Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD) currently constitutes the leading cause of chronic liver disease worldwide. Its global prevalence is estimated between 25% and 30% of the adult population, with figures exceeding 40% in regions with a high incidence of obesity and type 2 diabetes mellitus1. The recent nomenclature change, from NAFLD (non-alcoholic fatty liver disease) to MASLD, better reflects the close association with metabolic dysfunction and underscores that this disease is not an isolated epiphenomenon, but rather a hepatic manifestation of a multisystem syndrome2,3. The clinical relevance of MASLD clearly extends beyond hepatic progression to advanced fibrosis, cirrhosis, or hepatocellular carcinoma4–6. In fact, the principal cause of morbidity and mortality in these patients is of cardiovascular origin, not hepatic7–9.
The mechanisms underpinning this association are multiple and complex, in accordance with the “multiple hits” theory, which integrates insulin resistance, lipotoxicity, systemic inflammation, endothelial dysfunction, coagulation abnormalities, neurohormonal regulation, gut microbiota, and genetic determinants10–13 as well as social and commercial determinants (our Lancet). Notably, new mediators are emerging, such as imidazole propionate, a metabolite derived from microbial catabolism of histidine, which is becoming recognized as a key pathogenic marker and possible therapeutic target in the near future14–16.

Graphical abstract. Graphical summary. Cardiovascular risk in metabolic dysfunction-associated steatotic liver disease (MASLD).
Despite the magnitude of the evidence, clinical practice continues to focus almost exclusively on the detection and monitoring of hepatic fibrosis, while cardiovascular risk (CVR) assessment is scarce or nonexistent in most consultations. A national survey in Spanish hospitals showed that fewer than 17% perform a basic physical examination oriented to risk, only 10% use calculators, more than half do not request complementary tests, and barely 46% dedicate significant time to lifestyle counseling; only one in four has multidisciplinary circuits17. This care deficit, combined with the poor performance of classic risk scores, reinforces the need to systematically integrate vascular assessment in our consultations, supported by biomarkers, imaging techniques, and a multidisciplinary approach. In addition to this underassessment, classic prediction algorithms, such as the Framingham Risk Score or the Pooled Cohort Equations, present limited performance in this population, with poor discrimination and calibration, as demonstrated by the TARGET-NASH study18.
The growing recognition of MASLD as an independent determinant of vascular risk compels hepatologists’ to rethink their approach to this pathology. The magnitude of the problem requires not only understanding the pathophysiological mechanisms explaining its association with cardiovascular disease (CVD), but also clearly identifying the different clinical outcomes involved and having adequate tools for stratification. This framework of action aligns with recent guidelines from the European Association for the Study of the Liver (EASL), the European Association for the Study of Diabetes (EASD), and the European Association for the Study of Obesity (EASO), and with the hypertension guidelines from the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH) of 2024 and from the American College of Cardiology (ACC) and the American Heart Association (AHA) of 2025, which consolidate a comprehensive risk-based approach and offer the hepatologist an updated reference to integrate cardiovascular assessment into clinical practice19–22.
With this objective, this article offers a critical and structured review of the link between MASLD and CVR, aimed at expert hepatologists but not necessarily familiar with the technical aspects of vascular risk. The manuscript proposes a deliberate and progressive journey that moves from pathophysiological mechanisms to the systematic description of CVRs associated with MASLD, to subsequently analyze available assessment tools and conclude with a critical reflection on the existing gap in hepatology clinical practice and the necessary multidisciplinary strategies, including the approach to clinical management of CVR in this population. Our hope is to contribute to a paradigm shift in clinical practice, jointly addressing liver care and cardiovascular protection.
Pathophysiology of increased vascular risk in MASLD
The pathophysiology of MASLD is extraordinarily complex and responds to the multiple simultaneous impacts theory, which integrates metabolic, inflammatory, genetic, environmental, and microbiota factors10–13. However, to understand the increase in CVR in these patients, it is more appropriate to focus on mechanisms that, directly or indirectly, link the liver with cardiovascular pathology (Table 1 and Fig. 1).
Table 1. Pathophysiological mechanisms of increased vascular risk in MASLD
| Mechanism | Main cardiovascular effects |
|---|---|
| Insulin resistance | Hyperglycemia, hyperinsulinemia, endothelial dysfunction, accelerated atherogenesis |
| Lipotoxicity and chronic inflammation | Oxidative stress, production of interleukin-6 and tumor necrosis factor alpha, vascular and myocardial remodeling |
| Endothelial dysfunction and atherogenic dyslipidemia | Nitric oxide reduction, small dense LDL, dysfunctional HDL → early atherosclerosis |
| Hepatic fibrosis | Marker of cardiovascular mortality, multi-organ damage, atherosclerosis progression |
| Prothrombotic state | Elevation of factor VIII, protein C resistance → arterial and venous thrombosis |
| Gut microbiota (histidine axis) | Production of imidazole propionate by urdA → insulin resistance, endothelial inflammation, arterial stiffness. Emerging potential therapeutic target |
| Neurohormonal regulation | Sympathetic and RAAS activation → cardiac remodeling, heart failure with preserved ejection fraction |
| Genetics and causal heterogeneity | Variants such as PNPLA3/TM6SF2 promote steatosis but reduce VLDL; other variants increase coronary risk |
HDL: high-density lipoproteins; LDL: low-density lipoproteins; RAAS: renin-angiotensin-aldosterone system; VLDL: very low-density lipoproteins.
The pathophysiology of MASLD connects the liver, cardiovascular system, and metabolism, justifying the integrated cardio-hepato-metabolic approach.
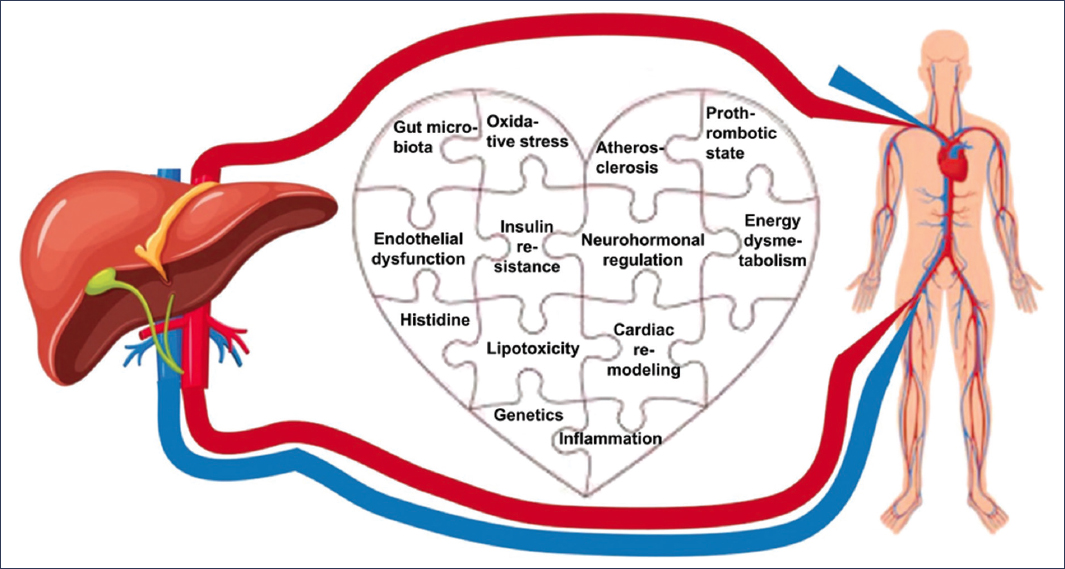
Figure 1. Mechanisms associated with the pathophysiology of increased vascular risk in MASLD.
Insulin resistance and energy dysmetabolism
Insulin resistance is the central axis. It leads to hyperglycemia, hyperinsulinemia, and excessive lipolysis, with influx of free fatty acids to the liver and lipid overload in multiple tissues. This situation not only promotes hepatic steatosis but deteriorates endothelial function by reducing nitric oxide bioavailability and activating pro-inflammatory pathways, laying the foundation for atherogenesis23.
Lipotoxicity, inflammation, and oxidative stress
Excess toxic lipids (diacylglycerols, ceramides) induce mitochondrial dysfunction and oxidative stress, activating the endoplasmic reticulum stress response and releasing cytokines such as tumor necrosis factor alpha and interleukin-6. The result is a low-grade systemic inflammatory state that accelerates the formation and progression of atherosclerotic plaques and contributes to myocardial remodeling24.
