polyphenol-enriched) support their beneficial effect on hepatic steatosis. The two largest and longest-duration (18 months) randomized clinical trials provide the best evidence of the valuable role of two modified Mediterranean diets. Gepner et al.15 demonstrated the superiority of the low-carbohydrate Mediterranean diet over the low-fat diet in terms of decreasing intrahepatic fat. Yaskolka Meir et al.16 found that the Mediterranean diet enriched with polyphenols and more strictly restricted in red and processed meat led to twice the hepatic fat loss compared to a regular isocaloric Mediterranean diet, despite achieving similar weight reduction. In both studies15,16, it was demonstrated that nutritional intervention is superior to standard nutritional counseling. The use of the Mediterranean diet, as well as its modified versions, low-carbohydrate Mediterranean diet and low-fat Mediterranean diet, has also resulted in significant reductions in liver enzymes, including ALT and AST, as well as fatty liver index (FLI) and fibrosis-4 index (FIB4)1,4,16–18.
Low-carbohydrate diets
Low-carbohydrate diets also effectively reduce hepatic steatosis and improve insulin resistance. Additionally, emerging dietary strategies, such as intermittent fasting and very low-energy ketogenic therapy, are being explored for their potential to reduce weight and fat mass and insulin resistance, improving metabolic health, emphasizing their effectiveness in improving hepatic outcomes associated with MASLD, as well as reducing inflammatory status3.
Low-carbohydrate diets can be classified into three categories according to intake. A moderate or reduced carbohydrate diet could be defined as a carbohydrate intake of 26% to 45% of total calorie intake per day, or > 130 g/day (and less than approximately 250 g/day on a 2000-calorie diet, for example). When carbohydrate intake is < 26% or > 30 g/day, it is classified as a low-carbohydrate diet. A step further, when carbohydrate intake is < 10%, or < 30 g/day, it can be categorized as a ketogenic or very low-carbohydrate diet. As the proportion of carbohydrate intake decreases, the proportion of fat and protein increases, so diets called “high-protein” or “high-fat” are generally reduced or low-carbohydrate diets. In general, despite the short-term superiority of the low-carbohydrate diet, in the long term, both appear to be equally effective in reducing hepatic fat and ALT as long as 7% weight loss is achieved19.
Dietary intervention, especially increasing dietary protein along with overall body weight control, has proven to be a promising strategy to decrease the impact of MASLD. Dietary proteins have been shown to reduce fat accumulation in the liver by increasing hepatic metabolism, generating satiety, increasing insulin sensitivity, and improving muscle mass retention, which together contribute to weight control. Both animal and plant proteins have benefits; however, plant proteins (mainly from legumes, nuts, and seeds) have shown greater metabolic advantages, which highlights the importance of obtaining sufficient protein from readily available and sustainable food sources. Furthermore, protein-rich diets, such as the Mediterranean diet, have been shown to delay the probability and progression of MASLD20,21.
Low-fat diets
A low-fat diet restricts the amount of energy obtained from fat sources. Typically, a low-fat diet limits energy from fat to no more than 30% of total daily calories22. Low-fat or low-carbohydrate diets offer alternative strategies, with evidence suggesting that reducing carbohydrate intake can reduce intrahepatic lipid accumulation, triglyceridemia, lipid oxidation, and insulin resistance1. A systematic review conducted by Jurek et al.1 found a clinical trial performed in patients with MASLD, in which hepatic triglyceride content decreased by 25% after 12 weeks of low-fat diet (p < 0.01), independent of body weight loss and caloric intake. In another meta-analysis conducted by Ahn et al.19, in an 18-month weight loss study with 139 participants with abdominal obesity or dyslipidemia, comparing a low-fat diet versus a Mediterranean or low-carbohydrate diet, found that a low-fat diet resulted in a 3.8% reduction in hepatic fat content (p < 0.001). In this study, the authors demonstrated that decreases in the proportion of hepatic fat content appear to act as a stronger mediator of favorable effects of the low-fat diet on some cardiometabolic markers than overall visceral fat reduction. It is important to note that in one of the previous studies, hepatic fat content reduction was greater in the Mediterranean or low-carbohydrate diet group (p < 0.05). Despite the findings, more recent studies suggest that a low-carbohydrate diet might be more suitable than a low-fat diet for treatment1,19.
