Introduction
In the first half of the last century, the first case reports appeared of patients with fatty liver who also had metabolic disorders such as type 2 diabetes mellitus (T2DM), obesity, or dyslipidemia, but did not consume large quantities of alcohol1. Since then, this condition has received different names; in 1980, Ludwig et al.2 called it nonalcoholic steatohepatitis to distinguish it from alcoholic steatohepatitis, and thus this disease gained its own identity. As more has become known about it, its names have changed, and it appears to be a disease in search of its own name; it is currently known as MASLD (metabolic dysfunction-associated steatotic liver disease)3.
Over the last 60 years, cases have multiplied worldwide. The paradox is that “progress” has favored a harmful lifestyle; now the excessive consumption of refined sugars and saturated fats is promoted on the one hand, and extreme sedentarism on the other. This combination exposes the population to diseases such as obesity, T2DM, arterial hypertension (AH), and dyslipidemia, which are components of metabolic syndrome and have a close relationship with MASLD.
With the name changes came changes in diagnostic criteria as well; however, the clinical outcome of NAFLD (non-alcoholic fatty liver disease), MAFLD (metabolic associated fatty liver disease), and MASLD is very similar4, as confirmed by a study in the Mexican population that revealed a 97% concordance of diagnostic criteria between NAFLD and MASLD, and 100% between MAFLD and MASLD, establishing that the results of NAFLD and MAFLD studies can be extrapolated to MASLD5.
Global epidemiology of metabolic dysfunction-associated steatotic liver disease
Prevalence
A recent meta-analysis compared global prevalence in the periods 1990-2006 and 2016-2019, and reported an increase of more than 50% between the two periods (25.26% and 38%, respectively); in Europe it was 25.1%, in Latin America 44%, and in childhood from 7% to 14%6.
Global prevalence for the year 2019 showed an alarming increase in cases in North Africa and the Middle East (42.62%), in North America (38.47%), and in Latin America (34.45%) (Table 1)6. Regarding sex, prevalence in men is 40% and in women 26%7.
Table 1. Worldwide prevalence of NAFLD in general population, over 20 years of age, in the year 2019
| Region | Prevalence (95% CI) |
|---|---|
| Western Europe | 32.47 (19.1-49.4) |
| United States | 38.47 (18.4-50.1) |
| Latin America | 34.4 (18.2-55.2) |
| North Africa and Middle East | 42.6 (26.9-59.9) |
| Global | 32.16 (18.4-50.1) |
95% CI: 95% confidence interval.
Modified from Younossi et al.6
In children, an autopsy study (n = 742) in California reported hepatic steatosis in 9.6% of the total sample and in 38% of children with obesity8. Other studies with hepatic ultrasound, in children with obesity or food insecurity and Latino ancestry, reported MASLD prevalences of 4.8% and 17%9–11.
Between 10% and 30% of subjects with steatosis can develop steatohepatitis or MASH (metabolic dysfunction-associated steatohepatitis), and global prevalence is estimated at 5.7% (± 2.63), with greater presence in Latin America (7.11% ± 3.55), particularly in patients with high rates of obesity or T2DM12,13.
Incidence
The incidence of MASLD is difficult to estimate due to the variability of samples and diagnostic methods used. Globally, it is 48.89 per 1000 persons/year; in Sri Lanka, South Korea, and Germany, the highest figures were found, with 80, 54, and 50 cases per 1000 persons/year, respectively6. In the analysis of the periods 1994-2006 and 2010-2014, it was found that there was a 58% increase, with figures of 37.41 and 59.11 cases per 1000 persons/year, respectively6.
Risk factors
MASLD is a very heterogeneous disease. By definition, it is associated with at least one cardiometabolic criterion, but can have more than one; thus, there are patients with T2DM and AH, or only with obesity, or with T2DM and dyslipidemia, or other combinations. Therefore, patients have differences in their clinical profile, lifestyle, and comorbidity, which determine the particular pathophysiology of each case and, consequently, the personalized treatment that corresponds. This is why personalized care must begin at the primary care level, with the identification of risk factors in each case. That is where effective and timely intervention must occur to achieve the best results. That is where the largest number of people at risk is concentrated, and the cost of care and prevention is not so high, and the possibilities of success are greater; on the contrary, when the disease progresses, comorbidity and its complications increase, interventions are more costly, and the results are not entirely satisfactory (Fig. 1).
