Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD) has emerged in recent years as a significant global health problem. This new terminology replaces “non-alcoholic fatty liver disease” (NAFLD), incorporating diagnostic criteria based on metabolic dysfunction and providing a more precise framework for patient identification and stratification1.
MASLD is defined by the presence of hepatic steatosis (detected by imaging, histology, or biomarkers) without excessive alcohol consumption, and associated with at least one of five cardiometabolic risk factors: overweight or obesity, prediabetes or type 2 diabetes, hypertension, dyslipidemia, or low levels of high-density lipoprotein cholesterol1.
Its global prevalence is currently estimated at approximately 38% of the adult population, representing a 50% increase over the past 20 years, with regional variations2. Particularly in Latin America, the prevalence of MASLD is estimated at 20-40% of the general population, but this figure increases to approximately 70% in people with type 2 diabetes or obesity3.
MASLD is also associated with a significant increase in mortality. Between 2006 and 2023, MASLD-related mortality in the United States increased from 0.25 to 1.27 per 100,000 inhabitants, and is projected to reach 2.24 in 2040, with the largest increase in people ≥ 65 years (from 3.69 to 7.12 per 100,000), especially in non-metropolitan areas4.
Given its high prevalence and clinical burden, the initial evaluation of a patient with suspected MASLD is fundamental. This should address five main domains: clinical profile, initial laboratory studies, imaging studies to detect steatosis, assessment of liver fibrosis, and approach to precision diagnosis. This literature review aims to describe the elements of the initial evaluation of the patient with suspected MASLD.
Clinical profile of the patient with MASLD
In clinical practice, patients with MASLD are usually referred due to incidental findings of hepatic steatosis on imaging studies or elevated liver function markers. It is important to consider that the reference values used by many laboratories exceed optimal limits for this context; in MASLD, the normal range for alanine aminotransferase (ALT) is 29-33 U/l in men and 19-25 U/l in women5.
The initial evaluation should include6:
- − Targeted clinical history, including:
- Weight history and recent changes.
- Medical comorbidities and current or recent medications.
- Family history of type 2 diabetes, NAFLD, or cirrhosis.
- Screening for obstructive sleep apnea.
- Alcohol consumption (amount, pattern, and duration).
- − Physical examination focused on6:
- Amount and distribution of body fat (body mass index [BMI], waist circumference, body fat percentage).
- Signs of insulin resistance (dorsocervical fat pad, acanthosis nigricans).
- Manifestations of advanced liver disease (firm hepatomegaly, splenomegaly, prominent abdominal veins, ascites, gynecomastia, spider angiomas, palmar erythema).
In cases with an atypical clinical profile – for example, absence of metabolic comorbidity – or presence of symptoms suggesting alternative etiologies, less common causes of steatosis or steatohepatitis should be ruled out, such as hypobetalipoproteinemia, lysosomal acid lipase deficiency, nutritional deficiencies, Wilson’s disease, or celiac disease7.
Likewise, some drugs (amiodarone, 5-fluorouracil, irinotecan, tamoxifen, methotrexate, corticosteroids) can induce or worsen steatosis and should be identified in the initial assessment3.
To determine risk and perform screening, international guidelines recommend a targeted search for those patients at highest risk of clinically significant fibrosis (≥ F2) or cirrhosis, particularly those with type 2 diabetes, obesity with metabolic complications, family history of cirrhosis, or significant alcohol consumption6,8.
The risk extends to first-degree relatives – who have up to 12 times greater likelihood of presenting advanced fibrosis – and to unrelated household members, due to exposure to similar environmental factors and lifestyles9.
Within the clinical evaluation, etiologies that present with findings similar to MASLD should be ruled out; table 1 shows the criteria and tests to exclude other causes of liver disease10.
