Introduction
In 2023, non-alcoholic fatty liver disease (NAFLD) was redefined as metabolic dysfunction-associated steatotic liver disease (MASLD), and likewise non-alcoholic steatohepatitis (NASH) was redefined as metabolic dysfunction-associated steatohepatitis (MASH). Within this new classification, a new category was proposed, currently termed MetALD (metabolic dysfunction and alcohol-related liver disease), which encompasses patients who meet criteria for MASLD and who also have risky alcohol consumption1, defined as 140-350 g/week (20-50 g/day) in women and 210-420 g/week (30-60 g/day) in men2. The proposal, according to this new nomenclature, for establishing MetALD criteria is shown in figure 1.
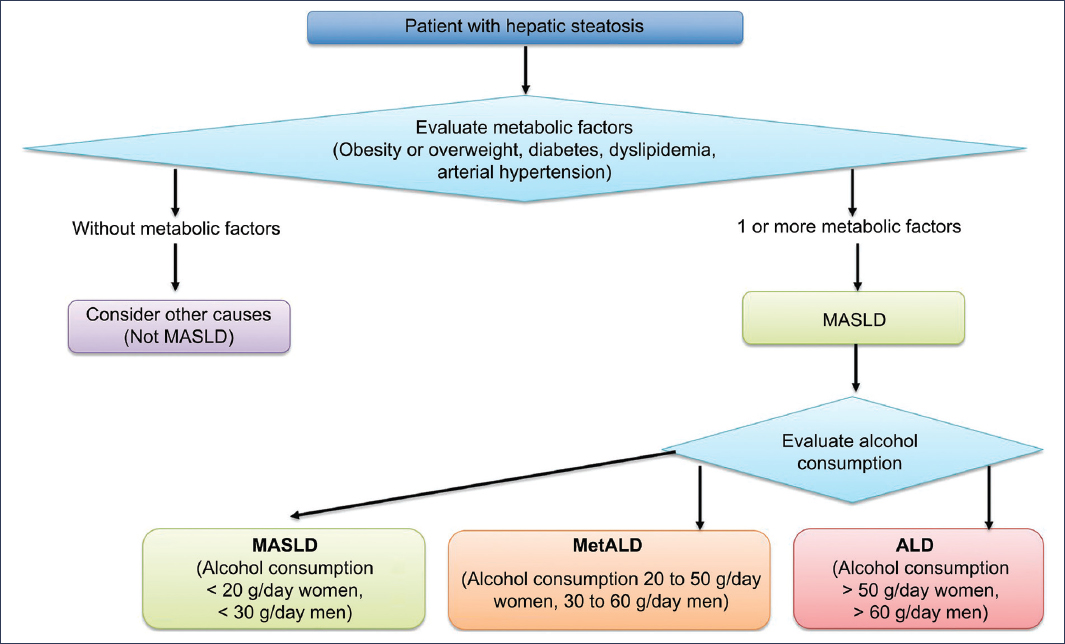
Figure 1. Criteria for considering MetALD within the new nomenclature for hepatic steatosis
Controversies regarding the new nomenclature and definition of MetALD
There is ambiguity in alcohol consumption thresholds, as the limits currently proposed to define risky alcohol consumption lack scientific robustness, have been mostly defined on an empirical basis, and vary across different publications3. On the other hand, the term MetALD represents a conceptually imprecise overlap between MASLD and alcohol-associated liver disease (ALD). The diagnostic distinction between metabolic or alcoholic predominance is not always clear and may depend excessively on subjective clinical judgment4. Furthermore, although it is recognized that alcohol consumption potentiates the progression of fibrosis, cirrhosis, and hepatocellular carcinoma, robust prospective studies are still lacking that clearly differentiate the risks in individuals with MetALD compared to MASLD or ALD alone5.
In the research field, another major challenge is that this new classification requires reevaluating historical databases and ongoing clinical trials, which may complicate longitudinal comparisons and question the validity of previous results6.
Epidemiology of MetALD
There is significant underreporting regarding alcohol consumption, since generally, to assess it, self-reported questionnaires are applied, which underestimate actual intake by 20-40%, potentially undervaluing the true burden of MetALD7.