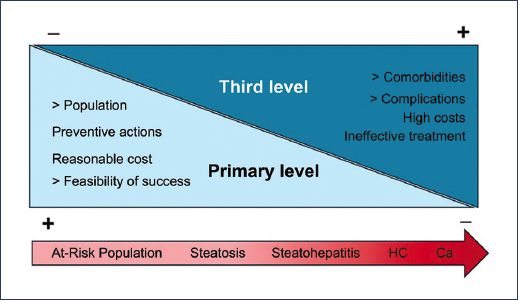
Figure 1. There are more opportunities for intervention at the primary care level
Lifestyle
In almost every part of the world, traditional diets were changed to a diet now dominated by ultra-processed foods, where excess saturated fats and refined sugars. For the food industry, it is more profitable to replace sucrose with fructose in almost all processed foods; the drawback is that fructose metabolism is different and its abuse promotes insulin resistance, T2DM, AH, obesity, dyslipidemia, dysbiosis, MASLD, and systemic inflammation that induces neoplasms14. At the same time, consumption of fiber, polyunsaturated fats, omega-3 fatty acids, and vitamins was reduced.
On the other hand, sedentarism has been encouraged. People walk less and outdoor recreational activities have been reduced; now mobility is inhibited, and we spend many hours in front of a screen, whether for work or in leisure activities, especially children15. Motorized vehicles have diversified, and having one, two, or more cars in a family is a sign of comfort and status16. The long-term effect has been regrettable, as sedentarism reduces energy expenditure and increases body fat deposits, cardiovascular risk, and MASLD.
Metabolic syndrome
MASLD has a bidirectional cause-effect interaction with the components of metabolic syndrome; it is the hepatic expression of metabolic syndrome and, according to the new nomenclature, all patients have at least one cardiometabolic risk criterion3 corresponding to the components of metabolic syndrome (Fig. 2). To the extent that a patient accumulates more components of metabolic syndrome, their risk of developing MASLD is greater, and their prognosis will be worse17.
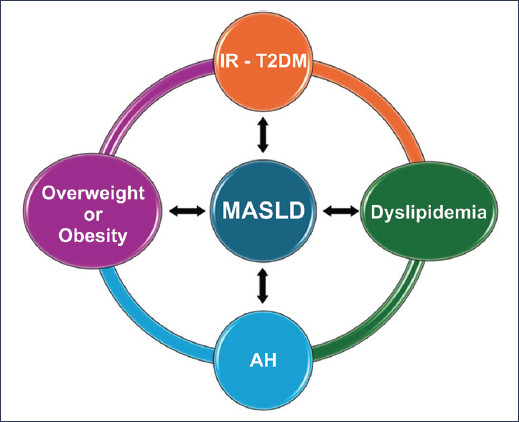
Figure 2. Bidirectional correlation between metabolic dysfunction-associated steatotic liver disease (MASLD) and components of metabolic syndrome. T2DM: type 2 diabetes mellitus; AH: arterial hypertension; IR: insulin resistance.
Type 2 diabetes mellitus
The prevalence of T2DM is increasing alarmingly worldwide: from 1980 to 2019, cases went from 108 to 463 million people. The worst is that the association of T2DM and MASLD is common in children and adults, because they share pathophysiological mechanisms such as insulin resistance and abnormal triglyceride metabolism18.
T2DM induces fibrosis and hepatocellular carcinoma in patients with MASLD19, and since 80% of them have insulin resistance, its timely detection is a priority at the primary care level20.
Obesity
Obesity has reached epidemic proportions due to lifestyle changes. It is now the most frequent condition in many patients with MASLD and, together with T2DM, is the two main promoters of fibrosis, poor evolution, more complications, and death in these patients21. In recent decades, a parallel increase in cases of obesity and MASLD has been observed in all age groups.
Arterial hypertension
AH per se is also associated with greater progression to hepatic fibrosis; the prevalence of AH is higher in patients with advanced fibrosis, and these have the highest number of cardiovascular complications and deaths²¹. Therefore, it is recommended that patients with AH and any other cardiometabolic risk factor be evaluated with a more complete screening to assess the possibility of MASLD21.