Ketogenic or very low-carbohydrate diet
The very low-carbohydrate ketogenic diet pattern is characterized by restricting carbohydrate intake to < 30-50 g daily (equivalent to 13-25% of total calories). This diet has a high fat content, representing between 60 and 70% of total calories, and a protein amount ranging between 0.8 and 1.2 g per kg of ideal body weight. On the other hand, the very low-carbohydrate and very low-calorie ketogenic diet is defined by a carbohydrate content < 50 g daily, protein intake of 1-1.5 g per kg of ideal body weight, and fat intake of 15-30 g per day, with a total caloric intake of approximately 500-800 calories daily. It is a highly restrictive diet, and guidelines establish that it should be under the supervision of a health professional and limited to 8 to 12 weeks, followed by a gradual increase in calories, carbohydrates, and food variety. Some studies have investigated the efficacy of the very low-carbohydrate ketogenic diet in patients with overweight and obesity, but long-term, high-quality evidence is limited. This diet was an effective strategy in some studies in terms of weight loss, visceral fat reduction, and improvement of metabolic parameters and inflammatory markers; however, conclusive results have not been obtained in long-term randomized clinical trials on its effect in MASLD4.
In the meta-analysis conducted by Ahn et al.19, it was found that neither the low-carbohydrate diet nor the very low-carbohydrate diet is better for improving hepatic steatosis or transaminase levels compared to a low-fat diet. Furthermore, there is evidence that, beyond the distribution of macronutrients in the diet, the type of calories can be considered. Regarding carbohydrates, the glycemic index, related to postprandial glycemic response, can be a useful tool in management. In this context, studies conducted by Ramon-Krauel et al.23 and Parker A et al.24 incorporated a low glycemic index diet into nutritional interventions and observed a positive effect on hepatic fat composition. In addition to promoting weight reduction, Kirk et al.25 observed that these diets can also have positive effects on liver disease by reducing insulin levels, lipogenesis, and fatty acid oxidation19,23,27
DASH diet
The DASH diet has been used as a lifestyle approach to treat and prevent hypertension. It consists of a diet low in saturated fats, high in protein, fiber, and minerals, and low in sodium. The DASH eating plan is based on fruits, vegetables, low-fat dairy products, fish, whole grains, poultry, nuts, seeds, and legumes, while reducing consumption of fat, red meat, and products with added sugar. The DASH diet has been shown to decrease all-cause mortality, cardiovascular disease, diabetes, and cancer; therefore, this dietary approach has proven to be promising for improving metabolic and hepatic parameters1,26.
Hekmatdoost et al.28 found an inverse relationship between adherence to the DASH diet and MASLD risk; subjects in the highest quartile of the DASH diet scale had a 30% reduction in risk (odds ratio [OR]: 0.70; 95% confidence interval [95% CI]: 0.61-0.80). Consistent with these results, in a review conducted by Torres et al.26, a long-term study that evaluated a large cohort was found, and after two decades of follow-up, showed that being in the highest tertile of DASH diet adherence was associated with lower risk (OR: 0.57-0.77). Evidence from some observational studies and one clinical trial23,28 supports that DASH dietary patterns could be a preventive tool; however, more clinical trials are needed to evaluate with greater depth the findings from a heterogeneous population of observational studies23,28.
Time-restricted feeding
Time-restricted feeding, also called “intermittent fasting,” is a nutritional strategy in which calories are consumed in a defined time window and involves various forms of time restriction4. The term “intermittent fasting” is used to describe various methods of energy restriction, ranging from alternating feeding and fasting periods to total food abstinence or very low energy consumption23.
Alternate-day fasting consists of consuming food normally for 24 hours and fasting for the next 24 hours. The 5:2 fasting consists of drastically reducing caloric intake for 2 days (approximately 500 calories per day), followed by 5 days of normal consumption. On the other hand, periodic fasting consists of intermittent fasting for more than 2 days with minimal caloric intake (≤ 500 calories) without repeated fasts. Time-restricted fasting consists of eating only during certain hours of the day (e.g., from 12 p.m. to 8 p.m.). It is hypothesized that intermittent fasting may have metabolic benefits in the liver, independent of caloric restriction and weight loss; this may also improve MASLD histology18.
In diets like intermittent fasting, patients may have the impression that during the eating window, also called “feast hour (or day),” they can eat whatever they want, regardless of whether it is healthy or not. This can lead to excessive consumption of sugars, saturated fats, and ultra-processed foods. Furthermore, there may be excess saturated fat and meat, especially red and processed meat, in low-carbohydrate and ketogenic diets. At minimum, unprocessed or very minimally processed fish and poultry should be preferred4.