Table 1. Criteria and tests to exclude other causes of liver disease
| Category | Examples | Suggested tests |
|---|---|---|
| Viral hepatitis | HBV, HCV | Serologies for HBsAg, anti-HCV |
| Autoimmune diseases | Autoimmune hepatitis, primary biliary cholangitis | Antinuclear, antimitochondrial, and anti-smooth muscle antibodies |
| Genetic | Hemochromatosis, Wilson’s disease, alpha-1 antitrypsin deficiency | Serum ferritin, transferrin saturation, ceruloplasmin, alpha-1 antitrypsin |
| Drugs | Methotrexate, amiodarone, corticosteroids, valproic acid, 5-fluorouracil, irinotecan | Detailed pharmacological history |
| Alcohol | Significant consumption (> 21 g/week in men and > 14 g/week in women) | History of consumption and pattern |
HBsAg: hepatitis B surface antigen; HBV: hepatitis B virus; HCV: hepatitis C virus.
Modified from Cusi et al.8 and Frączek et al.10.
Initial laboratory studies
The initial evaluation in patients with suspected or diagnosed MASLD should include a basic set of tests to characterize hepatic and metabolic status, identify comorbidities, and rule out other causes of liver disease. Recommended determinations include6:
- − Liver profile.
- − Complete blood count with platelets.
- − Fasting plasma glucose.
- − Glycosylated hemoglobin.
- − Serum insulin.
- − Lipid profile.
- − Creatinine.
- − Urinary microalbumin or microalbumin/creatinine ratio.
- − Hepatitis C serology if not previously evaluated.
Depending on the clinical context, it may be necessary to investigate other causes of steatosis or steatohepatitis, especially in the presence of elevated liver enzymes, through autoimmune serologies, transferrin saturation, ceruloplasmin, or determination of alpha-1 antitrypsin (genotype or phenotype)11.
The identification of metabolic alterations is key in the diagnosis of MASLD. It is considered that if a patient meets at least one of the five established criteria (Table 2), this disease can be diagnosed in the presence of hepatic steatosis.
Table 2. Cardiometabolic risk factors in the definition of MASLD
| Metabolic factor | Criteria in adults |
|---|---|
| Overweight or obesity | BMI ≥ 25 and waist circumference ≥ 94 cm in men or ≥ 80 cm in women |
| Prediabetes or type 2 diabetes | Prediabetes: HbA1c 5.7-6.4% or fasting plasma glucose 100-125 mg/dl or glucose 140-199 mg/dl at 2 h of OGTT; type 2 diabetes: HbA1c ≥ 6.5% or fasting glucose ≥ 126 mg/dl or glucose ≥ 200 mg/dl at 2 h of OGTT; or treatment for type 2 diabetes |
| Triglycerides | ≥ 150 mg/dl or lipid-lowering treatment |
| HDL cholesterol | ≤ 40 mg/dl in men or ≤ 50 mg/dl in women, or lipid-lowering treatment |
| Blood pressure | ≥ 130/85 mmHg or treatment for hypertension |
HbA1c: glycosylated hemoglobin; HDL: high-density lipoproteins; BMI: body mass index; OGTT: oral glucose tolerance test.
Modified from Rinella et al.1.
Clinical guidelines recommend priority evaluation of people with type 2 diabetes, abdominal obesity associated with another metabolic risk factor, or persistent elevation of liver enzymes12. Although liver biopsy remains the reference method for staging hepatic steatosis, its systematic use is limited by risks, costs, and necessary resources, and is therefore reserved for specific cases6. In clinical practice, non-invasive biomarkers have gained relevance for estimating the presence of advanced fibrosis and predicting hepatic outcomes, a function that previously corresponded almost exclusively to biopsy12–14. Table 3 summarizes the components of non-invasive tests for the identification of advanced fibrosis in MASLD.
