Introduction
Liver cancer is currently the sixth most common cancer worldwide and the third leading cause of cancer death1. The most recent studies report that metabolic dysfunction-associated steatotic liver disease (MASLD) is the cause of hepatocellular carcinoma (HCC) with the greatest global increase2. MASLD-related HCC (MASLD-HCC) has particular characteristics that distinguish it from HCC of other etiologies, such as a higher frequency in patients without liver cirrhosis, larger tumors, more advanced stage at diagnosis, older age, and higher prevalence of obesity and other metabolic comorbidity3–5. Due to these characteristics, current screening strategies appear to be suboptimal in these patients6. In addition, the treatment of MASLD-HCC presents some additional challenges, especially when diagnosed in advanced stages7.
The objective of this narrative review is to analyze and synthesize the available evidence on MASLD-HCC, addressing epidemiology, risk factors, pathogenesis, diagnosis, treatment, prevention, and future perspectives.
Method
A narrative review of the literature on the relationship between MASLD and HCC was conducted. To this end, an exhaustive bibliographic search was performed in PubMed/MEDLINE, Embase, and Cochrane Controlled Register of Trials (CENTRAL), covering publications from 2005 to 2025. The search strategy combined MeSH terms and free keywords: “cirrhosis,” “hepatocellular carcinoma,” “MASLD,” “MASH,” “NAFLD,” and “NASH.” The selection of studies was based on their thematic relevance and clinical value, prioritizing systematic reviews with or without meta-analyses, followed by randomized controlled trials, non-randomized intervention studies, observational studies (case-control, cross-sectional, cohorts), and finally, case series or reports and expert opinions. In addition, a manual screening of the reference lists of relevant articles was performed to complement the electronic search. Given the narrative nature of this review, formal systematic review criteria were not applied; instead, available evidence was synthesized in a critical and structured manner, with the aim of offering a comprehensive and updated view of the topic.
Epidemiology and global burden
MASLD is the most common chronic liver disease, with a global prevalence in the general population that has increased from 25% in 2016 to more than 30% in the most recent reports8,9. The clinical spectrum of the disease encompasses simple steatosis, metabolic dysfunction-associated steatohepatitis (MASH), liver fibrosis, cirrhosis, and even HCC10. It has been estimated that only 20% of individuals with MASLD have MASH, and 20% of these will develop cirrhosis over an average of 2 to 4 decades11.
Liver cancer is currently the sixth most common cancer worldwide and the third leading cause of cancer death1, with an incidence that is increasing, with the possibility of 1.4 million people diagnosed with liver cancer by the year 204012. HCC represents 90% of liver cancers, and most cases arise in the context of chronic liver disease, with liver cirrhosis being the main risk factor for its development13, with up to 30 times greater risk compared to patients without cirrhosis14.
MASLD is the fastest-growing cause of HCC worldwide2. MASLD-HCC represents approximately 10% of all HCC cases15. Using information from the United Network for Organ Sharing (UNOS) database for 2022, Koh et al.16 reported that MASLD is the most frequent cause of HCC among liver transplant candidates in the United States of America, surpassing chronic hepatitis C. In this registry, MASLD-HCC represented 24.3% of HCC cases on the waiting list. Using dynamic Markov modeling, it is estimated that the prevalence of MASLD-HCC by the year 2030 will increase by up to 130% in the United States of America17.
The annual incidence of HCC in patients with MASLD cirrhosis is currently 0.5% to 2.6%15; however, using the predictive model mentioned above, it has been predicted that the incidence by 2030 could increase by up to 122% in the United States of America17. On the other hand, the frequency of MASLD-HCC is significantly lower in patients without fibrosis, with an incidence ranging from 0.8 to 1.2 per 1000 person-years in patients with simple steatosis and MASH without fibrosis, respectively18.
Risk factors for HCC in MASLD
There are numerous risk factors related to the development of HCC in MASLD, from those associated with the progression of chronic liver disease, such as liver fibrosis, in which carcinogenesis is similar to that of HCC of other etiologies19, to those that lead to the activation of pathophysiological mechanisms not related to fibrosis progression, such as metabolic alterations or chronic low-grade inflammation that result in direct and indirect mutations of oncogenic factors, and which are postulated to be the cause of the greater susceptibility to develop HCC19,20.