Endothelial dysfunction and atherosclerosis
Endothelial dysfunction, characterized by reduced nitric oxide and increased oxidative stress, together with expression of adhesion molecules, favors leukocyte infiltration and plaque formation. The typical dyslipidemia of MASLD (small dense low-density lipoproteins [LDL], dysfunctional high-density lipoproteins [HDL], hypertriglyceridemia) is highly atherogenic and reinforces this process25,26.
Prothrombotic state
MASLD is associated with a procoagulant phenotype with elevation of factor VIII and resistance to activated protein C. This condition contributes to the higher incidence of arterial and venous thrombosis in this population27.
Gut microbiota: the emerging histidine axis
The gut microbiota represents a bridge between the liver and the cardiovascular system. Of particular relevance is the microbial metabolism of histidine, capable of generating imidazole propionate (ImP) through bacteria carrying the urdA gene. This metabolite, initially described in diabetes and obesity, induces insulin resistance, endothelial inflammation, and arterial stiffness, and appears frequently in conditions intimately linked to MASLD. The ImP+/urdA^high biotype concept has been proposed, which confers a phenotype of insulin resistance, steatosis, and inflammatory vascular risk. ImP is emerging as an emerging functional marker and a possible first-order therapeutic target for addressing vascular risk in MASLD14–16,28–30.
Neurohormonal regulation and cardiac remodeling
Activation of the sympathetic nervous system and the renin-angiotensin-aldosterone axis, favored by inflammation and insulin resistance, contributes to the development of heart failure, particularly with preserved ejection fraction31.
Genetics and causal heterogeneity
Mendelian randomization studies suggest that the relationship between hepatic steatosis and coronary disease depends on the biological mechanism. Variants such as PNPLA3 or TM6SF2 promote steatosis while reducing very low-density lipoprotein (VLDL) secretion, conferring a paradoxically less atherogenic lipid profile. In contrast, other variants associated with dyslipidemia favor a net higher CVR32.
In conclusion, the increase in CVR in MASLD results from the convergence of multiple pathophysiological pathways: insulin resistance, lipotoxicity and systemic inflammation, endothelial dysfunction, advanced fibrosis, prothrombotic state, and cardiac remodeling33,34. Among them, the microbial metabolism of histidine and ImP production may consolidate as an emerging mechanism of maximum relevance, which could redefine the therapeutic approach to vascular risk in this disease in the near future.
Fundamental concepts in vascular risk and major adverse cardiovascular events
Understanding vascular risk is essential for interpreting the prognosis of patients with MASLD. Although hepatology has traditionally focused its attention on hepatic outcomes, the burden of CVD associated with this pathology requires precise handling of concepts and tools usually employed in cardiology and internal medicine.
Definition of vascular risk
The term “vascular risk” refers to the probability of developing, within a defined time horizon, a clinically relevant cardiovascular or cerebrovascular event. Traditionally, it has been limited to the atherosclerotic sphere, but in a broad sense, it includes both arterial and venous events. Its magnitude depends on the sum of classic risk factors, such as hypertension, diabetes, dyslipidemia, smoking, and central obesity, and emerging determinants such as insulin resistance, low-grade systemic inflammation, endothelial dysfunction, and intestinal dysbiosis, whose pathophysiological and prognostic relevance is increasingly better documented8,35.
Major adverse cardiovascular events
Major adverse cardiovascular events (MACE) are the clinical outcomes with the greatest impact in clinical practice and research. Their strict definition, employed in most registry trials, includes non-fatal myocardial infarction and ischemic stroke and cardiovascular death (Fig. 2). In a broader sense, the term extends to encompass hospitalization for unstable angina or heart failure, coronary revascularizations, severe arrhythmias with life-threatening compromise, and resuscitation after sudden death. Heterogeneity in the use of the concept requires critical interpretation of literature results and makes comparisons between studies difficult36.
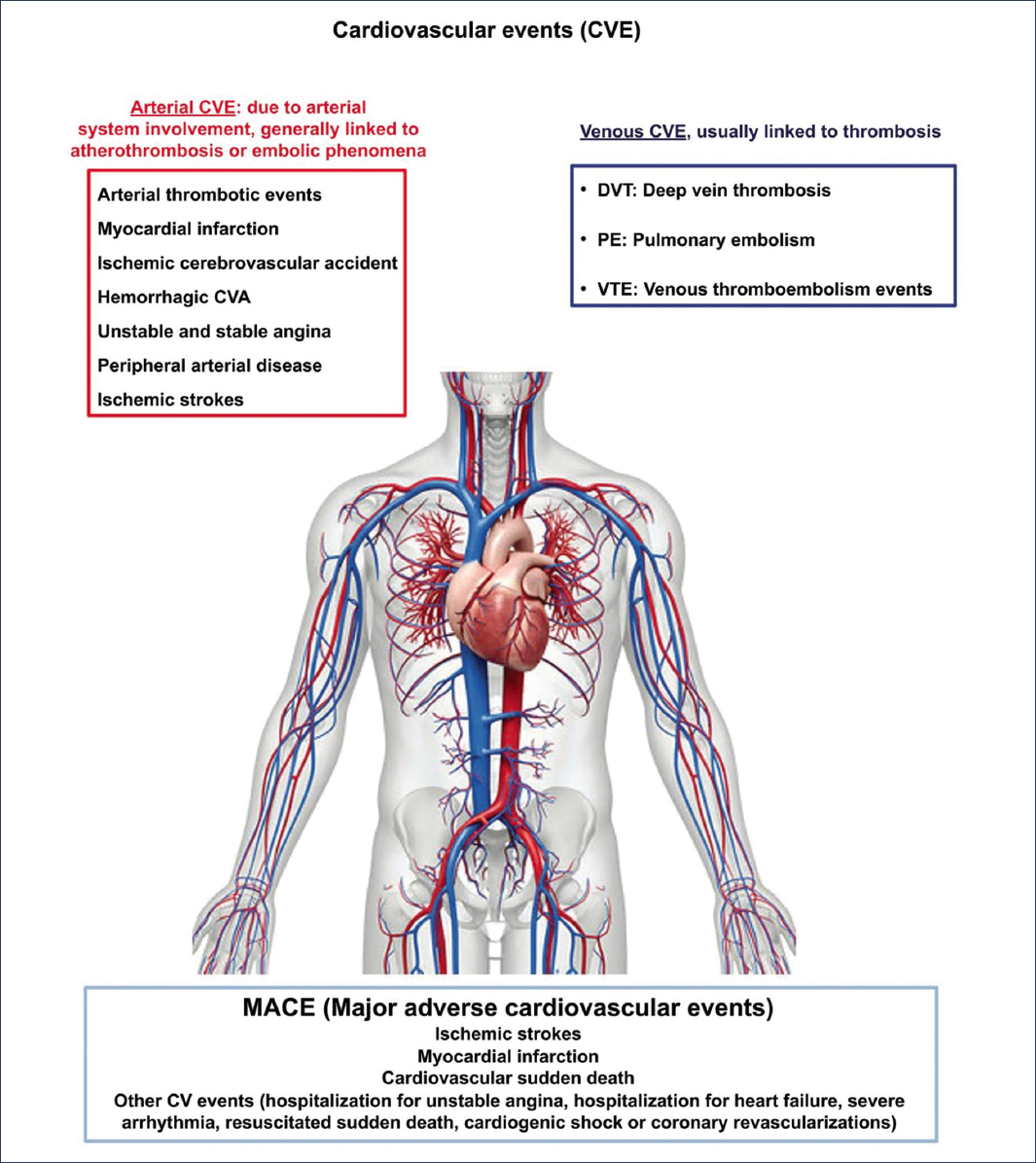
Figure 2. Cardiovascular events that may occur in patients with MASLD.
Clinical measurement of vascular risk
Correct estimation of this risk requires systematic collection of clinical and analytical parameters. Blood pressure should be measured under standardized conditions, with validated devices, after a rest period, and avoiding interpretations from isolated values. The complete lipid profile (total cholesterol, LDL cholesterol [LDL-C], HDL cholesterol [HDL-C], triglycerides, non-HDL cholesterol, and, when possible, apolipoprotein B) constitutes the basis of any prediction model. Lipoprotein (a) should be measured at least once in a lifetime (with mention of insulin resistance HOMA-IR, systemic inflammation hs-CRP, congestion NT-ProBNP, and myocardial damage Troponin I). Fasting glucose and glycated hemoglobin (HbA1c) are essential for detecting diabetes or prediabetes. Quantification of body mass index (BMI) and waist circumference allows assessment of global and central adiposity (and if possible, determination of body composition with measurement of visceral adipose tissue by BIA, DEXA, BOD-POD, CT, MRI, or PET), and a detailed smoking history should be collected systematically. Finally, determination of the albumin/creatinine ratio in urine adds critical information: microalbuminuria reflects microvascular damage not only renal but systemic and is associated with an independent increase in CVR and renal risk, even in individuals with preserved glomerular filtration37.