Table 3. Non-invasive tests for the identification of advanced fibrosis in patients with MASLD
| Indicator | Variables needed for calculation | Type of indicator |
|---|---|---|
| FIB-4 (fibrosis-4 index) | Age, AST, ALT, platelets | Serum score |
| NAFLD Fibrosis Score | Age, BMI, impaired fasting glucose (yes/no), AST/ALT ratio, platelets, albumin | Clinical-biochemical score |
| APRI (AST to platelet ratio index) | AST, upper normal limit of AST according to laboratory, platelets | Serum score |
| ELF (enhanced liver fibrosis test) | Hyaluronic acid, PIIINP, TIMP-1 | Serum collagen panel |
| Hepamet Fibrosis Score | Sex, age, HOMA-IR, presence of diabetes, AST, albumin, platelets | Clinical-biochemical score |
ALT: alanine aminotransferase; AST: aspartate aminotransferase; HOMA-IR: homeostatic model assessment for insulin resistance; BMI: body mass index; PIIINP: N-terminal propeptide of type III collagen; TIMP-1: tissue inhibitor of metalloproteinase 1.
Modified from Tacke et al.12, Abdelhameed et al.13, and Reinson et al.14.
Among these tests, the fibrosis-4 index (FIB-4) is the initial tool recommended for its availability and low cost6,8. This index is calculated from age, ALT, and aspartate aminotransferase (AST) values, and platelet count, with established cutoff points to determine whether the person has low, intermediate, or high risk of fibrosis. Figure 1 shows the algorithm for non-invasive evaluation of fibrosis in patients with MASLD. Low FIB-4 values allow ruling out advanced fibrosis and reevaluation every 1-3 years, while intermediate or high values require confirmation through hepatic elastography or, if not available, serum markers such as ELF (enhanced liver fibrosis test). In case of intermediate risk, intensive intervention on lifestyle and cardiometabolic factors for 12 months may be chosen, repeating FIB-4 before deciding on new tests12–14.
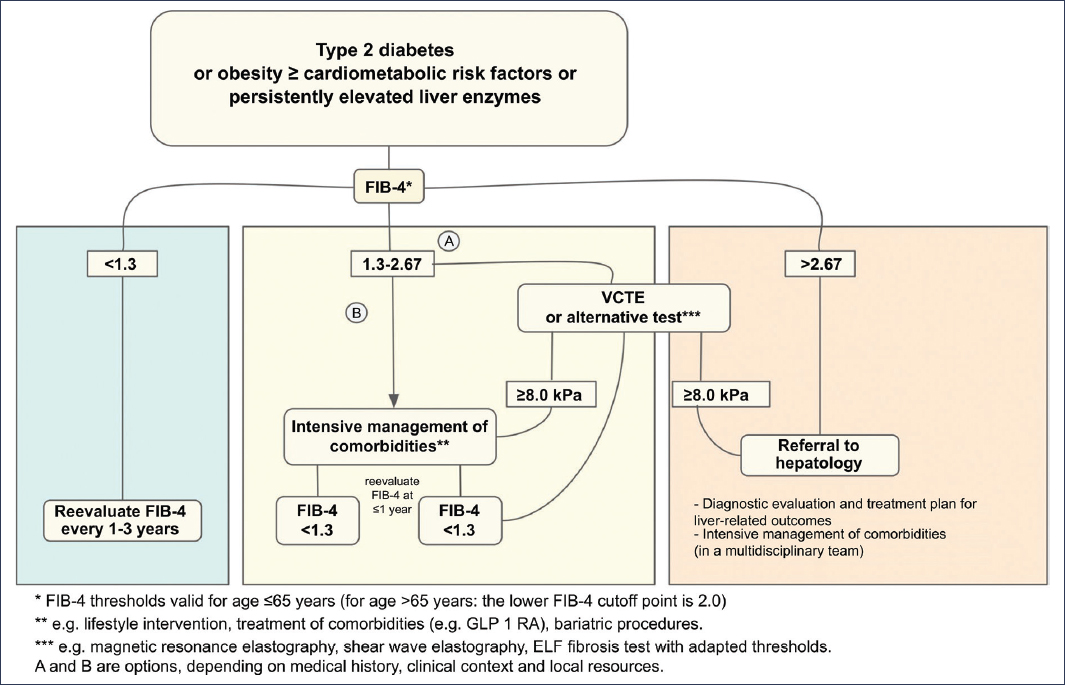
Figure 1. Proposed algorithm for non-invasive evaluation of fibrosis in patients with MASLD. This clinical algorithm proposes the stepwise use of non-invasive tools to stratify the risk of liver fibrosis in patients with MASLD. It begins with calculation of the FIB-4 index (fibrosis-4 index), using age, AST and ALT, and platelets. A low score allows ruling out advanced fibrosis and suggests periodic reevaluation every 1-3 years. In case of intermediate or high score, hepatic elastography is recommended, or alternatively, advanced serum tests such as ELF. Alternatively, intensive lifestyle intervention may be chosen, with subsequent reevaluation. This approach allows minimizing the use of invasive methods, such as biopsy, reserving them for cases in which diagnostic uncertainty persists. VCTE: vibration-controlled transient elastography. Modified from Tacke et al.12.