Liver fibrosis and cirrhosis
Hepatic inflammation (steatohepatitis) is the main driver for the development of liver fibrosis in MASLD, and the degree of fibrosis constitutes the most important prognostic factor for disease progression and its complications, including the development of HCC. A systematic review and meta-analysis demonstrated that the risk of HCC increases progressively according to the fibrosis stage21. Advanced fibrosis, defined as METAVIR stage ≥ 3, has been associated with the development of hepatic decompensation (ascites, variceal hemorrhage, and hepatic encephalopathy), liver transplantation, mortality, and HCC22–25.
As already mentioned, the proportion of patients with HCC without cirrhosis is notably higher in MASLD-HCC, with studies reporting frequencies ranging from 20% to 63%4,26,29 A systematic review and meta-analysis that included 19 studies with 168,571 patients reported a prevalence of HCC in MASH without cirrhosis of 38%, compared to 14.2% in patients with liver disease of other etiologies (p < 0.001), with a higher probability of developing HCC (odds ratio [OR]: 2.61; 95% confidence interval [95% CI]: 1.27-5.35; p = 0.009)3.
Obesity
Obesity is the most widely described risk factor for MASLD. Visceral fat is one of the most important components in the pathophysiology of the disease19. The presence, duration, and severity of obesity have been related to risk of liver disease progression, as well as fibrosis progression30, development of hepatic decompensation31, and up to a fourfold increase in HCC risk (OR: 4.3; 95% CI: 2.1-8.4), also reporting a synergistic relationship between obesity and other risk factors, such as alcohol and tobacco consumption32. Other studies have corroborated these results; Borena et al. reported that body mass index (BMI) is associated with a relative risk of primary liver cancer of 1.39 (95% CI: 1.24-2.58)33, while in a multivariate logistic regression analysis, Archambeaud et al.34 reported that obesity (BMI ≥ 30 kg/m2) is an independent risk factor for HCC development.
Type 2 diabetes mellitus
Type 2 diabetes mellitus (T2DM) is the second most important metabolic alteration in MASLD. The presence of T2DM has been associated with HCC development regardless of liver disease etiology19,35. In a European study that included 136,703 patients with MASLD or MASH, T2DM constituted the main factor associated with a tripled risk of HCC (hazard ratio [HR]: 3.51; 95% CI: 1.72-7.16)36. Another study conducted in the United States of America reported an even higher HCC risk (HR: 4.2; 95% CI: 1.2-14.2; p = 0.02), without a significant association between HCC risk and other metabolic factors such as BMI, dyslipidemia, or arterial hypertension37. Conversely, in the study by Kanwal et al.38, the risk increased progressively with the number of metabolic alterations (T2DM, hypertension, dyslipidemia, and obesity), reporting a risk of developing HCC or liver cirrhosis up to twice as high in patients with T2DM, obesity, dyslipidemia, or hypertension (HR: 2.6; 95% CI: 2.3-2.9). It has also been reported that T2DM duration has an impact on HCC risk, with greater risk in patients with T2DM of more than 10 years’ duration (adjusted OR: 2.2; 95% CI: 1.2-4.8)39.
Alcohol consumption
Alcohol has been shown to be an important risk factor for MASLD progression, with studies indicating that a small (5-9 drinks/week) to moderate (10-13 drinks/week) amount significantly increases the risk of liver fibrosis, with an additive and dose-dependent effect as the number of cardiometabolic risk factors increases40. In addition, alcohol consumption has been associated with HCC development in patients with liver cirrhosis of any etiology, with an increase of up to six times the risk of HCC (OR: 5.7; 95% CI: 2.4-13.7)32 and with a synergistic effect when combined with obesity (BMI ≥ 30 kg/m2) (HR: 3.82; 95% CI: 1.94-7.52; p < 0.01)41. Regarding MASLD-HCC, Kimura et al.42 demonstrated that even mild alcohol consumption (< 20 g/day) is associated with an increase in the rate of HCC occurrence compared to patients without alcohol consumption (6.5% vs. 1.4%; p = 0.02); the effect of alcohol consumption contributed significantly to HCC development, especially in patients with advanced fibrosis (F3-4). A meta-analysis that included 31 studies and more than 1 million individuals reported that alcohol consumption was associated with a 140% increase in MASLD-HCC risk (2.41 OR; 95% CI: 1.03-5.65)43.