Time-restricted feeding limits the daily food intake period to 8-10 hours most days of the week. This nutritional strategy shifts the daily proportion between feeding and fasting periods without necessarily reducing total daily caloric intake. However, in some studies (two randomized controlled trials and a single-group trial), an involuntary reduction in energy intake of 250 to 600 calories was observed. Periodic fasting showed a reduction of hepatic steatosis in 697 participants in a prospective observational study of subjects with and without type 2 diabetes4,16. Johari et al.29 demonstrated that with an alternate-day caloric restriction intervention for 8 weeks (fasting day, 30% of caloric requirement; non-fasting day, ad libitum), compared to a normal habitual diet (no intervention) as control, both hepatic steatosis and fibrosis scores were significantly reduced in age-adjusted body mass (AABM) compared to controls (both p < 0.01). A randomized controlled trial conducted by Holmer et al.30 in patients with MASLD compared three interventions for 12 weeks. Time-restricted feeding was in the form of a 5:2 diet, in which 2 non-consecutive days per week participants consumed 500 kcal/day for women and 600 kcal/day for men, and maintained a generally healthy diet on the remaining 5 days of the week. Time-restricted feeding was superior to standard dietary instructions for reducing weight and steatosis but was equally effective as a regular low-carbohydrate, high-fat diet4,29,31
Intermittent fasting includes different methods of energy restriction: time-restricted fasting, alternate-day fasting, and prolonged fasting. The daily time-restricted feeding regimen, with an 18-hour fasting period and a 6-hour feeding window (16/8), and alternate-day fasting, characterized by 24 hours of fasting at 25% of basal energy, have recently gained attention as possible interventions to improve management of metabolic conditions. Although they could be beneficial, the best intermittent fasting method for individuals with MASLD remains unknown1,4. The use of time-restricted feeding, including the 16/8 protocol, has reported improvements in liver function, demonstrating a reduction in liver enzymes (AST and ALT)23. More evidence is needed, with results and conclusions from long-term studies, to confirm the beneficial impact of intermittent fasting variations on the onset and evolution of MASLD.
Plant-based diets
Plant-based diets focus on high intake of plant foods and derivatives (i.e., cereals, legumes, vegetables, and fruits), and low intake of animal-origin foods. The vegetarian diet partially overlaps with the plant-based diet, while the vegan diet is characterized by total abstinence from animal products. Plant-based diets offer the benefit of preventing chronic diseases, but their effect on MASLD has not been demonstrated in long-term randomized clinical trials13.
Despite growing interest in plant nutrition in clinical care, highlighting health benefits, and plant diet strategies, evidence linking these diets with hepatic health is lacking, as research focused on the relationship between plant diet quality and hepatic complications, such as steatosis and fibrosis, is still limited. In a large cross-sectional study, adherence to plant-based diets, especially those prioritizing healthy plant foods, was associated with a lower probability of hepatic fibrosis, in addition to finding that high fructose consumption in plant-based diets was associated with a greater risk of hepatic steatosis. However, few studies have highlighted the relationship between plant-based diets and hepatic steatosis, with controversial results13,32,33.
It is important to differentiate between fructose sources. Whole fruits, rich in fiber and polyphenols, could have protective effects, while refined or concentrated sources could promote hepatic fat accumulation34. The vegetarian diet, in particular, has gained attention due to its potential to reduce hepatic fat, decrease systemic inflammation, and improve insulin sensitivity. However, despite growing interest, evidence of its impact on MASLD remains limited. Even considering these potential benefits, the use of plant-based diets should be approached with caution, particularly in people with insulin resistance, as high carbohydrate intakes from certain plant sources can lead to hepatic fat accumulation. Firm conclusions cannot be drawn about the relative merits of vegetarian versus Mediterranean diets in terms of outcomes in patients with MASLD. The optimal macronutrient composition (carbohydrates, fats, proteins) for MASLD management remains unclear, particularly when comparing a low-carbohydrate diet and a low-fat diet. While the Mediterranean diet is well studied in MASLD, the long-term effectiveness and adherence of other dietary patterns, such as the DASH diet, require further exploration, also in comparison with the Mediterranean diet. Furthermore, most studies have been conducted over a few months, which creates a gap in knowledge regarding the long-term effects of specific dietary models in MASLD16.
Diets oriented in the Mexican context
Some international health organizations are promoting traditional diets to combat increasing non-communicable diseases and obesity rates worldwide. Traditional diets are generally healthy diets because their core consists of plant-origin foods, such as grains, legumes, vegetables, fruits, and tubers, with low consumption of animal-origin foods35,36.