Risk stratification tools
Prediction of CVR in the general population relies on validated algorithms that combine age, sex, and conventional risk factors. We will analyze these tools later.
Subclinical cardiovascular damage
Beyond risk factors and clinical algorithms, detection of subclinical damage provides a complementary dimension38,39. We will address these aspects later.
In conclusion, the hepatologist must become familiar with the fundamentals of vascular risk and the MACE concept, as well as with the metrics and parameters necessary for its measurement. This conceptual framework is essential for interpreting the impact of MASLD on cardiovascular prognosis and for integrating the hepatic perspective into contemporary vascular prevention.
Cardiovascular risks associated with MASLD
MASLD is now configured as a systemic cardiovascular determinant. Its association with major clinical outcomes transcends the accompaniment of classic risk factors and encompasses a broad spectrum that includes atherothrombotic events, arrhythmias, predominantly diastolic heart failure, microvascular dysfunction, and specific risks in selected subgroups (Table 2). The magnitude of risk increases with hepatic fibrosis progression, persists after adjusting for cardiometabolic confounding, and even reaches populations traditionally considered lower risk, such as women and young adults40.
Table 2. Cardiovascular manifestations associated with MASLD and level of evidence
| Cardiovascular manifestation | Epidemiological evidence | Risk magnitude | Evidence level (EASL/AASLD) |
|---|---|---|---|
| Myocardial infarction | Cohorts and meta-analyses | ↑ 1.5-2 times | High |
| Ischemic stroke | Population cohorts | ↑ 1.5 times | Moderate |
| Atrial fibrillation | Population studies | ↑ 1.2-1.5 times | Moderate |
| Heart failure (with preserved ejection fraction) | Cohorts and meta-analyses | ↑ 2 times | High |
| Cardiovascular mortality | Meta-analyses (> 10,000 cases) | Significant increase | High |
AASLD: American Association for the Study of Liver Diseases; EASL: European Association for the Study of the Liver.
MASLD is associated with an independent increase in cardiovascular risk, with greater magnitude in the presence of advanced fibrosis. Heart failure with preserved ejection fraction and global cardiovascular mortality stand out as manifestations with the most solid evidence.
Atherothrombotic events (MACE): myocardial infarction, stroke, and cardiovascular death
Contemporary evidence confirms an excess risk of MACE that persists after adjusting for classic factors and is accentuated when significant fibrosis coexists40. In quantitative terms, the risk of ischemic heart disease and myocardial infarction is increased by approximately 25-30% in patients with MASLD, independently of age, sex, and cardiometabolic comorbidity, according to a meta-analysis that integrated approximately 1.2 million individuals41. Alon et al.42 confirm this and document significant increases in the risk of infarction, stroke, atrial fibrillation (AF), and heart failure in these patients. A subsequent meta-analysis demonstrated greater cardiovascular morbidity and mortality, including lean NAFLD phenotype43. In stroke, the relationship is equally consistent and presents a gradient with hepatic fibrosis. A meta-analysis published in Stroke showed that the risk of fatal and non-fatal cerebrovascular events increases with MASLD severity44. In those under 40 years, a systematic review and meta-analysis of 10.6 million people estimated a global hazard ratio (HR) of 1.63 for cardiovascular events, with significant increases in coronary heart disease, stroke, AF, and heart failure, requiring early vascular assessment when MASLD coexists at early ages45,46. This “hepatic signal” clinically translates into a greater burden of MACE as the disease progresses, justifying integration of fibrosis stratification into vascular risk assessment40. Real-world practice evidence corroborates these findings. In a UK Biobank cohort, MASLD emerged as an independent factor for all-cause mortality and adverse cardiovascular events; additionally, adding MASLD to traditional models improved prediction capacity for outcomes47. After myocardial infarction, a Fatty Liver Index (FLI) > 60 was associated with higher cardiovascular mortality at 12 years, independently of obesity and diabetes, in the Alpha Omega cohort48. In a longitudinal cohort, both persistence and development of steatosis were linked to a higher risk of infarction, stroke, heart failure, and mortality, compared to those without MASLD; steatosis regression was associated with a more favorable risk profile, pointing to a potentially reversible component of excess vascular risk49. In high-risk subpopulations, the magnitude of the problem is even greater, as we have already indicated. In type 2 diabetes, MASLD has been shown to be associated with a significant increase in CVD and all-cause mortality, with clinically relevant absolute risk differences50. Regarding cardiovascular mortality and sudden death, the literature shows heterogeneous signals. While some studies have observed a clear increase in cardiovascular mortality, others detect an excess of events that does not always translate into specific mortality, possibly due to competing risks and methodological differences43. The association with sudden cardiac death is emerging and requires prospective confirmation; in patients carrying a defibrillator, the presence of MASLD has been linked to greater burden of ventricular arrhythmias and adverse events51. MASLD constitutes an independent determinant of MACE, with a risk gradient related to fibrosis, high expression in type 2 diabetes, and significant presence in young adults. This reality requires systematic integration of the cardiovascular dimension in clinical assessment, especially when fibrosis or cardiometabolic phenotype are relevant40,47,52 (Table 3).
Table 3. Evidence from cohorts and studies on cardiovascular risk in MASLD
| Study/cohort | Population | Main findings | Implications in MASLD |
|---|---|---|---|
| MESA77 | Multiethnic population cohort | PCE showed a c-statistic ≈ 0.69 in moderate-severe steatosis; marked underestimation in women | Classic models underestimate risk, especially in women and advanced disease |
| UK Biobank47 | United Kingdom population cohort | MASLD associated with increased MACE and cardiovascular mortality | MASLD adds additional risk not captured by conventional algorithms |
| TARGET-NASH18 | Multicenter clinical practice cohort | Framingham, PCE, and PREVENT with c-statistic 0.58-0.60; poor calibration (overestimation in high risk, underestimation in low risk) | Serious limitations of classic models in MASLD |
| HEPAMET/ETHON78 | Clinical vs. population cohort | Patients with MASLD higher prevalence of cardiovascular events and more elevated SCORE categories; MASH and advanced fibrosis = independent predictors of high risk | Hepatic fibrosis adds prognostic power in risk stratification |
| Athens cardiometabolic cohort79 | Patients with cardiometabolic factors | FIB-4 consistently associated with subclinical arterial injury and cardiovascular events; modest discrimination improvement | FIB-4 can be integrated into cardiovascular prediction models in MASLD |
| NAFLD CV-risk score80 | Specific model (age, mean platelet volume, diabetes) | AUROC ≈ 0.83-0.84 for predicting MACE at 1 year; better than Framingham and QRISK2 | Promising, but pending extensive external validation |
| Meta-analysis in women82 | Women with MASLD | Higher CVR than in men, with amplification at greater hepatic severity | Stratification should consider sex as a key modifier |
| Type 1 diabetes and MASLD83 | Patients with type 1 diabetes and MASLD | Adjusted OR 4.26 for ASCVD | Coexistence of MASLD multiplies risk in vulnerable subgroups |
| Meta-analysis in youth45 | Population ≤ 40 years | Pooled HR 1.63 for cardiovascular events in MASLD | Justifies early integration of hepatic variables into prediction models |
| MASLD vs. MAFLD41,84,85 | Population studies | Higher CVR in MAFLD (51.1%) vs. MASLD (35.0%); differences in hepatic stiffness and metabolic severity | MAFLD could better capture atherosclerotic risk gradient |
| Pericardial/periaortic fat86 | Imaging studies | Greater volume associated with MASLD severity; improves prediction when added to classic factors | Possible additional marker in risk prediction |
ASCVD: atherosclerotic cardiovascular disease; AUROC: area under the ROC curve; FIB-4: fibrosis-4 index; HR: hazard ratio; MACE: major adverse cardiovascular events; OR: odds ratio; CVR: cardiovascular risk.
Evidence from cohorts shows that classic models underestimate cardiovascular risk in MASLD, with special impact on women, youth, and patients with advanced fibrosis. Specific models, such as the NAFLD CV-risk score, offer better discrimination but require external validation before widespread application.