Tobacco consumption
Tobacco consumption has been associated with a higher risk of HCC in patients with liver cirrhosis of any etiology, including MASLD43,44. The results of the systematic review by Abdel-Rahman et al.44, which included 81 epidemiological studies, report a combined OR for HCC development in current smokers of 1.55 (95% CI: 1.46-1.65; p < 0.00001), in former smokers of 1.39 (95% CI: 1.26-1.52; p < 0.00001), and in heavy smokers of 1.90 (95% CI: 1.68-2.14; p < 0.00001). In the aforementioned meta-analysis43, tobacco consumption was associated with a 30% higher risk of MASLD-HCC (1.3 OR; 95% CI: 1.08-1.57). More recently, a retrospective cohort study that included only MASLD patients reported similar results, with a higher incidence of HCC in those with current or previous tobacco consumption45.
Pathogenesis of HCC development in patients with MASLD
Oncogenesis in MASLD is a consequence of multiple deregulated pathways that contribute to hepatocellular damage and liver fibrosis development19. MASLD is characterized by lipid accumulation that causes lipotoxicity, that is, hepatocellular damage secondary to altered fat metabolism, resulting in excessive free fatty acid production that promotes reactive oxygen species formation and induces endoplasmic reticulum stress, mitochondrial damage, and gene transcription, activating inflammatory signaling pathways and causing a state of chronic inflammation that eventually leads to fibrogenesis20,46. These alterations contribute to carcinogenesis mediated by oxidative DNA damage and accumulation of mutations in genes such as FOXO1, CIDEB, and GPAM47. Multiple aberrant oncogenic pathways have been implicated in MASLD-HCC tumorigenesis, such as Wnt/β-catenin signaling, phosphatidylinositol-3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/Akt/mTOR), Myc, Hedgehog signaling, and epithelial-mesenchymal transition, which has been associated with metastasis and cell migration46.
Other pathophysiological mechanisms involved are angiogenesis, with increased expression of CD34 and vascular endothelial growth factor (VEGF), as well as disruption of the immune system with depletion of CD8+ T cells and CD4+ T cells, with possible implications for cancer immunotherapy response47.
Diagnosis of HCC in patients with MASLD
MASLD-HCC has particular characteristics that distinguish it from other causes of HCC. In addition to its occurrence in the absence of cirrhosis3, it has been reported that patients with mild to moderate fibrosis (F0-2 stages) present larger tumors (93 ± 52 vs. 53 ± 35 mm; p < 0.001) compared to patients with MASLD and advanced fibrosis (F3-4 stages), as well as a higher frequency of not being candidates for liver transplantation according to Milan and University of California criteria (73% vs. 36% and 66% vs. 25%, respectively)4. A systematic review and meta-analysis that included 61 studies and 3631 patients reported that those with MASLD-HCC were older, had higher BMI, and presented more metabolic comorbidity compared to those with HCC due to other etiologies, in addition to larger tumor diameter, with a mean difference of 0.67 cm (95% CI: 0.35-0.98; p = 0.0087), and a higher probability of uninodular lesions (OR: 1.36; 95% CI: 1.19-1.56; p = 0.0003). Despite this, no significant differences were observed between both groups in terms of the distribution by stages of the BCLC (Barcelona Clinic Liver Cancer) system, the TNM (Tumor, Node, Metastasis) system, functional status according to the ECOG (Eastern Cooperative Oncology Group) scale, the treatment provided, or survival5. More recently, analysis of an Italian database (ITA.LI.CA) has reported similar results: patients with MASLD-HCC were more frequently men, with a higher average age and with lower frequency of liver cirrhosis (84% vs. 94%) and clinically significant portal hypertension (75% vs. 82%)48. Regarding tumor burden, there was a higher frequency of large tumors (27% vs. 21%) and with extrahepatic metastases (7% vs. 3%). However, despite having a more advanced tumor stage at diagnosis, patients with MASLD-HCC had a lower risk of HCC-related death, although they did show a significantly higher risk of non-HCC-related mortality (p = 0.006)48.
Figure 1 describes the characteristics of MASLD-HCC.