In an effort to define the traditional Mexican diet, Valerino-Perea et al.35 conducted a systematic review of 61 scientific documents and found that representative food groups and their components are as follows: 1) grains and tubers, such as corn, amaranth, rice, wheat, cassava, potato, and sweet potato; 2) corn products, such as tortilla, tamales, and atole; 3) legumes, mainly beans; 4) vegetables such as squash, chayote, nopales, maguey, tomato, tomatillo, and quelites; 5) fruits such as anona, capulin, guava, jicama, mamey, plums, prickly pear, and sapote; 6) meats from turkey, chicken, duck, venison, rabbit, and beef; 7) herbs and condiments, such as chile, epazote, vanilla, salt, and onion; 8) oils and fats from avocado, pumpkin seed, and chia seeds; 9) beverages of chocolate and pulque (fermented maguey beverage); 10) insects, such as grasshoppers, maguey worms, ants and their larvae, chicatanas, and escamoles; and 11) sweets and sweeteners, such as honey, sugar, and sugar cane. It is important to note that some of the foods mentioned are not of Mexican origin, but were adopted from other countries and even from other continents35.
Regionalized diet
In a randomized clinical trial conducted by Cano et al.37 in patients with MASLD, those with moderate to severe steatosis demonstrated by ultrasound, aged 18 to 70 years, of Mexican nationality, and without alarm signs were selected. Participants were divided equally into two groups: the Mediterranean diet and the regional Mexican diet. It was found that the Mediterranean diet and the regional Mexican diet are equally effective in improving liver disease symptoms, particularly hepatic steatosis, which highlights the viability of using regionally adapted diets for fatty liver treatment37. The regional Mexican diet was characterized by wild vegetables, such as quelites, edible flowers, local fruits, whole grains (corn derivatives, amaranth), tubers (sweet potato), beans, healthy oils and fats, peanuts, chia, sunflower, pumpkin, avocado, and greater consumption of white meats.
Milpa diet
Milpa is a traditional agricultural production system in Mesoamerica, particularly in Mexico, that has been used since pre-Hispanic times. The milpa diet, rich in proteins, fiber, vitamins, and bioactive compounds, aligns with recommendations for MASLD. It provides greater dietary diversity and contributes to sustainable production. One of its limitations is that the availability of described foods varies according to the Mesoamerican region; most of them are available in the central regions of America, such as Mexico. However, there is little information on this topic, and most of the data found are related to individual components and not to the overall dietary pattern, as is the case with the Mediterranean diet, which has solid evidence38 (Table 1).
Table 1. Dietary interventions and better metabolic outcomes
| Comparison of dietary models in MASLD | Study characteristics | AST | ALT | GGT | CAP | LSM | FLI | Fib4 | Liver pathology staging | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| MED vs. LFD | 27 male participants with overweight/obesity Diagnosis: ultrasound Age 27-42 years Randomization 1:1 14 MED vs 13 LFD Duration: 12 Weeks | p = 0.017 | p = 0.128 | p = 0.224 | NA | NA | p = 0.021 | NA | NA | Ristic-Medic et al.45 (2020) |
| TMD vs. LCMD vs. LFMD | 63 participants with insulin resistance and obesity Diagnosis: ultrasound Age 18-65 years Duration: 12 weeks Simple random sampling Group 1 TMD: 40-45% C, 15-20% P, 35-40% L Group 2 LCMD: ≤ 35% C, 15-20% P, > 45% L Group 3 LFMD: ≥ 55% C, 15-20% P, 20-25% L | p = 0.010 | p = 0.410 | p = 0.010 | NA | NA | p = 0.003 | p = 0.020 | NA | Kestane and Bas46 (2024) |
| TRF 16/8 vs. CN | 45 participants TRF group: 22 CN group: 23 Age: 18-50 years Hepatic steatosis grade 2 (CAP) Randomized controlled trial Duration: 12 weeks | p = 0.010 | p = 0.013 | p = 0.026 | p = 0.009 | p < 0.001 | NA | NA | NA | Kord-Varkaneh et al.47 (2023) |
| DASH vs. LCD | 40 participants with obesity DASH diet group: 20 Control group: 20 Double-blind RCT Duration: 8 weeks | p = 0.008 | p = 0.149 | NA | NA | NA | NA | NA | NA | Rooholahzadegan et al.48 (2023) |
| CR vs. ND | 60 participants Randomized controlled clinical trial Age: 20-60 years Diagnosis: ultrasound CR group: 20 Control group: 30 Duration: 12 weeks Diets contained 17% P, 30% L, and 53% C | p = 0.020 | p = 0.010 | NA | NA | NA | NA | NA | p = 0.350 | Asghari et al.49 (2022) |
| LOV-D vs. SWL-D | 75 participants with overweight or obesity Randomized clinical trial Duration: 12 weeks | p = 0.08 | p = 0.04 | NA | NA | NA | NA | NA | NA | Garousi et al.50 (2023) |
ALT: alanine aminotransferase; AST: aspartate aminotransferase; CAP: Controlled Attenuation Parameter; CN: normal control diet; CR: calorie-restricted diet; DASH: Dietary Approaches to Stop Hypertension; FLI: fatty liver index; GGT: gamma-glutamyl transferase; C: carbohydrates; L: lipids; LCD: low-calorie diet; LCMD: low-carbohydrate Mediterranean diet; LFD: low-fat diet; LFMD: low-fat Mediterranean diet; LOV-D: lacto-ovo-vegetarian diet; MASLD: Metabolic-associated steatotic liver disease; MED: Mediterranean diet; ND: normal diet; P: proteins; SLM: Standard Lifestyle Modification; SWL-D: standard weight-loss diet; TMD: Traditional Mediterranean Diet; TRF: time-restricted feeding 16/8.