The prevalence of MetALD is higher in Hispanic populations and Eastern Europe, while it is lower in Asia, but with an increasing trend among young people in urban centers8.
Studies in the general population conducted in the United States and Europe suggest that MetALD affects approximately 3-5% of the adult population9,10. In Asia, figures are reported between 1% and 3%, probably related to cultural differences in alcohol consumption11. In Latin America, where high obesity prevalence and high alcohol consumption coexist, prevalences range between 8 and 10%12. However, the prevalence of MetALD increases when evaluated in patients with a pre-established diagnosis of MASLD, in whom it has been reported that between 15% and 30% meet MetALD criteria13.
The risk of having MetALD is particularly high in middle-aged men with visceral obesity and type 2 diabetes mellitus14. It is also known that patients with MetALD have a higher risk of advanced fibrosis compared to those who only meet MASLD criteria15. Finally, studies have shown that the risk of hepatocellular carcinoma is substantially higher (incidence rate of 1.847 per 1000 person-years) in patients with MetALD and fibrosis-4 index (FIB-4) ≥ 1.3, compared to other etiologies or MASLD alone16.
Pathogenesis of ALD and MASLD: differences and similarities
Although ALD and MASLD differ in their primary triggering factors (excessive alcohol consumption in ALD and insulin resistance/obesity in MASLD), they share multiple common pathophysiological pathways that explain why there is an overlapping clinical spectrum and even the mixed condition MetALD4. In MetALD, on the one hand, chronic and excessive ethanol consumption converges, causing direct toxicity in hepatocytes, generating alterations in lipid metabolism, and promoting inflammation17, and on the other hand, insulin resistance, visceral obesity, and metabolic syndrome, which induces a state of hepatic lipotoxicity18 (Table 1).
Table 1. Comparison of the pathogenesis of ALD and MASLD
| Mechanism | ALD | MASLD | Key convergence points |
|---|---|---|---|
| Primary triggering factor | Chronic and excessive alcohol consumption | Insulin resistance, visceral obesity, metabolic syndrome | Both require a sustained external stimulus that alters hepatic homeostasis |
| Lipid metabolism | Acetaldehyde and excess NADH inhibit β-oxidation and promote lipogenesis | Hyperinsulinemia stimulates SREBP-1c and ChREBP → increases fatty acid synthesis | Accumulation of triglycerides (hepatic steatosis) |
| Oxidative stress | Ethanol metabolism via CYP2E1 → ROS and acetaldehyde adducts | Lipotoxicity from free fatty acids → lipid peroxidation | Oxidative stress and mitochondrial damage as central axes |
| Inflammation and innate immunity | Intestinal dysbiosis, increased permeability → LPS activates TLR4 in Kupffer cells → TNF-α, IL-1β | Dysfunctional adipose tissue releases TNF-α, IL-6; decreased adiponectin | Activation of Kupffer cells and chronic inflammatory response |
| Fibrogenesis | Acetaldehyde and ROS activate hepatic stellate cells | Chronic metabolic signals (lipotoxicity, insulin resistance) activate stellate cells | Progressive fibrosis mediated by extracellular matrix deposition |
| Carcinogenesis | Direct mutagenesis by acetaldehyde, ROS, and epigenetic alterations | Chronic inflammation, obesity, hyperinsulinemia → activation of IGF/PI3K-AKT pathways | HCC can appear even in the absence of cirrhosis |
| Clinical spectrum | Steatosis → alcohol-related hepatitis → fibrosis → cirrhosis → HCC | Steatosis → MASH → fibrosis → cirrhosis → HCC | Similar progressive spectrum course |
HCC: hepatocellular carcinoma; ChREBP: carbohydrate response element-binding protein; CYP2E1: cytochrome P2E1; IGF/PI3K-AKT: insulin-like growth factor/phosphoinositide 3-kinase – protein kinase B; IL-1β: interleukin 1 beta; IL-6: interleukin 6; LPS: lipopolysaccharide; MASH: metabolic dysfunction-associated steatohepatitis; NADH: reduced nicotinamide adenine dinucleotide; ROS: reactive oxygen species; SREBP-1c: sterol regulatory element-binding protein 1c; TNF-α: tumor necrosis factor alpha; TLR4: toll-like receptor 4.