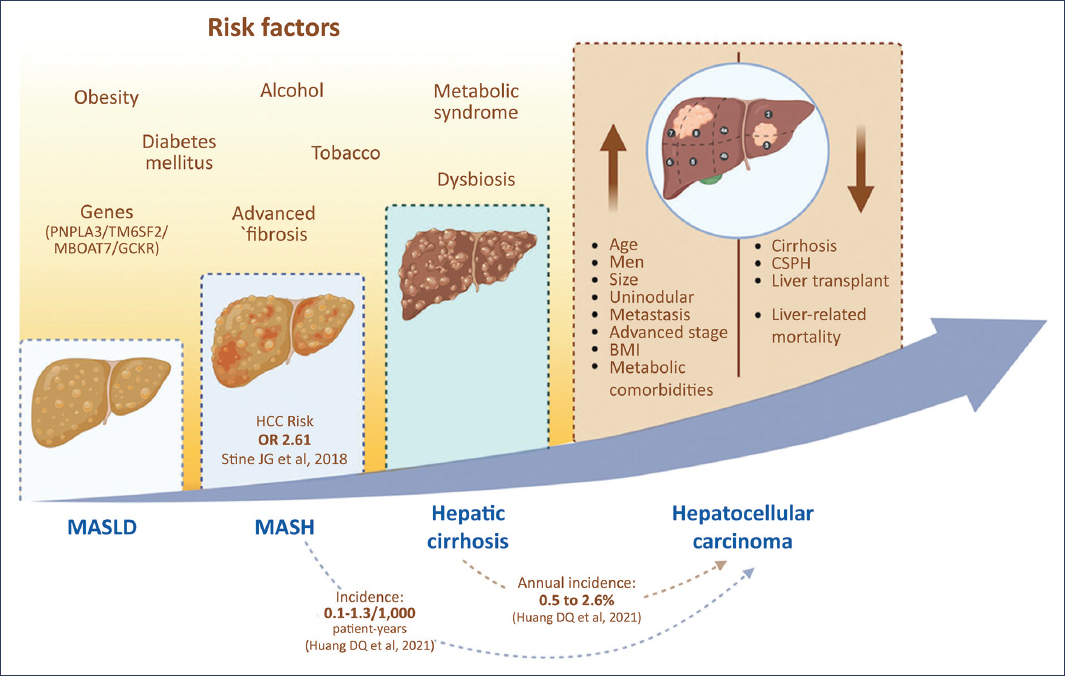
Figure 1. Risk factors and clinical characteristics of MASLD-HCC. CSPH: clinically significant portal hypertension; BMI: body mass index; MASLD: metabolic dysfunction-associated steatotic liver disease; MASH: metabolic dysfunction-associated steatohepatitis.
Because liver function in MASLD-HCC is usually preserved, a significant proportion of patients are not in a screening program, and HCC diagnosis is often incidental20,46. Current guidelines establish that HCC diagnosis can be made non-invasively through imaging studies, either by magnetic resonance imaging (MRI) or multiphase contrast-enhanced computed tomography (CT) in patients with liver cirrhosis or chronic hepatitis B49, observing arterial phase hyperenhancement (APHE) and subsequently contrast washout and capsule presence in the portal-venous or delayed phases, reporting results according to the standardized terminology of the LI-RADS (Liver Imaging Reporting and Data System) system50. If MRI and CT are not available, contrast-enhanced ultrasound may be used, although it is a tool with limited utility for staging. Invasive diagnosis by liver biopsy should be reserved for patients without cirrhosis or in cases of diagnostic doubt49.
As with HCC of other etiologies, HCC diagnosis in patients with MASLD cirrhosis can be established through standard imaging studies (MRI or CT). A systematic review and meta-analysis that included 170 patients demonstrated that those with MASLD-HCC have a similar frequency of APHE, although with a lower frequency of washout and capsule; the overall estimated percentages of APHE, washout, and capsule were 94.0% (95% CI: 89.1-96.7), 72.7% (95% CI: 63.3-80.4), and 57.5% (95% CI: 45.1-69.1), respectively51. Compared to CT, MRI showed a higher pooled percentage of capsule (67.1% vs. 44.7%; p = 0.02)51. Similarly, studies conducted in patients without cirrhosis have demonstrated that the radiological appearance of HCC on CT and MRI is typical in more than 80% of cases, with no significant differences in the imaging patterns of patients with and without cirrhosis52, suggesting that non-invasive criteria should be extended to this group of patients. In this regard, Barat et al.53 conducted a retrospective study that demonstrated the applicability of the LI-RADS diagnostic system (v2018) in patients with MASLD-HCC, finding no significant differences between MRIs of patients with MASLD and patients with chronic viral hepatitis.