Imaging evaluation to detect steatosis
According to the previous definition, one of the requirements for establishing the diagnosis of this condition has been the demonstration of steatosis through some complementary study. The method for diagnosing steatosis should be chosen according to its availability, validity, and reliability. In addition to demonstrating steatosis, these methods can, to a greater or lesser extent, quantify the percentage of fat and classify it into grades.
Ultrasonography
It is an imaging method that stands out for being widely accessible. However, there are limitations in its performance, such as operator dependence, subjective evaluation, and limited capacity to quantify fat. The accepted criteria for visual evaluation of steatosis are15:
- − Bright liver with increased liver-kidney contrast.
- − Blurring of intrahepatic vessels and the diaphragm.
- − Loss of echoes in the posterior hepatic segments.
In a meta-analysis of 49 studies comparing liver histology with ultrasonography, 22 of which reported intra- or interobserver variability, with a total of 4720 patients, the capacity of ultrasound to discern between absence of steatosis and presence of moderate to severe steatosis was reported, with sensitivity (S) of 84.8%, specificity (E) of 93.6%, positive likelihood ratio of 13.3 and negative 0.16, and area under the curve of 0.93. Even at lower percentages of steatosis, S and E remained close to 90%16.
Another meta-analysis involving 2921 patients from 12 studies reported the following values: S 82%, E 80%, positive likelihood ratio 4.0, and negative 0.23. As the amount of steatosis increases, these values improve slightly17.
A third meta-analysis evaluated the capacity of ultrasound for the detection of steatosis with different cutoff points, and its performance was as follows15:
- − First group (> 0%, > 2%, > 5%): S 73.3% and E 84.4%.
- − Second group (> 10%, > 15%, > 20%): S 90.5% and E 69.6%.
- − Third group (> 25%, > 30%, > 33%): S 85.7% and E 85.2%.
- − Fourth group (> 50%, > 60%, > 66%): S 91.1% and E 91.9%.
Therefore, ultrasound is considered a tool with adequate performance for initial detection of hepatic steatosis, especially as the percentage of fat infiltration (demonstrated by biopsy) increases.
Controlled attenuation parameter
The controlled attenuation parameter (CAP) is a measurement performed by transient elastography. This technique has the ability to be measured simultaneously with fibrosis. CAP is an estimate of total ultrasonic attenuation at the central frequency of the probe used, and is expressed in dB/m. Its advantages include not requiring an imaging probe, not depending on operator subjectivity, and not requiring advanced imaging for its performance. Its value is not affected by the etiology of liver disease, which allows estimating the comorbidity of steatosis even in the case of liver disease from other causes18.
In a study that included 159 patients who underwent liver biopsy and CAP determination on the same day, a gradual increase in CAP was observed alongside the increase in the percentage of fat infiltration determined by histology, and the following findings in terms of sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and area under the ROC curve (AUROC) were reported19:
- − S0: 184.5 (127-200) dB/m.