Alterations in lipid metabolism
In both ALD and MASLD, intrahepatic accumulation of triglycerides (steatosis) occurs, and there is an increase in de novo lipogenesis and an alteration in mitochondrial β-oxidation. However, in ALD, ethanol oxidation generates an excess of reduced nicotinamide adenine dinucleotide (NADH), which in turn favors lipogenesis and blocks fatty acid oxidation, while in MASLD, hyperinsulinemia stimulates key transcription factors that regulate lipogenesis; among the most relevant are sterol regulatory element-binding protein 1c (SREBP-1c) and carbohydrate response element-binding protein (ChREBP), inducing greater fatty acid synthesis17–20.
Oxidative stress and mitochondrial damage
In ALD, ethanol metabolism via cytochrome P2E1 (CYP2E1) produces reactive oxygen species (ROS) and acetaldehyde, which damage proteins and deoxyribonucleic acid17. In MASLD, excess free fatty acids induce lipotoxicity, mitochondrial dysfunction, and lipid peroxidation20,21. Thus, in MetALD, oxidative stress and mitochondrial damage are mechanisms that trigger inflammation and progression to fibrosis17,20,22
Inflammation and innate immune system
Ethanol increases intestinal permeability, favoring bacterial translocation and activation of toll-like receptor 4 (TLR4) by lipopolysaccharide (LPS), which is capable of triggering an inflammatory response (tumor necrosis factor alpha [TNF-α], interleukin 1β [IL-1β], and interleukin 6 [IL-6)17,19,22. On the other hand, in MASLD, insulin resistance and lipid peroxidation favor a proinflammatory profile (TNF-α, IL-6, leptin) and reduction of adiponectin20,21. In both conditions, there is activation of Kupffer cells and recruitment of immune cells, which amplifies hepatic inflammation19,22.
Fibrogenesis
In both ALD and MASLD, chronic inflammation activates hepatic stellate cells, promoting extracellular matrix deposition and favoring fibrosis progression17,19,21–23. In ALD, acetaldehyde and ROS act directly on stellate cells22,23, while in MASLD, fibrosis is more related to chronic metabolic signals (lipotoxicity, insulin resistance) that activate stellate cells21.
Carcinogenesis
In ALD, there is direct mutagenesis by acetaldehyde and oxidative stress, in addition to epigenetic effects induced by alcohol17,19,21. In MASLD, carcinogenesis is associated with obesity, diabetes, and chronic inflammation, even in the absence of cirrhosis. In both diseases, inflammation, mitochondrial dysfunction, and altered insulin or insulin-like growth factor signaling contribute to the development of hepatocellular carcinoma16,17,21,22.
Evaluation of MetALD
The diagnosis of MetALD requires the integration of four fundamental aspects: 1) evidence of hepatic steatosis, 2) meeting MASLD criteria, 3) demonstrating risky alcohol consumption, and 4) stratifying fibrosis risk1,3,4 (Fig. 2).
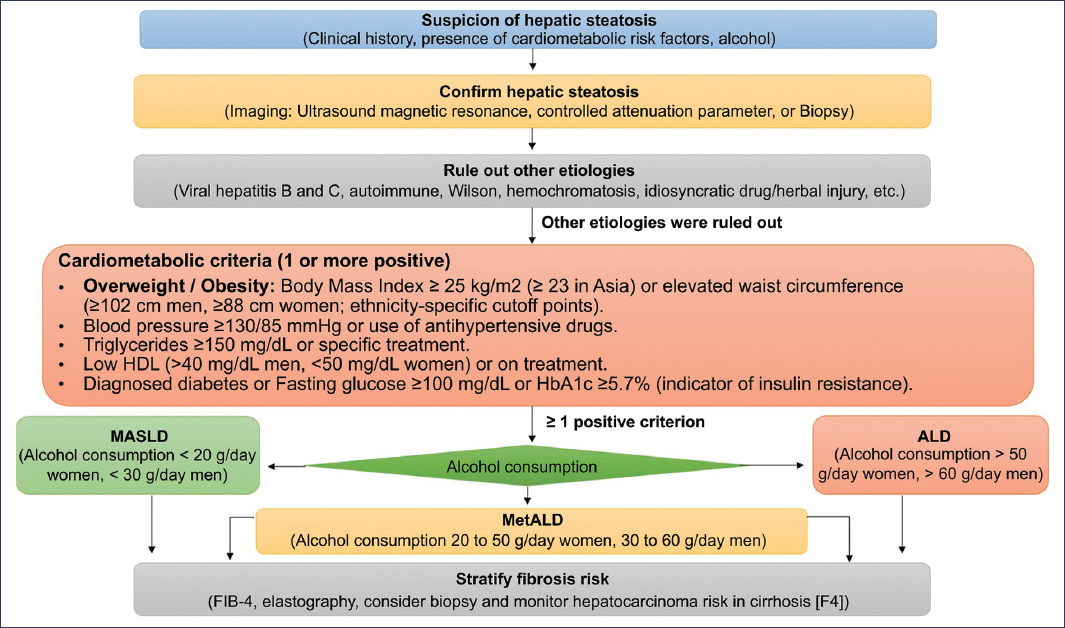
Figure 2. Diagnostic algorithm for MetALD.