Management of HCC in patients with MASLD
Current HCC management is based on the BCLC staging system, which classifies patients into five stages (0, A-D) considering tumor burden, liver function, and cancer-associated health status, proposing an evidence-based therapeutic approach updated with surgical options, locoregional treatments, and systemic treatment options for patients in advanced stages54. The treatment of HCC in MASLD presents some additional challenges, as mentioned previously, since patients with MASLD have a higher prevalence of cardiometabolic comorbidity and are usually diagnosed in more advanced stages, so therapeutic options could be limited46.
Management of HCC in early stages (BCLC 0-A)
Regarding early stages, curative therapies include surgical resection, liver transplantation, and ablative treatments. Various studies have evaluated the efficacy of these therapies in patients with MASLD. A meta-analysis of 17 retrospective studies reported that patients with MASLD-HCC may have higher overall survival (HR: 0.87; 95% CI: 0.75-1.02) and recurrence-free survival (HR: 0.93; 95% CI: 0.84-1.02) after hepatectomy, as well as a similar rate of perioperative complications and mortality to patients with HCC of other etiologies55. Studies that have evaluated the effectiveness of ablative therapies (radiofrequency or microwave) in patients with MASLD-HCC report that there is no difference in terms of survival, morbidity, or tumor recurrence after ablation56,57.
MASLD has become the second most frequent cause of liver transplantation in the United States of America58. It has been postulated that patients with MASLD may have a higher rate of peri- and post-transplant complications due to the higher prevalence of cardiovascular disease59; however, a recent meta-analysis of 10 comparative studies with 51,761 patients demonstrated that survival at 1, 3, and 5 years, as well as disease-free survival after liver transplantation, is similar in patients with MASLD-HCC and those with HCC of other etiologies60. In conclusion, studies suggest that, despite the increase in comorbidity prevalence, including cardiovascular disease, and the risk of larger tumors, there is no difference in the application of curative therapies in patients with MASLD-HCC5, although some studies report up to a 47% lower probability of receiving any curative treatment due to the more advanced stage at diagnosis56.
Management of HCC in intermediate stage (BCLC B)
Locoregional therapies are the treatment of choice for patients with intermediate-stage HCC with well-defined lesions and preserved portal flow54. These include TACE (transcatheter arterial chemoembolization) and TARE (transcatheter arterial radioembolization). A retrospective study demonstrated that TACE is equally effective in patients with MASLD-HCC compared to patients with HCC of other etiologies, despite having larger lesions (4.9 ± 5.8 vs 3.1 ± 2.4 cm), and without an increase in complication rate61. Studies that have evaluated the effectiveness and safety of TARE in patients with MASLD-HCC report similar results in terms of safety, overall survival, and local or hepatic progression-free survival62,63.
Management of advanced HCC (BCLC C)
The management of advanced HCC is based on the administration of systemic treatment with immunotherapy using immune checkpoint inhibitors (ICI)54. Based on the results of the IMbrave15064 and HIMALAYA65 clinical trials, current treatment guidelines establish that the combinations atezolizumab-bevacizumab and durvalumab-tremelimumab are the first-line options49,54. According to the update of the BCLC therapeutic algorithm for 2025, the combination of nivolumab plus ipilimumab is incorporated as an additional immunotherapy option in first-line in selected clinical scenarios, a recommendation based on the results of the CheckMate 9DW trial, which demonstrated improved overall survival compared to sorafenib, along with greater durability of response, supporting the role of dual immune blockade (PD-1/CTLA-4) in the treatment of advanced HCC. (Reig M, et al. BCLC 2025 strategy for prognosis prediction and treatment allocation in hepatocellular carcinoma. J Hepatol. 2025.)