- − S1: 213.5 (193-234) dB/m, AUROC 0.822, S 81.6%, E 76.5%, PPV 92.7%, and NPV 53.6%.
- − S2: 262.5 (237-94) dB/m, AUROC 0.956, S 93.5%, E 84.1%, PPV 84.6%, and NPV 93.2%.
- − S3: 324.0 (303-345) dB/m, AUROC 0.976, S 95.1%, E 89.0%, PPV 75.0%, and NPV 98.1%.
Furthermore, this same study, in its multivariate analysis, showed an independent correlation between higher CAP levels and other variables (BMI > 25, dyslipidemia, and MASLD etiology)19.
A meta-analysis of nine studies involving 1297 patients with MASLD demonstrated by biopsy showed that CAP has an S of 87% and an E of 91%20. Interestingly, in the subanalysis by degrees of steatosis, CAP showed a lower ability to quantify severe steatosis, attributing this deficiency, in part, to technical difficulties related to the distance between the skin and the liver capsule, which has been described as better when it is < 25 mm20. Such technical difficulties should be considered a priori with the objective of selecting the correct probe (M vs. XL) and improving diagnostic performance.
A meta-analysis of 61 studies with 10,537 patients reported the following performance through AUROC: S1 0.924, S2 0.794, and S3 0.778. Similarly, in this meta-analysis, it was observed that BMI ≥ 30 reduced the diagnostic capacity of CAP21.
Thus, CAP determination is considered to have limitations, but it represents a useful tool with the capacity to detect steatosis and discern among its various grades, with the significant advantage of being able to establish the degree of fibrosis simultaneously.
Magnetic resonance imaging with proton density fat fraction
Magnetic resonance imaging with determination of proton density fat fraction (MRI-PDFF) is considered an imaging study with the capacity to be quantitative and allow a reliable, reproducible diagnosis. PDFF is a tissue property that reflects the concentration of mobile triglycerides within that tissue, calculated as a rate and expressed as a percentage. Despite this, the PDFF percentage is not numerically equivalent to the histological fat percentage, but rather a correlation22.
A study evaluated, through linear regression, the cutoff points for establishing and quantifying the percentage of fat infiltration by MRI-PDFF. It established that S and E of 95% were achieved with cutoff points23:
- − ≥ 10%: S 3.7% and E 6.3%.
- − ≥ 20%: S 5.5% and E 8.0%.
- − ≥ 30%: S 8.0% and E 9.1%.
- − ≥ 40%: S 10.0% and E 10.1%.
A meta-analysis evaluated the capacity of magnetic resonance imaging for the detection of steatosis with different cutoff points, and its performance was as follows15:
- − First group (> 0%, > 2%, > 5%): S 82% and E 89.9%.
- − Second group (> 10%, > 15%, > 20%): S 90.0% and E 95.3%.
- − Third group (> 25%, > 30%, > 33%): S 97.4% and E 76.21%.
MRI-PDFF is considered an accurate, precise, reliable, reproducible method with less intra- and interobserver variability than other methods, with the advantage of being non-invasive. Furthermore, it can be used for quantitative evaluation of treatment response, which has only been done in clinical research protocols22.
The selection of the non-invasive diagnostic method for demonstrating steatosis will depend mainly on accessibility and availability. However, it should also depend on the reliability and reproducibility of its results, and on the objectives set when performing each study. In theory, a single non-invasive study should be sufficient to confirm or rule out steatosis. To date, it has not been established that the level of steatosis is clinically relevant or has an influence on the evolution of MASLD, so its repeated monitoring is reserved as a therapeutic objective in clinical trials24.
Evaluation of fibrosis
Histology
Liver biopsy with histological analysis of the obtained tissue remains the reference method for diagnosing steatosis. Its limitations are related to its invasive nature, the potential for inadequate sampling, and the low feasibility of performing repeated measurements during patient follow-up19. The interpretation and scoring of biopsies may present significant intra- and interobserver variability22.