Dyslipidemia
Dyslipidemia is another component of metabolic syndrome. It is characterized by elevated triglycerides and low levels of high-density lipoprotein cholesterol; in these cases, atherogenic lipoprotein subparticles, small low-density proteins, increase cardiovascular risk, which is the leading cause of death in patients with MASLD22.
Genetics
Although MASLD affects a large percentage of the world population, its distribution is not homogeneous and some ethnic groups are more affected than others. Several epidemiological studies conducted in the United States of America, where the population is very diverse, showed that subjects of Hispanic origin, and very particularly Mexicans, had the highest prevalence, with 58%, followed by Caucasians with 44% and African Americans with 35%23. This peculiarity has been associated with a genetic predisposition related to the PNPLA3 and TM6SF2 genes, which are involved in hepatic lipid metabolism24; however, these are not the only ones and there are others with similar effects, shown in table 2. The prevalence of the PNPLA3 I148M genetic variant in the Mexican population is 78%, one of the highest in the world25.
Table 2. Main genes associated with the pathogenesis and progression of MASLD
| Gene | Function | Effect |
|---|---|---|
| PNPLA-3 | Lipid metabolism | MASLD, MASH, fibrosis, and hepatocellular carcinoma |
| TM6SF2 | VLDL and cholesterol secretion and mobilization | MASLD, MASH, and fibrosis |
| GCKR | De novo lipogenesis, regulates glycemia | MASLD, MASH, and fibrosis |
| MBOAT7 | Phosphatidylinositol remodeling | MASLD, MASH, and fibrosis |
| IGFBP2 | Insulin-like growth factor transport | MASLD |
| miR-122 | Regulates lipid metabolism and fibrosis | MASLD, MASH, fibrosis, and hepatocellular carcinoma |
VLDL: very low-density lipoproteins.
Martínez-López et al.28
Age
Global prevalence of MASLD increases significantly from 50 years of age onwards, although there may be some regional differences6. Naturally, with the passage of years, exposure to metabolic disorders that induce steatosis and liver damage increases and accelerates the progression of fibrosis, hepatocellular carcinoma, and death; that is why it is considered that after 50 years, the risk of poor prognosis of MASLD increases24.
Male sex
Initially, MASLD was considered to be more frequent in women, since the first cases were mostly in females; however, current evidence confirms the predominance in men, and male sex is considered a risk factor not only for MASLD, but also for the development of fibrosis, HC, and death. Figures are very variable; a North American series with 5532 individuals reported an odds ratio (OR) of 1.3 with a 95% confidence interval (95% CI) of 1.05-1.61 (p = 0.019)26.
Menopause
Although MASLD is more frequent in men, in women, from menopause onwards, the risk increases 2.4 times, which, added to age > 50 years, increases the risk of developing fibrosis, even in women with normal weight. This condition is attributed to a protective effect of estrogens before menopause, which physiologically declines at this stage27.
Dysbiosis
The functions of the intestinal microbiota are very diverse and necessary to maintain homeostasis; therefore, when an imbalance or dysbiosis occurs, there can be repercussions at different levels.
It has been shown that a diet rich in fructose and saturated fats alters the microbiota, damages the intestinal barrier, produces local low-grade inflammation, and increases intestinal permeability14. All this facilitates bacterial translocation and increases nutrient absorption; direct tissue damage occurs from bacterial endotoxins that flow freely through the portal system and increase fat deposits in the liver, resulting in steatosis and steatohepatitis, which are the initial phases of MASLD14,24.
Social determinants
Social determinants of health create complex pathways through which socioeconomic inequalities translate into disease disparities. Individuals from low socioeconomic strata have higher rates of MASLD due to limited access to health care, poor dietary quality, and few opportunities for physical activity, while in high-income countries, MASLD cases are due to excessive consumption of ultra-processed foods and sedentary lifestyles29. Educational level is also important; some studies have correlated higher cardiometabolic risk and advanced fibrosis with lower educational levels29.
Food insecurity creates a paradoxical situation of “excess in scarcity”, which favors more MASLD and fibrosis compared to those with food security, operating through irregular dietary patterns, dependence on energy-dense but nutritionally deficient foods, and increased systemic inflammation30.
Risk factors in childhood
MASLD in childhood and adolescence is associated with obesity and other elements of metabolic syndrome, as in adults10,31. However, it can also occur in those with a normal body mass index (BMI), but who have dyslipidemia and insulin resistance32. Other factors associated with MASLD in pediatric age are maternal obesity33 and reduced muscle mass34,35.