Arrhythmias: atrial fibrillation and others
AF is the arrhythmia most solidly linked to MASLD. A significant increase in AF risk has been observed in patients with MASLD, an association that persists after adjusting for cardiometabolic confounders. A meta-analysis of cohorts confirmed the increased risk of AF (along with infarction, stroke, and heart failure)42. In a large national cohort, the incidence of AF was higher in patients with MASLD and increased progressively with hepatic fibrosis; the excess was evident even in young adults and was accentuated with advanced fibrosis53. Additionally, a study conducted using Mendelian randomization suggests that more severe phenotypes (e.g., MASH [metabolic dysfunction associated steatohepatitis]) could be causally related to AF, while for global MASLD, the evidence is less conclusive54. Overall, findings support considering AF surveillance in patients with MASLD and significant fibrosis. Additionally, alterations in repolarization (QTc prolongation and dispersion) and a possible increase in ventricular arrhythmias have been described, probably mediated by systemic inflammation, autonomic dysfunction, and diffuse myocardial fibrosis55. The association between hepatic fibrosis and AF reinforces the role of hepatic structural damage as a marker of arrhythmic risk56. In MASLD with significant fibrosis or high-risk phenotypes, it is advisable to incorporate opportunistic screening for AF (pulse/electrocardiogram) and review proarrhythmic factors (electrolytes, QT prolongers).
“MASLD cardiomyopathy”: diastolic dysfunction and heart failure with preserved ejection fraction
The cardiomyopathy associated with MASLD manifests through left atrial and ventricular remodeling, diastolic dysfunction, and elevated filling pressures. A meta-analysis of 31 studies demonstrated greater left atrial volume, left ventricular mass, and, above all, deterioration of diastolic function in MASLD57. This structural-functional alteration translates into greater risk of incident heart failure, with a more marked excess for heart failure with preserved ejection fraction versus reduced ejection fraction in population analyses58. A review by Mantovani et al.59 synthesizes the clinical and translational evidence linking MASLD with heart failure with preserved ejection fraction, proposing MASLD as a phenotype of adverse cardiometabolism with diffuse myocardial and vascular involvement. In patients with MASLD and exertional dyspnea, edema, or elevation of N-terminal pro-brain natriuretic peptide (NT-proBNP), suspicion of heart failure with preserved ejection fraction should be raised early; coexistence of obesity, type 2 diabetes, hypertension, and hepatic fibrosis increases the pretest probability and justifies a structured cardiologic evaluation.
Microvascular and subclinical disease
Markers of silent vascular damage are elevated in MASLD. In a review that included 59 studies (16,179 cases and 26,120 controls), MASLD was associated with an absolute increase of 0.123 mm in carotid intima-media thickness (≈20.6%)60. Arterial wall stiffness is increased in both MASLD and MAFLD (Metabolic-Associated Fatty Liver Disease), with stronger associations observed in MAFLD and in women61. Regarding coronary microcirculation, MASLD is associated with microvascular dysfunction (reduced flow reserve), suggesting a functional basis for the greater propensity to significant ischemia62. In people with type 2 diabetes and MASLD, fibrosis evaluated by the Fibrosis-4 Index (FIB-4) or by the NAFLD Fibrosis Score (NFS) is independently associated with coronary artery calcium (CAC) and modestly improves coronary stratification25. In parallel, steatosis regression has been associated with lower CAC progression, supporting modifiability of vascular risk63. Renal microvascular involvement (albuminuria, glomerular filtration) is more prevalent in MASLD, with approximately 1.45 times more risk of incident chronic kidney disease in the long term64. The presence of subclinical vascular damage reinforces the indication to intensify cardiovascular prevention. In hepatology, its utility is greater when this evaluation can change decisions (e.g., reclassify doubtful risk or prioritize cardiometabolic therapies).
Other specific risks
Type 1 diabetes
In adults with type 1 diabetes, the coexistence of steatosis and significant fibrosis is associated with a very substantial increase in estimated CVR at 10 years, compared to type 1 diabetes without steatosis or with steatosis without fibrosis. This finding was maintained in all subgroups by sex, age, HbA1c, and renal function65.
Hypertension
MASLD increases the risk of incident hypertension by 66%, with higher prevalences as blood pressure strata increase; additionally, coexistence of MASLD is associated with greater total and cardiovascular mortality in hypertensive subjects, which may translate into more cases of resistant hypertension66,67.
Cognitive impairment and dementia
The relationship between hepatic integrity and cognitive function is a topic under evaluation. A national study showed that high FIB-4 is associated with 18% more risk of incident dementia, even after broad adjustments68. However, other population analyses (UK Biobank) have not confirmed an overall excess when FLI is used as a proxy for MASLD, so causality remains open69.
Venous thromboembolism
Recent population studies suggest a higher risk of venous thromboembolism in patients with MASLD. In Korea, the presence of MASLD was associated with more venous thromboembolism events in follow-up, while in the Tromsø Study, elevated FLI values were related to incident venous thromboembolism, although part of the association was explained by general adiposity70,71. The signal is emerging and requires confirmation with direct liver fat measurements and fine adjustment for central obesity.
Performance of general risk scores and incorporation of the “hepatic signal”
Classic algorithms (PCE/SCORE2/Framingham) show calibration and discrimination limitations in MASLD, with a tendency to underestimate risk in women and moderate-severe steatosis. Incorporation of non-invasive hepatic markers, especially FIB-4, provides incremental value over conventional systems and appears to mediate part of its effect through vascular injury, according to contemporary analyses of the Athens Cardiometabolic Registry and integrated cohorts72. Additionally, the Steatosis-associated Fibrosis Estimator score (SAFE) shows utility for stratifying fibrosis and facilitating care circuits that connect hepatic and cardiovascular decisions73.
Genetics and causality
Mendelian randomization studies indicate that the link between steatosis and coronary heart disease depends on the underlying lipid mechanism: when variants that reduce hepatic VLDL secretion (e.g., PNPLA3, TM6SF2, APOB, and MTTP) are excluded, the association with coronary artery disease emerges more clearly; conversely, aggregating them dilutes or reverses the signal, as they are accompanied by “apparently favorable” lipoprotein profiles74,75. This divergent biology helps explain why not all “hepatic fat” confers the same CVR and reinforces the clinical emphasis on phenotyping beyond steatosis (fibrosis, atherogenic dyslipidemia, insulin resistance, inflammation).
Available evidence positions MASLD as a systemic cardiovascular determinant: it increases MACE, favors AF, is associated with a heart failure with preserved ejection fraction phenotype, presents a broad microvascular/subclinical footprint, and aggravates risks in specific scenarios (type 1 diabetes, hypertension, possible cognitive impairment, venous thromboembolism). The fibrosis gradient is a transversal element. In hepatology, this implies systematically documenting the cardiovascular dimension, recognizing high-risk subgroups, and considering early referral to vascular risk units when risk burden or target organ damage warrant it.
Cardiovascular risk assessment in MASLD
Importance of risk classification
CVR stratification constitutes an essential pillar in the care of patients with MASLD, since CVD is its main cause of mortality. Adequate classification allows for the identification of individuals without clinical manifestations CVD but are at high risk, enabling early implementation of preventive measures, in addition to guiding intervention intensity and setting specific therapeutic targets for blood pressure, LDL-C, or HbA1c.
Clinical measurement of risk factors
The first step consists of systematic clinical measurement of classic risk factors: blood pressure, complete lipid profile, glucose and HbA1c, BMI, abdominal perimeter, smoking, and family history of premature CVD. This basic approach constitutes the pillar on which prognostic stratification in MASLD is built (Fig. 3).
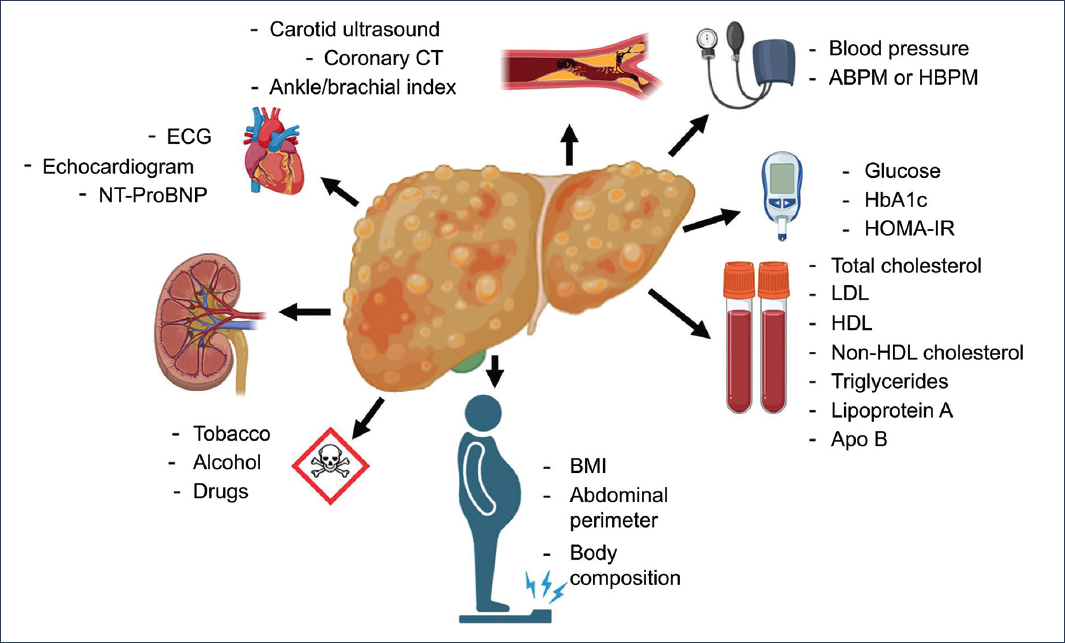
Figure 3. Cardiovascular risk assessment in the MASLD patient. HBPM: home blood pressure monitoring; ECG: electrocardiogram; HbA1c: glycated hemoglobin; HDL: high-density lipoproteins; BMI: body mass index; LDL: low-density lipoproteins; ABPM: ambulatory blood pressure monitoring; CT: computed tomography.