The results of experimental and retrospective studies, and even a meta-analysis of randomized clinical trials, initially suggested that HCC etiology could influence the response to immunotherapy7,66. The mentioned meta-analysis reported that ICIs were significantly more effective in viral hepatitis-associated HCC than in HCC of other etiologies. Conversely, liver disease etiology did not appear to influence the efficacy of tyrosine kinase inhibitors or anti-VEGF therapies7. This lower treatment response described in patients with MASLD-HCC could be related to tumor infiltration by CCR2+ and CX3CR1+ macrophages, as well as impaired “functional fitness” of T lymphocytes, which could contribute to therapeutic resistance to ICIs47. In addition, it has been described that HCC is usually an immunologically “cold” tumor, that is, an immunodeficient tumor or one with impaired innate immune functions, with low PD-L1 expression, low mutational burden, decreased expression of molecules involved in antigen presentation, and limited T lymphocyte infiltration67.
Apart from a possible lower efficacy, the use of systemic treatment in these patients could be conditioned by the presence of metabolic syndrome, since arterial hypertension and cardiovascular disease can be aggravated with the use of these drugs20. However, more recent studies have provided opposite results. Thus, a post hoc analysis of the IMbrave150 study did not identify significant differences in terms of objective response evaluated with RECIST and modified RECIST criteria, or in progression-free survival or overall survival, between patients with MASLD-HCC and those with HCC of other etiologies68.
In agreement with these results, a retrospective study that included 295 patients treated with atezolizumab–bevacizumab showed that the number of cardiovascular risk factors had no impact on overall survival or progression-free survival69. In this same vein, a recent meta-analysis that included the CheckMate 459, KEYNOTE-240, IMbrave150, COSMIC-312, HIMALAYA, LEAP-002, RATIONALE-301, and camrelizumab–rivoceranib clinical trials demonstrated that immunotherapy offers a significant survival advantage in both viral and non-viral etiology HCC, with the HIMALAYA trial showing the greatest benefit with ICI, highlighting the importance of CTLA-4 inhibition in this patient subgroup70. These findings appear more robust, since in the initial studies, the reported results were based on subanalyses, without defining what percentage of patients with non-viral cirrhosis were truly MASLD patients7. To date, there are no randomized controlled clinical trials specifically designed to evaluate the response to immunotherapy in patients with MASLD-HCC71, so first-line treatment continues to be that recommended by the most recent clinical guidelines.
Prevention of HCC in patients with MASLD
Current guidelines recommend HCC screening in all patients with liver cirrhosis, regardless of etiology. If liver function permits oncological treatment, screening should include abdominal ultrasound every 6 months with or without determination of serum alpha-fetoprotein concentrations49; however, the performance of abdominal ultrasound may be suboptimal in patients with obesity, which is one of the most prevalent metabolic factors in this disease6, with a decrease in sensitivity and specificity due to possible inadequate visualization of the liver parenchyma in patients with MASH47. To date, systematic screening in patients without advanced fibrosis is not justified72,73. In addition to ultrasound, biomarkers such as the fibrosis-4 index (FIB-4, with a cutoff point > 3.2)26 and increased liver stiffness assessed by elastography74 are associated with increased HCC risk.
Various risk stratification models for HCC in patients with liver cirrhosis have been designed, some of them specific for MASLD. The model by Ioannou et al.75, based on seven predictors (age, sex, T2DM, BMI, platelet count, serum albumin, and aspartate aminotransferase to alanine aminotransferase ratio), showed an area under the ROC curve (AUROC) of 0.75. Another Italian model confirmed that the combination of parameters such as the PNPLA3 genotype, liver function tests, and the presence of portal hypertension was useful for estimating HCC risk76. More recently, Lambrech et al.77 developed the APAC score, based on age, soluble platelet-derived growth factor receptor beta, alpha-fetoprotein, and creatinine, with good predictive capacity for HCC in patients with MASLD (AUROC 0.75)35.
Future perspectives and precision medicine
The upward trend in MASLD prevalence is a key determinant of the future global burden of MASLD-HCC17. As described previously, MASLD-HCC presents specific characteristics that make it a diagnostic and therapeutic challenge7, which leads to the imminent need to implement programs and improve HCC screening tools in patients with MASLD and advanced fibrosis, with the integration of serum biomarkers and even the application of ultrasound models based on artificial intelligence (machine learning) to detect MASLD-HCC in earlier stages20, as well as prospective studies on the response to immunotherapy treatment in the different phenotypes of MASLD19.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical considerations
Protection of human and animal subjects. The authors declare that no experiments were conducted on humans or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data or require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.