Epidemiology in Mexico
Epidemiological studies of MASLD in Mexico are limited, and, as in the rest of the world, when the populations studied and diagnostic methods are different, the results tend to yield discordant results.
In 2020, the research fund of the Mexican Association of Gastroenterology (AMG), in collaboration with the Institute of Medical-Biological Research of the Universidad Veracruzana, sponsored an epidemiological study on MAFLD in 585 volunteers from the open population36. Family and personal history of metabolic dysfunction, physical examination, anthropometry with body composition analysis, nutritional and physical activity assessment, socioeconomic status, laboratory tests, hepatic ultrasound, and Fibroscan® were evaluated. A representative sample size of the general population was calculated; some of its characteristics are presented in table 3.
Table 3. Significant differences of patients with and without MAFLD, in a Mexican population
| Characteristics | Without MAFLD | With MAFLD | p; OR; 95% CI |
|---|---|---|---|
| Male sex | 49.7% | 50.3% | p < 0.006; OR: 1.67; 95% CI: 1.14-2.43 |
| Female sex | 62.2% | 37.8% | |
| Average age | 49 | 50 | |
| Overweight | 37.6% | 29.8% | 97.7% with BMI > 25 |
| Obesity | 30.6% | 66.9% | |
| BMI | 27.3 | 31.9 | p < 0.001 |
| Abdominal perimeter | 88.5 cm | 102 cm | p < 0.001 |
| Visceral fat | 2.5Kg | 3.4 Kg | p < 0.001 |
| Poor or very poor diet | 37.3% | 46.3% | p < 0.03; OR: 1.43; 95% CI: 1.01-2.02 |
| Metabolic syndrome | 27.4% | 62.4% | p < 0.001; OR: 1.82; 95% CI: 1.18-2.81 |
| Arterial hypertension | 20.9% | 36.9% | p < 0.00001; OR: 2.27; 95% CI: 1.54-3.34 |
| Hypercholesterolemia | 17.6% | 23.8% | p < 0.05; OR: 1.49; 95% CI: 0.97-2.28 |
| Hypertriglyceridemia | 36.5% | 64.3% | p < 0.00000; OR: 3.26; 95% CI: 2.28-4.67 |
| Type 2 diabetes mellitus | 8.8% | 21.3% | p < 0.00001; OR: 2.86; 95% CI: 1.71-4.77 |
BMI: body mass index; OR: odds ratio; 95% CI: 95% confidence interval.
Modified from Bernal-Reyes et al.36
Among the most important results is an updated prevalence with MASLD criteria of 42.5%37, one of the highest in the world. A higher prevalence was observed in men from 50 years of age onwards, sedentary, with poor eating habits and obese. Male sex, age > 50 years, metabolic syndrome, and elevated alanine aminotransferase were identified as risk factors. The prevalence of the disease in non-obese subjects was 3.3%. A worrying fact is that almost 40% of cases with MAFLD already had advanced fibrosis (F3-F4) by Fibroscan®.
Impact of nomenclature changes
The multiple changes in the nomenclature of fatty liver are due to the need for a definition that adheres to the most current knowledge. The current denomination, MASLD, emphasizes three fundamental elements:
- – The damage: steatosis.
- – The organ: liver.
- – The cause: metabolic dysfunction.
More than the name, it is the changes in diagnostic criteria that determine some modification in the epidemiological profile of patients. For example, initially it was said that nonalcoholic steatohepatitis, whose core component was steatohepatitis, was more frequent in women and of low prevalence (10-15%)6; later, with the change to NAFLD, the spectrum was expanded beyond steatohepatitis and cases with steatosis without another known cause were included, and even though subjects with alcohol consumption > 20 g/day for women and > 30 g/day for men were excluded, prevalence increased with variations between 17% and 46%, with predominance in males38.
Years later, with the change to MAFLD, emphasis was placed on the metabolic background of the disease, and those who had other chronic liver diseases, including alcoholic liver disease, were included. By then, prevalence already reached 30% of the world population, more as a global epidemiological phenomenon than as a result of the new denomination.
When NAFLD series are compared against MAFLD, prevalence is slightly higher in MAFLD, concordance between both is greater than 75%, and the risk of death is greater in MAFLD (2.4 times)39, because the diagnostic criteria include older patients with more comorbidity.