There are various classification systems that allow assessment of MASLD severity through biopsy. Fibrosis is characterized by beginning in acinar zone 3, with a chicken-wire appearance, macronodular aspect, or mixed.
The Brunt system evaluates the severity of steatosis, ballooning, and inflammation, and evaluates fibrosis as follows: 0 = no fibrosis, 1 = perisinusoidal or portal fibrosis, 2 = perisinusoidal and portal/periportal fibrosis, 3 = bridging fibrosis, and 4 = cirrhosis.
The NASH-CRN (Non-Alcoholic Steatohepatitis Clinical Research Network) system includes description of steatosis, lobar inflammation, hepatocyte ballooning, and severity of fibrosis, assigning a numerical activity value ranging from 1 to 8.
The SAF (Steatosis Activity Fibrosis) system establishes the diagnosis of MASH based on inflammation and ballooning25.
Non-invasive evaluation
Non-invasive evaluation encompasses various methods in terms of their degree of complexity. Some of them are combined serum biomarkers that have been validated with the objective of predicting degrees of fibrosis significant for clinical and therapeutic decision-making.
NAFLD-FIBROSIS SCORE (NFS)
Derived from a group of 733 patients with MASLD demonstrated by biopsy, it involves as variables age, BMI, hyperglycemia, AST and ALT, platelets, and serum albumin. Values less than –1.455 correlate with ≤ F2 and values < 0.675 with ≥ F3. It has an NPV of 88-93% and a PPV of 82-90%26. It was validated in a 20-year cohort that included 302 patients, with follow-up of 11.9 ± 3.9 years, in which it had adequate performance for mortality27. Age ≥ 65 years has been an adjustment factor in this score, as it overestimates the figures and results in an increase in false positives, so in this age group < 0.12 for ≤ F2 and < 0.675 for ≥ F3 have been proposed28.
FIB-4
It is useful for ruling out advanced fibrosis with scores < 1.3, with an NPV of 90%. Its performance can be affected by age ≤ 35 or ≥ 65 years. An adjustment for age > 65 years has been proposed, leaving the cutoff point at 229.
HEPAMET FIBROSIS SCORE
Derived from a group of 242 patients from four countries, all with MASLD demonstrated by biopsy, it involves the variables sex, age, presence of diabetes, glycemia, serum insulin, HOMA-IR, AST, albumin, and platelet count. Cutoff points validated in several cohorts allow determining a low risk of fibrosis with scores < 0.12, intermediate with 0.12-0.47, and high with > 0.47. S 97.2%, E 74%, NPV 92%, PPV 76.3%, positive likelihood ratio 13.2, and negative 0.31 have been reported30.
ELF
It is composed of the determination of procollagen type III peptide, hyaluronic acid, and tissue inhibitor of metalloproteinase-1. Scores < 7.7 are interpreted as very low risk of fibrosis, while values ≥ 9.8 identify patients with advanced fibrosis and risk of clinical events related to liver disease and cirrhosis29.
DA-GAG
In a study involving 791 patients (Caucasian and Asian) with MASLD demonstrated by biopsy, logistic regression analyses of categorical variables were performed to identify patients with fibrosis grade ≥ 2. From this analysis, the DA-GAG score (diabetes, age, gamma-glutamyl transferase [GGT], AST, platelets, globulin, total proteins) was obtained, calculating an area under the curve in a validation group of 0.8031. It is important to note that scores are not diagnostic of fibrosis, but a screening tool used in open populations, whose objective is to make decisions within a diagnostic tree for the implementation of more advanced tests. Its objective is to be sufficiently sensitive to optimize diagnosis by avoiding unnecessary tests.
ELASTOGRAPHY
Elastographic methods are divided into:
- − Ultrasound-based:
- Qualitative.