Therapeutic strategies
The treatment of MetALD is based on alcohol abstinence, strict control of metabolic factors, and surveillance for complications, complemented with therapies under investigation. There is not yet a specific approved drug, but the combination of behavioral strategies, pharmacological treatments for comorbidity, and hepatic surveillance constitutes the current therapeutic basis4 (Table 2).
Table 2. Therapeutic strategies in MetALD1–4
| Strategy | Specific intervention | Evidence (key) | Expected hepatic effect | Level of recommendation#x002A; | Safety considerations |
|---|---|---|---|---|---|
| Alcohol | Total abstinence or intensive reduction; structured counseling; management of alcohol use disorder | Meta-analyses and cohorts: reduce decompensation and mortality | Reduce inflammation, fibrosis progression, and HCC risk | A | Assess withdrawal syndrome; refer to addiction services; objective tests (PEth/EtG) when possible |
| Drugs for alcohol | Naltrexone, acamprosate, baclofen (cirrhosis) | RCTs and series: increase abstinence; baclofen is useful in liver disease | Reduce relapses → indirect hepatic benefit | B | Naltrexone: avoid in acute hepatitis; baclofen: sedation; acamprosate: adjust in kidney impairment |
| Weight loss | Hypocaloric diet 500-1000 kcal/d; target 7-10% | RCTs and observational studies in MASLD: improve MASH and fibrosis | Reduce steatosis, NASH/MASH, and possibly fibrosis | A | Individualize if sarcopenia; avoid rapid losses in decompensated cirrhosis |
| Exercise | Aerobic + resistance ≥ 150 min/week | RCTs: reduce hepatic fat independent of weight | Reduces steatosis and increases insulin sensitivity | A | Adapt in cirrhosis; prevent sarcopenia |
| Dietary pattern | Mediterranean diet; less fructose and trans/saturated fats | RCTs and cohort studies: reduce steatosis and insulin resistance | Reduce lipotoxicity and inflammation | B | Sustained adherence; clinical nutrition support |
| T2DM: GLP-1R agonists | Semaglutide/liraglutide | RCTs: MASH resolution; weight loss | Reduces steatohepatitis; possible reduction in fibrosis (under study) | A/B | GI, cholelithiasis; monitor in previous pancreatitis |
| T2DM: SGLT2 inhibitors | Empagliflozin/dapagliflozin | RCTs: reduce hepatic fat by imaging | Reduce steatosis and inflammation | B | Fungal UTIs; euglycemic ketoacidosis (rare) |
| T2DM: metformin | Metabolic first line | Cohort studies: CV benefit; no clear effect on NASH | Systemic metabolic benefit | C | GI; avoid in advanced kidney impairment with acidosis risk |
| Dyslipidemia | Statins | Trials and cohort studies: safe in compensated liver disease; reduce CV events; signal of HCC reduction | CV benefit; possible HCC reduction | A | Monitor transaminases; safe in compensated cirrhosis |
| Arterial hypertension | Individualized intensive control | Cohort studies: reduce CV events | Indirect on prognosis | B | ACE-I/ARB with caution in advanced ascites |
| Procedures | Bariatric surgery (selected) | Cohort studies and RCTs: MASH resolution and fibrosis reduction | Reduce steatohepatitis and fibrosis | B | Rigorous selection; perioperative risk in cirrhosis |
| Investigational agents | PPAR pan-agonist (lanifibranor), FXR (cilofexor/obeticholic), ACC/FASN inhibitors | Phase II–III studies: histological and enzymatic improvement | Reduce inflammatory activity and lipogenesis; potential antifibrotic effect | B (under investigation) | Access in trials; AE pruritus (FXR), lipid profile (ACC) |
| Surveillance and complications | Elastography/FIB-4; US/AFP every 6 months in advanced fibrosis | Guidelines | Reduces mortality through early HCC diagnosis | A | Ensure adherence; referral pathway to liver transplant program |
| Liver transplantation | Decompensated cirrhosis or incurable HCC | Standard | Long-term survival | A | Selection criteria; addiction support |
* Guidance recommendation level (A: strong/high quality; B: moderate quality; C: limited quality/consensus).