Comparative analyses of NAFLD, MAFLD, and MASLD series are limited, but they consistently show that MASLD prevalence is slightly higher and that it predominates in men. In a 27-year analysis, all-cause mortality was higher in MAFLD and MASLD, with 1.18 (95% CI: 1.04-1.33) and 1.19 (95% CI: 1.06-1.34), respectively, compared to NAFLD40.
In the AMG study35, the diagnostic criteria of NAFLD, MAFLD, and MASLD were compared, and it was found that 97.6% of patients with NAFLD criteria met MASLD criteria, and all those with MAFLD met MASLD criteria, with very good correlation (NAFLD – MAFLD: r = 0.955 and p < 0.0001; NAFLD – MASLD: r = 0.979 and p < 0.001; MAFLD – MASLD: r = 0.970 and p = 0.0001) and concordance (NAFLD and MAFLD: kappa = 0.954; NAFLD and MASLD: kappa = 0.974; MAFLD and MASLD: kappa = 0.975)5.
For now, there is little evidence of significant differences between NAFLD, MAFLD, and MASLD regarding the presence of CVD, HC, and malignant neoplasms39.
Socioeconomic impact
MASLD is a systemic disease with multiple comorbidities, whose care costs are high and increasing. Figures show that MASLD is more prevalent in developing countries, including Mexico, where changes in diet and lifestyle have been more pronounced, while in developed countries prevalence is lower, which is attributed to a better educational level and more effective public health strategies41. On the other hand, it is not ruled out that in the poorest countries, due to their deficient health infrastructure, there may be underreporting of cases.
In countries with the highest prevalences of MASLD, such as Mexico, the population most affected is the one with low income and food insecurity, in whom the consumption of ultra-processed foods of poor nutritional quality is habitual, nutritional information is scarce, and preventive programs are ineffective42.
Until now, the impact of drugs such as glucagon-like peptide-1 (GLP-1) receptor agonists and sodium-glucose cotransporter-2 (SGLT2) inhibitors has been minimal in the global cost of the disease, and if the current trend of obesity is not reversed, by 2039 the cost of care will have almost doubled43.
It has been calculated that resmetirom, which was recently approved by the Food and Drug Administration, would have a cost per patient of $66,700 with a gain of 1.24 quality-adjusted life years, so restricted prescription is now recommended only to gastroenterologists and hepatologists44.
In 2023, in the United States of America, the annual cost of caring for patients with MASLD was $45 billion, and in 20 years could reach $890 billion, while in Europe $35 billion euros of annual expenditure are estimated45. To these figures must be added the costs of premature deaths and work disabilities, both of the patients and their caregivers.
Impact on quality of life
MASLD is a systemic disease accompanied by various comorbidity that affects quality of life. The components of metabolic syndrome are the most common diseases, but there are others, such as polycystic ovary syndrome, psoriasis, hypothyroidism, obstructive sleep apnea, chronic kidney disease, CVD, and malignant neoplasms, to mention some of the most frequent, of deleterious course.
In a review from 1990 to 2019, it was observed that, in parallel with a global increase in the prevalence of MASLD, disability days and the number of deaths also increased significantly46. Regarding this trend, there is concern among international health organizations in countries such as Mexico, India, Qatar, Australia, and the United States of Americ47.
MASLD alone causes few symptoms; the most common in these patients are those related to their multiple comorbidities. However, fatigue can be disabling and has been reported in up to 33%. It has also been associated with depression (OR: 16.2; 95% CI: 6.02-43.6), CVD (OR: 3.64; 95% CI: 2.04-6.51), and death (hazard ratio [HR]: 2.31; 95% CI: 1.37-3.89)45
There are few studies that have analyzed the quality of life of patients with MASLD. In one of them, the HRQOL (health related quality of life) questionnaire was applied to 3333 patients with NAFLD and 5982 healthy controls. Twenty-two percent of the patients said their health was poor or fair, compared to only 10% of the control group, and reported deterioration of physical health 68.3% versus 61.3% (p < 0.01) and deterioration of mental health 62.6% versus 64%; the percentage of days in the last month in which some alteration of physical or mental health prevented carrying out daily activities was 84.4% versus 79.8%. After adjusting for age, sex, race, and BMI, it was concluded that patients with NAFLD had greater impairment in their quality of life, with an 18% to 20% greater likelihood of seeing their daily activities affected by deterioration of their health (p < 0.0001)48. As expected, deterioration in quality of life increases as the disease progresses and fibrosis advances49.