Cardiovascular risk calculators and classification (Table 4)
Table 4. Cardiovascular risk determination models and applicability in MASLD patients
| Model | Main variables included | Limitations in MASLD | Possible adjustments or practical considerations |
|---|---|---|---|
| Framingham Risk Score (FRS) | Age, sex, smoking, blood pressure, total cholesterol, HDL, glucose | Tends to underestimate cardiovascular risk in MASLD (c-statistic ≈ 0.58 in specific cohorts) | Can be complemented with hepatic markers (e.g., FIB-4). |
| Pooled Cohort Equations (PCE) | Age, sex, lipids, hypertension, diabetes, smoking | Similar performance to FRS; underestimates residual risk in MASLD | Adjust with hepatic fibrosis or vascular imaging. |
| SCORE2/SCORE2-OP | Age, sex, blood pressure, lipids, smoking, European region of risk | Designed for the European population; in MASLD also tends to underestimate risk | Possible recalibration incorporating fibrosis or hepatic biomarkers |
| PREVENT (AHA, 2023) | Classic factors + renal (eGFR, albumin/creatinine), HbA1c, social indicators | Not specifically validated in MASLD; slightly better performance than FRS/PCE (c-statistic ≈ 0.60 in some cohorts) | Potentially useful in MASLD with metabolic and renal comorbidity, still without formal validation |
| PREVENT submodels | PREVENT-CVD (global cardiovascular disease), PREVENT-ASCVD (atherosclerotic disease), PREVENT-HF (heart failure) | Increases risk granularity but is not validated in MASLD | May help identify heart failure with preserved ejection fraction risk relevant in MASLD |
| SAFE score | Cardiovascular risk integrated with hepatic biomarkers (e.g., FIB-4, ELF) | Initial validation in MASLD cohorts, still limited | Very promising as cardio-hepato-metabolic tool |
ELF: enhanced liver fibrosis; FIB-4: fibrosis-4 index; HbA1c: glycated hemoglobin; HDL: high-density lipoproteins; eGFR: estimated glomerular filtration rate. Classic cardiovascular risk models have been validated in the general population and are widely used in clinical practice, but show limited performance in patients with MASLD, as they tend to underestimate residual risk because they do not include hepatic or metabolic variables specific to this disease. The PREVENT model, developed by the American Heart Association in 2023, represents an advance over traditional equations by incorporating renal parameters (glomerular filtration, albuminuria), glycemic (HbA1c), and social (deprivation index), in addition to offering different submodels (PREVENT-CVD, PREVENT-ASCVD, PREVENT-HF). Although it modestly improves discrimination, it has not yet been specifically validated in MASLD and should be used with caution in this context. The U-Prevent platform is not a model itself but a digital tool that allows calculating different models (including FRS, SCORE2, PREVENT) and comparing them in clinical practice. It may be useful for hepatologists as a practical resource. The SAFE score, which integrates cardiovascular and hepatic factors (e.g., FIB-4, ELF), emerges as a model adapted to MASLD and could become the most appropriate tool for cardio-hepato-metabolic stratification, although its validation is still preliminary. In practice, the best strategy is to combine traditional models with additional hepatic information (fibrosis, biomarkers, or non-invasive imaging) until models validated specifically for MASLD are available.
Prediction of CVR in the general population is based on validated algorithms that combine age, sex, and conventional risk factors, among which stand out the Pooled Cohort Equations (PCE) recommended in the United States of America, SCORE2 for extended use in Europe, and the Framingham algorithm, considered the reference standard77,78. These models estimate the probability of experiencing a major cardiovascular event within 10 years and allow individuals to be classified into risk categories (low, moderate, high, or very high), forming the basis for preventive19 and therapeutic decision-making. However, they present limitations in certain subgroups, such as young women and patients with metabolic liver disease, in whom they tend to underestimate real risk19,38. In this context, the ESC 2021 guidelines recommend SCORE2 as a reference tool19, while the ACC/AHA 2025 guideline introduces a global risk-based approach through the PREVENT calculator, emphasizes the need to institute early treatment, and adapts recommendations to specific subpopulations such as pregnant women and patients with kidney disease, resistant hypertension, or cognitive impairment, thus configuring itself as a practical and accessible tool for risk stratification in clinical practice22. Finally, SAFE, initially conceived for fibrosis stratification in primary care, has also been proposed as a cardiovascular prioritization strategy20.
Limitations of risk prediction systems in patients with MASLD
Traditional models were not designed for populations with liver disease and present discrimination and calibration defects. In the MESA study, the PCE showed modest discriminative capacity (c-statistic: ≈0.69), which decreased to almost 0.65 in patients with moderate-severe steatosis, with marked risk underestimation in women77. Concordantly, in the UK Biobank, the presence of MASLD was associated with an increase in MACE and cardiovascular mortality, but inclusion of this variable in traditional models barely improved prediction47.
The multicenter TARGET-NASH study confirmed that Framingham, PCE, and PREVENT performed poorly in MASLD, with a c-statistic between 0.58 and 0.60 and poor calibration18. These defects translate into risk underestimation in women, young adults, and the metabolically healthy phenotype.
An additional aspect is the influence of competing events (cirrhosis, hepatocarcinoma), which can bias risk estimates if standard Cox models are applied. In this context, it is preferable to employ cumulative incidence functions or Fine-Gray subdistribution models to more precisely analyze CVR and non-cardiovascular risk in patients with MASLD80,81.
Evidence from clinical cohorts and biomarkers
In the HEPAMET registry, compared with the ETHON population cohort, patients with MASLD presented a higher prevalence of cardiovascular events and more frequently reached elevated SCORE categories, even after excluding previous CVD. The presence of MASH and, especially, advanced fibrosis, was independently associated with high risk, underscoring the prognostic relevance of the hepatic signal78. Among biomarkers, FIB-4 has demonstrated a consistent association with subclinical arterial injury and cardiovascular events, modestly improving model discrimination and associating with higher mortality at elevated cut-off points79. Likewise, specific models have been developed, such as the NAFLD CV-risk score, which combines age, mean platelet volume, and diabetes, with a receiver operating characteristic area under the curve (AUROC) of 0.83-0.84 for predicting MACE at 1-year, surpassing Framingham and QRISK2, although still requiring external validation80. SAFE, initially conceived to stratify hepatic fibrosis, has also been validated as a useful tool for identifying individuals with greater vascular vulnerability20,79. These findings illustrate a fundamental aspect: biomarkers and indices designed for hepatic stratification provide, with a change of perspective, cardiovascular prognostic value.
Clinical subgroups and diagnostic phenotypes
Certain clinical subgroups present specific vulnerabilities. In a meta-analysis, women with MASLD showed higher CVR than men, with risk amplification the hepatic severity increased82. In type 1 diabetes, coexistence of MASLD is associated with a marked increase in atherosclerotic risk (adjusted odds ratio: 4.26 for atherosclerotic cardiovascular disease [ASCVD])83. In the young population (≤ 40 years), a meta-analysis reported a pooled HR of 1.63 for cardiovascular events in MASLD45.
Diagnostic phenotypes also influence. The MAFLD construct (not previously mentioned or defined), by integrating metabolic criteria, better captures the atherosclerotic gradient than MASLD. Studies have shown a higher risk in MAFLD (51.1%) than in MASLD (35.0%), with differences in hepatic stiffness and metabolic severity41,84,85. On the other hand, pericardial and periaortic fat volume is associated with greater MASLD severity and improves prediction when added to classic metabolic syndrome factors86.