ACC: acetyl coenzyme A carboxylase; AFP: alpha-fetoprotein; ARB: angiotensin II receptor antagonists; HCC: hepatocellular carcinoma; CV: cardiovascular; T2DM: type 2 diabetes mellitus; AE: adverse events; RCT: randomized controlled trials; FASN: fatty acid synthase; FIB-4: fibrosis-4 index; FXR: farnesoid X receptor; GI: gastrointestinal; GLP-1R: glucagon-like peptide 1 receptor; ACE-I: angiotensin-converting enzyme inhibitors; kidney impairment: renal insufficiency; UTI: urinary tract infections; PEth/EtG: phosphatidylethanol/ethyl glucuronide; PPAR: peroxisome proliferator-activated receptor; SGLT2: sodium-glucose cotransporter 2; liver transplant program: liver transplant program; US: ultrasound.
Conclusions
- − The introduction of the MetALD concept is conceptually valuable but faces significant challenges in its clinical and epidemiological application.
- − Prospective studies are needed to validate its prognostic utility and define clinically relevant thresholds with evidence.
- − It is important to avoid stigmatizing patients; the ideal approach should integrate multiple risk factors and emphasize patient-centered interventions.
- − Until more evidence is available, it would be prudent to consider alcohol as a modulator of continuous risk within MASLD, rather than delimiting rigid categories.
- − MetALD already affects millions of individuals worldwide, with prevalences of 3-10% in the general population and up to 30% in patients with metabolic risk.
- − Current data are from retrospective studies and self-reports, which limits the precision of estimates.
- − There is an urgent need for standardized definitions to estimate the global burden and for objective biomarkers of alcohol consumption and metabolic susceptibility, and prospective multinational studies are required to determine the true burden of advanced fibrosis and hepatocarcinoma attributable to MetALD.
- − MetALD should be understood as an emerging disease with significant epidemiological impact, whose early recognition will allow the design of more effective preventive strategies.
- − ALD and MASLD share key pathogenic mechanisms (steatosis, oxidative stress, inflammation, fibrogenesis), but differ in the initial stimulus (alcohol or metabolic dysfunction) and in some predominant mediators (acetaldehyde and dysbiosis in ALD, and insulin resistance and adipokines in MASLD). This pathophysiological convergence explains the emergence of the MetALD concept, which recognizes the synergistic interaction between alcohol and metabolic dysfunction in the development and progression of liver damage.
- − Current therapy for MetALD is based on strategies focused on maintaining alcohol abstinence and emphasizing metabolic control.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
F. Higuera-de la Tijera is a speaker for Sanfer, Grünenthal, Gilead, Abbott, Medix, and Adium. A. Servín-Higuera has no conflicts of interest.
Ethical considerations
Protection of human subjects and animals. The authors declare that no experiments were performed on human subjects or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data nor requires ethical approval. SAGER guidelines do not apply.
Statement on the use of artificial intelligence. The authors declare that they used artificial intelligence for writing this manuscript, specifically ChatGPT and Gemini to create the algorithms, figures, and tables for this article.