Metabolic dysfunction-associated steatotic liver disease as a public health problem
The main problem of MASLD is its high prevalence, which goes hand in hand with the increase in cases of T2DM and obesity. Today, it affects 38% of the world population6. In Mexico, it affects at least 43% of adults, cases continue to increase37, and it is already the most frequent and fastest-growing chronic liver disease; however, very little has been done about it50.
Morbidity
According to the new nomenclature, patients with MASLD have at least one cardiometabolic risk factor that expresses diseases such as T2DM, AH, dyslipidemia, and obesity. Additionally, association with other pathologies such as HC, chronic kidney disease, and cancer is common. Table 4 shows the risk of the different clinical outcomes in patients with MASLD.
Table 4. Meta-analysis of 129 longitudinal studies that evaluated the risk of incidence of clinical outcomes in people with MASLD
| Clinical outcomes | Hazard ratio | 95% CI | p |
|---|---|---|---|
| Hepatocellular carcinoma | 4.37 | 1.79-10.67 | < 0.01 |
| Metabolic syndrome | 2.57 | 1.13-5.85 | 0.02 |
| Type 2 diabetes mellitus | 2.56 | 2.10-3.13 | < 0.01 |
| Arterial hypertension | 1.75 | 1.46-2.08 | < 0.01 |
| Cancer in general | 1.54 | 1.35-1.76 | < 0.01 |
| Cardiovascular disease | 1.43 | 1.27-1.6 | < 0.01 |
| Chronic kidney disease | 1.38 | 1.27-1.50 | < 0.01 |
95% CI: 95% confidence interval.
Modified from Chan et al.59
Metabolic syndrome
The prevalence of MASLD in people with metabolic syndrome is 73%, while the prevalence of metabolic syndrome among those with MASLD is 43%. In these cases, the prevalence of advanced fibrosis is higher, and when all components of metabolic syndrome are present, it can affect up to 30% of patients51–53.
Type 2 diabetes mellitus
In a large meta-analysis, it was established that the global prevalence of MASLD in patients with T2DM is close to 70% and in Eastern Europe can reach 80%54, while the prevalence of T2DM in patients with MAFLD is 28.3% (95% CI: 25.2-35.6), more than double the global prevalence, which is estimated at just over 10%; the highest figures are reported in Colombia (50.4%), Brazil (42%), and Mexico (32.2%), and the lowest in Nordic countries (< 15%). The incidence is 26.9 per 1000 persons/year18.
Overweight and obesity
Recent reports indicate that the cumulative prevalence of MASLD in overweight people is 70%, and in obese people it can reach up to 75%, while MASH occurs in 34% equally. On the other hand, the presence of hepatic fibrosis has been documented in 20% of overweight patients and in 22% of obese patients; in these cases, 7% had advanced fibrosis (F3-F4)12,55.
Arterial hypertension
Between 40% and 60% of patients with MASLD may have AH39. The incidence of AH in early stages of MASLD has been reported at 6.5 per 100 persons/year, while in advanced stages it reaches 14.5 per 100 persons/year, when the patient is already in the cirrhosis phase²¹. A retrospective study with more than 270,000 patients with MASLD showed that those who had AH and dyslipidemia had a risk of progression to cirrhosis or cancer of 1.8 compared to subjects without cardiometabolic risk factors56.
Dyslipidemia
Prevalence estimates vary widely. Some reports from Eastern countries establish a prevalence of up to 50% in patients with NAFLD57, but in Western countries, it is usually not so high.
In the AMG epidemiological study, in Mexico, hypertriglyceridemia was found in 36.5% of the control group and in 64.3% of cases with MAFLD (95% CI: 2.28-4.67; p < 0.00000), and hypercholesterolemia in 17.6% of the control group and 23.8% of cases with MAFLD (95% CI: 0.97-2.28; p < 0.05)37.
Chronic kidney disease
The incidence of chronic kidney disease increases with the degree of hepatic fibrosis because these patients experience a systemic inflammatory state; activation of proinflammatory cytokines and release of reactive oxygen species cause structural kidney damage, and glomerulosclerosis and interstitial fibrosis develop58.