Non-invasive assessment of subclinical cardiovascular disease (Table 5)
Table 5. Biomarkers and imaging techniques for subclinical cardiovascular risk evaluation in MASLD patients
| Parameter | Description | Findings in MASLD | Clinical implications |
|---|---|---|---|
| Microalbuminuria87 | Classic marker of endothelial dysfunction and early renal damage | Associated with greater arterial stiffness and cardiovascular mortality, even without established kidney disease | Identifies early vascular dysfunction; simple and cost-effective screening |
| High-sensitivity CRP/IL-624 | Inflammatory biomarkers | Elevated in MASLD; correlates with atherosclerosis progression and higher event incidence | Indicate systemic inflammation; potential for additional risk stratification |
| High-sensitivity troponin88 | Marker of subclinical myocardial damage | Higher levels in MASLD with advanced fibrosis, even without clinical heart disease | Detects early ventricular dysfunction; useful in high risk |
| NT-proBNP88 | Marker of cardiac wall stress | Elevated in MASLD; associated with advanced fibrosis and heart failure risk | Helps identify heart failure with preserved ejection fraction risk |
| FIB-4/MASLD fibrosis score25,79 | Serum indices of hepatic fibrosis | Associated with coronary atherosclerosis, CAC progression, and cardiovascular events | Hepatic biomarkers that provide cardiovascular prognostic value |
| SAFE score79 | System designed for fibrosis in primary care | Useful for identifying individuals with greater vascular vulnerability | Practical prioritization tool in integrated stratification |
| CAC63 | Computed tomography for coronary calcification quantification | Most robust predictor of subclinical atherosclerosis; faster progression in MASLD; slowing with steatosis regression | Justifies reclassification to high risk; an independent marker of events |
| Carotid ultrasound89 | Assessment of plaques and intima-media thickness | Higher prevalence of atheromatosis in MASLD, independent of classic factors | Accessible technique; useful for early detection in intermediate risk |
| Pulse wave velocity90 | Measure of arterial stiffness | Increased in MASLD; correlates with degree of hepatic fibrosis | Reinforces concept of vascular remodeling parallel to hepatic |
| Cardiac magnetic resonance91 | Advanced technique for myocardial structure and function | Detects ventricular dysfunction and diffuse fibrosis in MASLD without previous heart disease | Research tool; anticipates heart failure with preserved ejection fraction risk |
CAC: coronary artery calcium; FIB-4: fibrosis-4 index; NT-proBNP: N-terminal pro-brain natriuretic peptide; CRP: C-reactive protein.
Analytical biomarkers and imaging techniques provide information complementary to classic models, allowing detection of subclinical vascular or myocardial damage in patients with MASLD. CAC and carotid ultrasound are the most validated tools for risk reclassification, while biomarkers such as troponin, NT-proBNP, and FIB-4 offer innovative perspectives for cardio-hepato-metabolic integration.
Clinical approximation may be insufficient to capture the real magnitude of risk. In this context, non-invasive evaluation acquires a crucial role.
Analytical biomarkers
Microalbuminuria, a marker of endothelial dysfunction, is associated in MASLD with arterial stiffness and cardiovascular mortality, even without established kidney disease87. High-sensitivity C-reactive protein and cytokines such as interleukin-6 correlate with atherosclerotic progression and higher event incidence24. High-sensitivity troponin and NT-proBNP detect subclinical myocardial dysfunction, with higher levels in patients with advanced fibrosis even without known heart disease88. HOMA-IR. Additionally, serum indices of hepatic fibrosis, such as FIB-4 and MASLD fibrosis score, have been linked to a higher prevalence of coronary atherosclerosis and CAC progression, even in diabetic patients25.
Imaging techniques
CAC is the most robust marker of subclinical atherosclerosis and predicts event incidence in patients with MASLD, with faster progression than in controls and slowing if steatosis regression is achieved63. Carotid ultrasound shows higher plaque prevalence and increased intima-media thickness (technique in disuse) in MASLD, independently of classic factors89. Arterial stiffness assessed by pulse wave velocity correlates with hepatic fibrosis90 ECG and echocardiogram. Finally, cardiac magnetic resonance has allowed detecting ventricular dysfunction and diffuse fibrosis in patients with MASLD without known heart disease, anticipating risk of heart failure with preserved ejection fraction91.
Clinical guidelines and practical applicability
Recent guidelines reinforce the need to integrate these findings. The ESC/ESH 2024 hypertension guideline highlights the importance of measuring blood pressure in a standardized manner and identifying target organ damage21. The EASL-EASD-EASO 2024 guidelines recognize MASLD as an independent risk factor and advanced fibrosis as a prognostic determinant20. For its part, the ACC/AHA 2025 guideline recommends PREVENT as a global model, with specific sections for pregnant women and patients with kidney disease, resistant hypertension, or cognitive impairment22.
The applicability of these tools in clinical practice should not be understood as a universal mandate. Their greatest utility is observed in patients at intermediate risk, in those with discordant results between clinical findings and traditional scoring systems, or when there is suspicion of vascular damage not explained by usual factors. In most cases, advanced evaluation and interpretation of these findings should be carried out by CVR specialists, ideally in multidisciplinary units integrating hepatology, cardiology, endocrinology, and nephrology. The hepatologist’s role is to identify patients who are candidates for this assessment and to facilitate their referral, thus ensuring truly comprehensive care in a disease whose main prognostic threat transcends the liver.
Key messages for practice
- – CVR stratification should be performed in all patients with MASLD, even without known cardiovascular disease.
- – Significant fibrosis (≥ F2) justifies reclassifying risk toward higher categories.
- – Classic risk determination systems can be complemented with hepatic, metabolic, cardiovascular biomarkers or vascular imaging tests.
- – Detection of subclinical CVD requires intensive preventive measures.
- – Referral to multidisciplinary units improves care integration and cardiovascular prevention in patients with MASLD.
Management of cardiovascular risk in MASLD
Evidence-based recommendations for diet, exercise, and weight loss
Treatment of MASLD is primarily based on intensive lifestyle modification, as it is the most effective strategy for improving both hepatic complications and CVR. Currently, there is no pharmacological therapy that equals the benefits of weight loss, dietary improvement, and increased physical activity20,92,93.
Clinical guidelines recommend sustained weight loss of 7-10%, with intermediate goals of 3-5% in the first months to favor adherence20. Diet should be personalized according to patient preferences, culture, and lifestyle, prioritizing healthy dietary patterns94. The Mediterranean diet is the most supported, especially in its polyphenol-rich variant95–97, but the DASH (Dietary Approaches to Stop Hypertension) diet and healthy vegetarian patterns are also valid options98–100. The key is a hypocaloric diet that the patient can maintain long-term, with reduction of free sugars, especially fructose and sugar-sweetened beverages101–103. Strategies such as intermittent fasting or time-restricted eating may also be considered, always under professional supervision104,105.
Regarding substance consumption, total alcohol abstinence is recommended, even in small amounts, due to its negative effect on hepatic fibrosis and CVR106. In contrast, regular coffee consumption (2-3 cups per day) is associated with hepatic and metabolic benefits, thanks to its antioxidant compounds107,108.
Physical activity is fundamental in MASLD management, even without significant weight loss. A combination of aerobic and strength exercise is recommended, with a minimum of 150 minutes of moderate aerobic activity or 75 minutes of vigorous activity per week, in addition to two or three weekly strength training sessions109,110. High-intensity interval exercise is also an effective option111,112. It is important to reduce sedentary time, incorporating active breaks during the day and fostering everyday movements such as walking, climbing stairs, or brief stretching, which favors long-term adherence113.
Other recommendations
Sleep should be evaluated as part of the comprehensive MASLD approach, as obstructive sleep apnea is frequent in these patients and is associated with a higher risk of hepatic fibrosis114. Systematic screening is recommended (Stop-Bang scale or Epworth sleepiness scale) and, if risk is detected, referral to a sleep unit. Treatment with continuous positive airway pressure, together with weight loss, can improve hepatic evolution and CVR115.
Smoking also contributes to MASLD progression, so complete cessation should be recommended19,116,117. Complementarily, it has been identified that the exposome, that is, exposure to environmental pollutants, such as fine particles and nitrogen dioxide, increases the risk of developing or worsening the disease, so it should be considered within the preventive approach118,119.
Finally, the success of any intervention in MASLD largely depends on behavioral support20. It is essential to establish realistic goals, conduct structured follow-up, identify individual barriers, and offer continuous support. Tools such as self-registration, frequent feedback, use of mobile technologies, and support from multidisciplinary teams, including psychologists or health coaches, are key to maintaining long-term changes and improving clinical outcomes113,120.