Education, prevention, and patient empowerment
Guidelines recommend implementing medical-nutritional treatment through comprehensive nutritional education programs aimed at promoting self-care39.
Approaches for lifestyle modification
Lifestyle modification approaches use diverse intervention contents, such as assessment of participants’ lifestyle behaviors, information, education, goal setting, counseling, and feedback40,41. Education about lifestyle modification in patients with MASLD can be delivered in various ways; some are more traditional, such as an individual consultation with a health professional. From a practical point of view, personalized dietary counseling and patient food education are essential to ensure long-term adherence to a healthy eating pattern. Other forms of education involve more modern approaches, such as web programs or the use of smartphone applications. Regardless of the method, it is important to achieve weight loss goals by improving diet quality and increasing physical activity. Various methods and their combinations can be useful in this case20.
Mobile technologies for MASLD
Several studies have demonstrated the importance of using mobile technologies to modify the lifestyle of patients with MASLD. In a randomized controlled trial42, it was found that the use of a special mobile application in the intervention group demonstrated a significantly higher probability of achieving weight loss ≥ 5% at 6 months, with a relative risk of 5.2 (95% CI: 1.8-15.4; p = 0.003). Improvements occurred in anthropometric indices, such as waist circumference and body weight, along with reductions in liver enzyme levels. Patients with MASLD may lack motivation or adherence to disease management; interventions using eHealth with diverse contents would motivate their behavior and ultimately improve sustainable management20,40,42.
A study conducted in Thailand demonstrated that providing information about healthy lifestyles and education about MASLD through a social media application significantly improves liver stiffness in patients with MASLD compared to standard of care, highlighting the potential of social media platforms to improve clinical outcomes in MASLD management43.
With more than 25% of patients seeking dietary advice through platforms like YouTube, the quality and reliability of information are fundamental. However, the disparity in educational value and engagement metrics between professional and non-professional content remains unexplored. In a study conducted by Tur44, the dual challenge of ensuring high-quality health information while simultaneously maximizing interaction on digital platforms is emphasized. The findings point to the need for collaboration between health professionals and content creators to provide scientifically rigorous knowledge that is, at the same time, accessible. Through the adoption of narrative techniques and greater regulatory oversight, the quality and accessibility of online dietary guidance can be improved, ultimately favoring better health outcomes in patients with MASLD44.
Conclusions
Nutritional intervention is fundamental in the management of MASLD, with 7-10% weight loss being crucial to improve steatosis and reverse fibrosis in early stages. The Mediterranean diet is currently the dietary pattern with the most evidence; it stands out for its abundance of monounsaturated fats, polyphenols, and omega-3, which manage to modulate inflammation and improve hepatic metabolism. However, the clinical heterogeneity and associated comorbidity in patients with MASLD require an individualized nutritional approach, adapted to the genetic, metabolic, and cultural characteristics of each patient to ensure long-term adherence. Other strategies, such as low-carbohydrate diets, the DASH diet, vegetarian diets, and intermittent fasting, also show benefits, but with less evidence. We must consider that regionalized diets, such as the traditional Mexican diet and the milpa diet, offer viable alternatives adapted to the cultural context. Finally, nutritional education and the use of mobile technologies are key tools to improve adherence and clinical outcomes in patients with MASLD.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
A.D. Cano-Contreras is a speaker for Medix. M.R. Francisco declares no conflict of interest for conducting this research work.
Ethical considerations
Protection of human and animal subjects. The authors declare that the procedures followed conformed to the ethical standards of the responsible human experimentation committee and in accordance with the World Medical Association and the Declaration of Helsinki. The procedures were authorized by the institution’s Ethics Committee.
Confidentiality, informed consent, and ethical approval. The authors have followed the confidentiality protocols of their institution and have approval from the Ethics Committee. The recommendations of the SAGER guidelines have been followed, according to the nature of the study.
Statement on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.