- Quantitative: vibration-controlled transient or shear wave.
- − Magnetic resonance-based.
A systematic review and meta-analysis evaluated the capacity of different elastographic techniques to predict fibrosis, with the results shown in table 432.
Table 4. Summary of the performance of elastographic techniques
| Technique | Fibrosis stage | |||
|---|---|---|---|---|
| F1 | F2 | F3 | F4 | |
| VCTE | ||||
| S | 78% | 80% | 80% | 76% |
| E | 72% | 73% | 77% | 88% |
| AUC | 0.82 | 0.83 | 0.85 | 0.89 |
| MRE | ||||
| S | 71% | 78% | 83% | 81% |
| E | 85% | 89% | 89% | 90% |
| AUC | 0.87 | 0.91 | 0.92 | 0.90 |
| pSWE | ||||
| S | 64% | 69% | 80% | 76% |
| E | 76% | 86% | 86% | 88% |
| AUC | 0.77 | 0.86 | 0.89 | 0.90 |
| 2DSWE | ||||
| S | NA | 71% | 72% | 78% |
| E | NA | 67% | 72% | 84% |
| AUC | NA | 0.75 | 0.72 | 0.88 |
AUC: area under the curve; 2DSWE: 2D shear wave elastography; E: specificity; MRE: magnetic resonance elastography; NA: not applicable; pSWE: point shear wave elastography; S: sensitivity; VCTE: vibration-controlled transient elastography.
Modified from Selvaraj et al.32
The most accessible, explored, and validated technique is vibration-controlled transient elastography or FibroScan®. This technique has adequate performance for diagnosis and staging of fibrosis, in addition to being useful in follow-up to detect disease progression, defined as an increase of > 20% in liver stiffness, which translates into its capacity to predict future cirrhosis and mortality10.
There are also scores that combine transient elastography with biochemical markers, such as FAST (Fibroscan-AST), Agile 3+, and Agile 4, which have shown adequate performance in predicting significant fibrosis33–35.
Toward precision diagnosis
Precision medicine uses individual information encompassing genes, proteins, environment, and lifestyle, with the objective of preventing, diagnosing, or treating disease. Diagnostic precision would allow clinicians to establish individual risk of suffering from a disease, detect and catalog the condition, predict prognoses, select therapies with specific targets, and monitor the disease longitudinally36.
To date, it has been observed that diagnostic and staging methods in MASLD may have different results among subgroups, for example, according to age, sex, BMI, presence of metabolic comorbidity, and liver enzyme levels37. Therefore, the establishment of different cutoff points could be an initial step toward a more precise diagnosis.
Conclusions
MASLD represents a growing challenge in public health due to its high prevalence, its association with cardiometabolic comorbidity, and its risk of progression to advanced liver disease. A systematized initial clinical evaluation is essential for its timely detection, allowing stratification of the risk of significant fibrosis and guiding diagnostic and therapeutic decisions.
The initial approach should include a targeted clinical history, focused physical examination, specific laboratory tests, and imaging studies that allow identifying hepatic steatosis and estimating the severity of fibrosis. Non-invasive tools, such as biochemical scores (FIB-4, NAFLD-FS, Hepamet) and elastography, have proven useful for discriminating patients who require advanced evaluation, reserving liver biopsy for selected cases.
The choice of diagnostic method should be adapted to the clinical context, available resources, and patient characteristics, considering that factors such as age, BMI, and comorbidity can modify the diagnostic performance of the tools used. The approach toward precision medicine in MASLD – which considers interindividual variability and adjustment of diagnostic thresholds – constitutes a necessary horizon for improving detection, prognosis, and follow-up of this disease.
In summary, a thorough, integrated, and contextualized initial evaluation is the cornerstone for adequate management of patients with suspected MASLD, favoring early interventions that can modify the course of the disease.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical considerations
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data and does not require ethical approval. The SAGER guidelines do not apply.
Statement on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.