Rational use of pharmacological therapies in MASLD: an integrated cardio-RENO-hepato-metabolic framework (Table 6)
Table 6. Therapeutic options in MASLD and cardiovascular risk
| Intervention | Hepatic effect | Cardiovascular effect | Cost and availability |
|---|---|---|---|
| Weight loss (7-10%) | ↓ Steatosis, improves fibrosis | ↓ Global risk | Very cost-effective |
| Mediterranean or DASH diet | Improves steatosis | ↓ Cardiovascular events | High availability |
| Regular exercise | ↓ Steatosis, improves insulin sensitivity | ↓ Cardiovascular risk, improves VO2 max | Very cost-effective |
| Statins | Safe, ↓ mild steatosis | ↓ Cardiovascular mortality | Low cost |
| GLP-1 receptor agonists | ↓ Hepatic fat, improves NASH | ↓ MACE (in type 2 diabetes) | High cost |
| SGLT2 inhibitors | ↓ Hepatic steatosis | ↓ Heart failure and cardiovascular mortality | High cost |
| Resmetirom | ↓ Histological fibrosis | No robust cardiovascular data yet | High cost, restricted availability |
DASH: Dietary Approaches to Stop Hypertension; GLP-1: glucagon-like peptide-1; MACE: major adverse cardiovascular events; SGLT2: sodium-glucose cotransporter-2; VO2 max: maximal oxygen consumption.
The most cost-effective interventions remain lifestyle modification and statin use. Innovative drugs (GLP-1 agonists, SGLT2 inhibitors, resmetirom) should be prioritized in high-risk subgroups, due to their limited availability and high cost.
Pharmacotherapy in MASLD should be understood as part of a global strategy that is not limited to the liver but integrates prevention and treatment of CVR and metabolic risk. Both the 2024 joint European EASL-EASD-EASO guidelines20 and the 2023 American Association for the Study of Liver Diseases (AASLD) guideline121 agree that hepatic fibrosis, more than inflammation, is the most robust prognostic determinant of global and cardiovascular mortality. This implies that the intensity of pharmacological intervention should be graduated according to the presence of significant fibrosis (≥ F2), which marks an advanced vascular risk phenotype and requires an intensive strategy for controlling lipids, blood pressure, glucose, and body weight.
Statins and other lipid-lowering agents: the forgotten foundation
Statins constitute a cornerstone of pharmacological management in MASLD. They are capable of reducing LDL-C and apoB, and consequently vascular risk safely. A post hoc analysis of the GREACE study demonstrated that their use in patients with coronary disease and mild hepatic alterations not only reduced cardiovascular events but even improved hepatic parameters122. However, they remain underprescribed in clinical practice. In high or very high-risk patients, lipid objectives should be strict, defined in terms of LDL-C, non-HDL-C, and apoB, following ESC 2021 recommendations19. When the goal is not reached with high-intensity statins, ezetimibe should be added, and with risk persistence, resort to association with bempedoic acid, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors or inclisiran123. Lipoprotein (a) constitutes a hereditary factor of residual risk and may in the future justify the use of specific therapies, such as pelacarsen or olpasiran124.
Incretins and glucagon-like peptide-1 (GLP-1) receptor agonists: a paradigm shift
GLP-1 receptor agonists have gone from being hypoglycemic agents to becoming multi-organ modulation therapies. In March 2025, the Food and Drug Administration (FDA) approved semaglutide for MASH with significant fibrosis by producing histological NASH (Non-Alcoholic Steatohepatitis) resolution in 62.9% of patients125 and fibrosis improvement. Additionally, the SELECT trial, with more than 17,600 patients with obesity and CVD without diabetes, documented a 20% reduction in MACE126. Liraglutide also showed NASH resolution in the LEAN trial (39% vs. 9% with placebo)127 and reduction of cardiovascular events in the LEADER study128. Semaglutide, for its part, reduced MACE in the SUSTAIN-6 trial129.
The immediate future is even more ambitious. Dual GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonists, such as tirzepatide, have demonstrated weight losses exceeding 20% and significant hepatic improvements130. GLP-1 and glucagon agonists, such as cotadutide, have shown reductions in hepatic fat and triglycerides131. And triple agonists, such as retatrutide, have achieved 24% weight reductions and notable decreases in hepatic fat in phase II studies132. Thus, incretin drugs have ceased to be “antidiabetics” to position themselves at the center of a cardio-hepato-metabolic arsenal.
Sodium-glucose cotransporter-2 (SGLT2) inhibitors: nephro- and cardioprotection in one step
SGLT2 inhibitors have consistently demonstrated their impact on heart failure and chronic kidney disease. In the EMPA-REG OUTCOME study, empagliflozin reduced cardiovascular mortality in type 2 diabetes patients at high risk133. Their utility in MASLD is enhanced when albuminuria, reduced glomerular filtration rate, or heart failure coexist134. The combination of an SGLT2 inhibitor and a GLP-1 RA offers metabolic and hemodynamic complementarity, adding for weight, glucose, and arterial stiffness control.
Pioglitazone: histological efficacy, selective use
Pioglitazone maintains a place in the treatment of patients with NASH and type 2 diabetes. In a long-term trial, it improved necroinflammation and fibrosis, although with adverse effects of weight gain and edema135. Therefore, its use is recommended only in selected profiles, when insulin resistance is prominent and the benefit-risk balance is favorable.
Resmetirom: the first approved antifibrotic
In 2024, the FDA approved resmetirom as the first antifibrotic for MASH with F2-F3 fibrosis, for demonstrating fibrosis improvement and MASH resolution, in addition to reducing LDL-C, apoB, and triglycerides136.
Clinical sequencing and adherence
In fibrosis ≥ F2 or with very high CVR, the sequence should begin with high-intensity statins, add ezetimibe and a PCSK9 inhibitor, if necessary, introduce a GLP-1 agonist (semaglutide, liraglutide), consider an SGLT2 inhibitor in the presence of heart failure, albuminuria, or reduced glomerular filtration rate, add resmetirom in MASH with F2-F3 fibrosis, and reserve pioglitazone for patients with NASH and type 2 diabetes.
To this is added a frequently forgotten aspect: therapeutic adherence. Studies in hypertension and secondary prevention show that up to 40% of patients chronically abandon preventive treatments, which is associated with an increase in events and mortality137. Motivational interviewing, simplification of dosing schedules, and use of digital reminders are tools that should be integrated into clinical follow-up. Ultimately, pharmacotherapy in MASLD must leave behind the hepatocentric vision and assume it as an integrated cardio-RENO-hepato-metabolic strategy.
Longitudinal follow-up of cardiovascular risk in patients with MASLD: practical and integrated framework
CVR in patients with MASLD is not a static photograph but a dynamic process that should be addressed within the framework of a cardio-hepato-renal-metabolic continuum. Fibrosis ≥ F2 should be considered a threshold of advanced CVR20,46,138.
Baseline assessment: mapping risk
The first clinical act should be exhaustive and integrate a complete cardiometabolic history (background, life habits, tobacco and alcohol consumption, diet, physical activity, and sleep), a targeted examination (BMI, waist circumference, protocolized blood pressure), and laboratory tests with lipid profile, including apoB, HbA1c, creatinine with glomerular filtration estimation and albumin/creatinine ratio. Determination of lipoprotein (a) at least once in life adds prognostic value. In parallel, fibrosis should be stratified using FIB-4, and when the result is intermediate or elevated, or there is discordance with the clinical phenotype, complemented with elastography to assess hepatic stiffness. This core should be completed with a global cardiovascular health metric, such as Life’s Essential 8 (LE8), which evaluates diet, physical activity, smoking, sleep, BMI, non-HDL cholesterol, glucose, and blood pressure. A high LE8 is associated with lower MASLD incidence and better prognosis139,140. Albuminuria deserves special attention, as it constitutes a marker of target organ damage and increased CVR, conditions both blood pressure objectives and drug selection (angiotensin-converting enzyme inhibitors [ACEIs], angiotensin II receptor antagonists [ARBs], SGLT2 inhibitors, finerenone in diabetic nephropathy), and its persistence should be confirmed in at least two of three determinations37.
Dynamic and stratified follow-up
The re-evaluation rhythm should be adapted to risk. In patients without fibrosis or major comorbidity, an annual review with blood pressure, waist circumference, LE8, lipids/apoB, HbA1c, albumin/creatinine ratio, transaminases, and FIB-4 is sufficient. In patients with diabetes, hypertension, or dyslipidemia, the interval shortens to 6-12 months. In those with fibrosis ≥ F2 or established CVD, close control is recommended every 3-6 months during the first year of intensive intervention, with FIB-4 repetition at 6-12 months and annual elastography (or earlier in case of clinical or biochemical progression). Short-term review (3-6 months after first visit) should focus on verifying adherence to diet, exercise, and medication, identifying possible barriers, and adjusting the plan. In this period, basic parameters are sufficient in low-risk patients, while in those with significant fibrosis, it is advisable to add lipid profile and transaminases.