In a review of more than 12,000 people from the NHANES III (Third National Health and Nutrition Examination Survey) database, the prevalence of chronic kidney disease could be related to the degree of hepatic fibrosis, evaluated with the NFS (NAFLD Fibrosis Score), as shown in table 5.
Table 5. Prevalence of chronic kidney disease and degree of fibrosis in patients with MAFLD (NHANES III)
| Degree of fibrosis (NFS) | Chronic kidney disease (%) |
|---|---|
| F0-F1 | 19 |
| F1-F2 | 18 |
| F2-F3 | 37 |
| F3-F4 | 53 |
Adapted from Sun et al.58
Malignant neoplasms
Malignant neoplasms present as a relatively frequent complication in patients with MASLD; in fact, they are the second leading cause of death. The digestive system is the most affected, and hepatocellular carcinoma is the most frequent, followed by cancers of the colon, esophagus, and pancreas; others are located in the uterus, breast, and prostate. This association with various neoplasms is attributed to the systemic inflammatory state in which these patients find themselves59. The annual incidence of hepatocellular carcinoma in patients with MASLD and HC is 0.2 to 2.6, and in patients without HC the 10-year incidence is 2.7%60,61.
Mortality
Reported mortality figures are also variable, depending on the population studied, the type of analysis performed, and the diagnostic criteria used. However, the most important series agree that CVD is the main cause of death in patients with MASLD, followed by cancer and thirdly HC4.
Epidemiological studies conducted with NAFLD criteria show that the risk of fatal and non-fatal cardiovascular events is increased; myocardial infarction, coronary or carotid atherosclerosis, and valvulopathy correlate with disease severity62,63. In a meta-analysis with almost 500,000 subjects, mortality with and without NAFLD was compared, and all-cause mortality was found to be higher in NAFLD (HR: 1.34; 95% CI: 1.17-1.54)64.
There are two studies that analyzed the NHANES III database (1988-1994) with a follow-up of more than 25 years and more than 7500 people, and compared mortality according to NAFLD, MAFLD, and MASLD criteria. In general terms, all-cause mortality was higher and very similar in patients with MAFLD and MASLD, older than 65 years, but not in those classified as NAFLD, apparently because, in these cases, the diagnostic criteria exclude subjects with excessive alcohol consumption and other chronic liver diseases40,65. It is concluded that the criteria of the different denominations do impact fatal outcomes, because MAFLD and MASLD include a greater number of subjects with more comorbidity and worse prognosis66.
Mortality from HC secondary to MASLD is approximately 100,000 per year worldwide, with about half of the deaths in Asia, almost a quarter in Europe, another quarter in America, and 17,000 in Africa. In Mexico, around 1250 deaths are estimated, with the highest rate in Yucatán (12.4 per 100,000 inhabitants)67.
Mortality from cancer secondary to MASLD in the world is around 41,000 deaths per year, of which some 25,000 correspond to Asia. In our country, Veracruz and Yucatán are the most affected states, with about 0.6 deaths per 100,000 inhabitants67.
Information regarding mortality in children and adolescents with MASLD is limited. A cohort study that evaluated 718 children and young adults (< 25 years) with biopsy-confirmed NAFLD revealed an absolute risk of mortality of 7.7% in patients with NAFLD, compared to 1.1% in controls. Excess mortality was attributable mainly to cancer, liver disease, and cardiometabolic disease68.
Conclusions
Despite the increase in MASLD cases, containment efforts made in the field of public health are few and uncoordinated, and global health policies still underestimate the magnitude of the problem.
Health infrastructure and budgets for MASLD care and its comorbidities are very unequal. In developed countries, spending is in the millions, but insufficient, and in developing countries, the outlook is even worse, as health systems are overwhelmed to care for cases and contain the high incidence, and Mexico is an example of this.
The general population, health personnel, and decision-makers must be more aware of the problem; health policies must be reoriented, because until now, recognition and attention to the problem have been insufficient. An urgent and key measure for control lies in the primary care level; it is there that prevention must be emphasized, the at-risk population identified and channeled to specialized clinics with multidisciplinary management where their control is rigorous, their self-esteem reinforced, and self-care incentivized with mutual commitment, both from the health system and from patients and their families.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical considerations
Protection of people and animals. The authors declare that no experiments were carried out on humans or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data or require ethical approval. SAGER guidelines do not apply.
Statement on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.