Vascular imaging and reclassification
In intermediate-risk scenarios or in case of discordance between traditional risk scores and clinical phenotype, vascular imaging is determinant. The CAC score is the most validated test: values ≥ 100 Agatston units require intensifying cardiovascular prevention. In MASLD, the prevalence of CAC ≥ 300 is elevated, and an intermediate or high FIB-4 predicts accelerated CAC progression39,77. If CAC determination is not available, measurement of carotid intima-media thickness or detection of carotid plaques adds value, and in MASLD with biopsy, fibrosis is associated with elevated intima-media thickness and greater plaque burden89. Pulse wave velocity offers an additional phenotyping of arterial stiffness with prognostic value90.
Intervention and adjustments according to results
Follow-up should not be limited to monitoring but requires intensive optimization of risk factors:
- – Lipids: high-intensity statins as first line, with objectives < 40 mg/dl in extreme risk, < 55 mg/dl LDL-C in very high risk, and < 70 mg/dl in high risk, and apoB < 65 mg/dl as complementary target139. If not reached, add ezetimibe, bempedoic acid, and consider a PCSK9 inhibitor or inclisiran.
- – Blood pressure: objective < 130/80 mmHg if tolerated; initiate ACEI/ARB in the presence of albuminuria, add calcium antagonist and/or thiazide or similar diuretic, and spironolactone if it persists outside objectives140.
- – Glucose and weight/adiposity: semaglutide reduces cardiovascular events in patients with obesity without diabetes126 and with diabetes with/without obesity, improves hepatic histology125; SGLT2 inhibitors reduce hospitalizations for heart failure, renal events, and cardiovascular mortality133; resmetirom has demonstrated histological benefit and a cardioprotective lipid profile136.
- – Fibrosis ≥ F2 confirmed: closer follow-up, consider resmetirom, and prioritize GLP-1 agonists or SGLT2 inhibitors according to metabolic phenotype.
- – Significant subclinical atherosclerosis (CAC ≥ 100 or elevated carotid intima-media thickness or carotid plaque or ankle-brachial index): apply intensive prevention strategies as in very high-risk patients, even without events.
Adherence and long-term goals
The success of follow-up requires structuring SMART (Specific, Measurable, Achievable, Relevant, Time-bound) objectives: 7-10% weight loss in 6-12 months, ≥ 150-300 min/week of moderate aerobic exercise or 75-150 min of vigorous exercise plus 2-3 strength sessions, 7-8 h of regular sleep, tobacco cessation, and achievement of personalized lipid, blood pressure, and glucose goals. LE8 should be recalculated every 6-12 months to direct intervention toward the poorest-scoring domains. Therapeutic adherence is critical: about 50% of patients abandon chronic preventive treatments within 1 year, with increased total and cardiovascular mortality137. In MASLD, where prevention is multimodal and prolonged, this phenomenon is especially damaging. Follow-up should incorporate adherence assessment, digital tools (reminders, telemonitoring, telecoaching), motivational interviewing, and screening for social determinants (food insecurity, obesogenic environments), with referral to community resources when possible. Finally, outcome analysis should encompass both cardiovascular events and hepatic progression, as well as non-cardiovascular mortality. To minimize biases, in competing risk contexts, Fine-Gray subdistribution models should be employed81.
In summary, longitudinal follow-up in MASLD requires a care model that combines precise initial stratification, risk-adjusted periodic re-evaluation, integration of hepatic, cardiovascular, and renal biomarkers, vascular imaging when pertinent, phenotype-based therapeutic intensification, and robust adherence strategies. The hepatologist must assume that caring for the liver in MASLD is inseparable from preventing atherothrombosis, protecting the kidney, and preserving the heart.
Conclusions (Table 7)
Table 7. Decalogue of practical advice in MASLD patient consultation
| No. | Practical advice (what to do in consultation) | References |
|---|---|---|
| 1 | Weight: set objective of ≥ 7-10% loss in 6-12 months; design maintenance plan and periodic follow-up; avoid unrealistic goals that demotivate | 92,93 |
| 2 | Diet: prioritize Mediterranean diet (including variants such as green-Med); alternatives DASH or plant-based; reduce free sugars, fructose, and ultraprocessed foods; adapt to the patient’s context | 94,97,99,103 |
| 3 | Exercise: prescribe ≥ 150 min/week moderate or ≥ 75 min/week vigorous; combine aerobic + strength; consider high-intensity interval exercise in motivated patients; break sedentarism with active breaks | 109,111,113 |
| 4 | Sleep and obstructive sleep apnea: screen apnea with STOP-Bang in at-risk patients (snoring, resistant hypertension, somnolence); refer for study and treatment | 114,114 |
| 5 | Alcohol and coffee: reinforce absolute alcohol abstinence; recommend coffee consumption (2-3 cups/day) unless contraindications, due to its association with lower fibrosis risk | 106,107,108 |
| 6 | Behavioral and organizational: accompany all recommendations with goal setting, self-registration, periodic feedback, and digital support; refer to multidisciplinary programs if failure at 3-6 months | 20 |
| 7 | Tobacco: inquire at each consultation; give brief advice; refer to specialized cessation programs, given its association with fibrosis progression and cardiovascular risk | 116,117 |
| 8 | Environmental exposome: raise awareness about pollution (PM2.5, PM10, NO2); recommend reducing exposure (e.g., avoid exercise during pollution peaks) | 118,119 |
| 9 | Motivation: use motivational interviewing; center communication on achievable accomplishments; avoid counterproductive messages about rapid weight losses | 113 |
| 10 | Personalization and care network: adapt plan to age, comorbidity, social situation, and preferences; foster multidisciplinary approach (nutrition, psychology, rehabilitation, cardiovascular risk) | 20 |
This decalogue synthesizes practical evidence-based measures for hepatology consultation in patients with MASLD. It integrates lifestyle interventions with emphasis on personalization and multidisciplinary work, aimed at improving adherence and cardiovascular and hepatic prognosis.
MASLD constitutes one of the most prevalent chronic diseases, affecting more than a quarter of the world’s adult population, with a growing trend and special impact on individuals of working age141. Its character is not exclusively hepatic; it is a systemic condition, closely linked to vascular risk, to the point that CVD constitutes the principal cause of mortality in these patients142. Advanced fibrosis must be recognized as the main prognostic marker, integrating hepatic and cardiovascular prevention143. This knowledge requires overcoming the dichotomy between hepatology and cardiology, understanding risk in a cardio-hepato-renal-metabolic continuum. In practice, this implies two simultaneous actions: screen MASLD in patients with CVD, and vice versa, screen CVD in all patients with MASLD, independently of whether they present classic risk factors or not144. Vascular risk assessment should be supported by validated scales, non-invasive fibrosis tests (FIB-4, elastography), and, when there is intermediate risk or discordance, implementation of complementary techniques to reclassify CV risk145.
In terms of treatment, MASLD presents two critical aspects: hepatic and cardiovascular. Intensive lifestyle modification remains the cornerstone: weight loss, cardio-healthy dietary pattern (Mediterranean diet as reference), aerobic and strength exercise, sleep hygiene, and tobacco146 and alcohol cessation. In parallel, the development of drugs with dual efficacy (GLP-1, SGLT2, resmetirom) opens a new era of cardio-hepatic cardio-RENO-hepatic-metabolic intervention147. Optimal approach requires multidisciplinary teams, integrating hepatologists, cardiologists, endocrinologists, internists, nephrologists, and primary care professionals, guaranteeing coordinated screening, prevention, and intervention circuits148. This vision has been reflected in the multidisciplinary European guidelines, which emphasize the need for full integration into health systems149.
In conclusion, MASLD is a prevalent, multisystem disease with a high health burden, whose impact transcends the liver. It significantly increases mortality, especially from cardiovascular causes, and requires rigorous and periodic vascular risk assessment, as well as bidirectional screening between hepatology and cardiology. Treatment should combine lifestyle changes and dual-benefit therapies, under the leadership of multidisciplinary teams capable of addressing the cardio-RENO-hepatic-metabolic continuum. Only through this will it be possible to reduce the clinical and social burden of MASLD and improve the global prognosis of these patients150.
Funding
This project has received funding from the European Horizon’s Research and Innovation Programme HORIZON-HLTH-2022-STAYHLTH-02 (agreement No 101095679) and from the Health Research Fund, Carlos III Health Institute, Spain (PI22/01853).
Conflicts of interest
The authors declare no conflicts of interest.
Ethical considerations
Protection of persons and animals. The authors declare that no experiments were conducted on humans or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data or require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The authors declare that they used artificial intelligence for writing this manuscript, specifically ChatGPT5 Thinking for final editing of the chapter and configuration of tables.
