Introduction
First-line treatment for metabolic dysfunction-associated steatotic liver disease (MASLD) consists of intensive lifestyle changes, with the goal of achieving weight loss greater than 7-10% to obtain histological improvement1. However, it has been shown that less than 10% of patients achieve these goals in a sustained manner with conventional dietary and medical therapy, which underscores the need for additional therapeutic strategies2.
In recent decades, metabolic and bariatric surgery has become a promising treatment for MASLD in people with obesity3. The latest clinical practice guidelines from the European associations for the study of the liver, diabetes, and obesity (European Association for the Study of the Liver, European Association for the Study of Diabetes, and European Association for the Study of Obesity) on MASLD recommend that surgery be considered a therapeutic option for patients without cirrhosis or with compensated cirrhosis (metabolic dysfunction-associated steatohepatitis [MASH])4.
In this context, bariatric surgery has proven to be the most effective intervention for significant and sustained weight loss, in addition to inducing improvement or resolution of steatosis, inflammation, and hepatic fibrosis5,6. Nevertheless, its indication may be limited in patients with a lower degree of obesity, high surgical risk, or medical contraindication, which has motivated the development of minimally invasive endoscopic techniques, such as endoscopic sleeve gastroplasty, intragastric balloons, and duodenal exclusion devices (Fig. 1), which seek to replicate the effects of surgical interventions with lower risk and greater accessibility7.
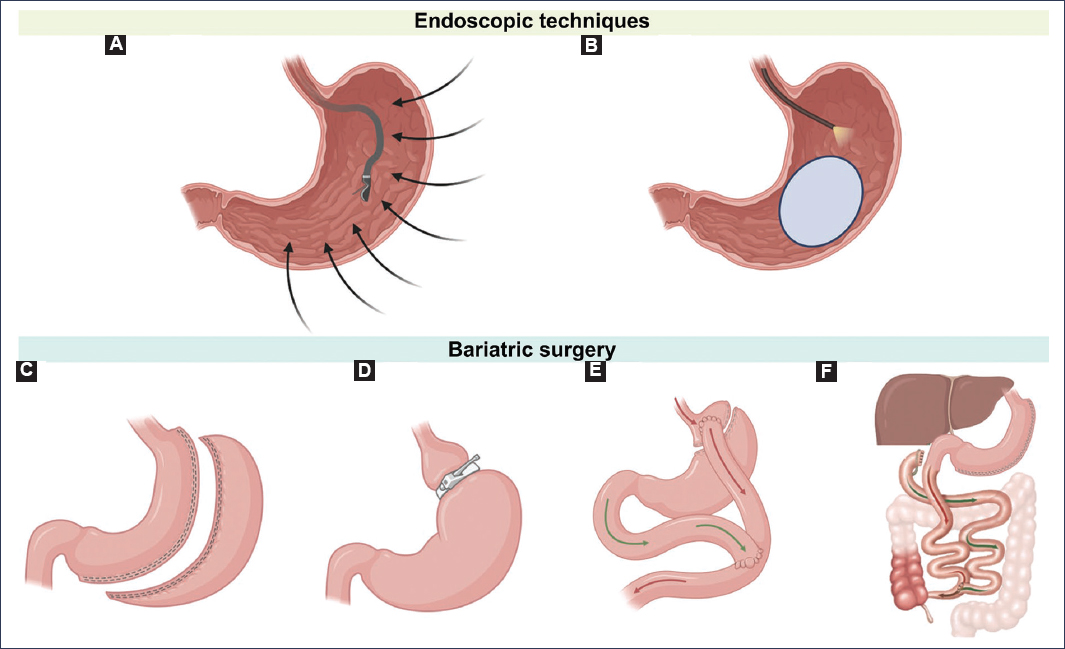
Figure 1. Endoscopic techniques and bariatric surgery in MASLD. A: endoscopic sleeve gastroplasty. An endoscope with a suturing device is used to reduce the size and volume of the stomach through permanent sutures along the greater curvature, from the antrum to the fundus. B: intragastric balloon. A collapsed balloon is inserted into the stomach through an endoscopic device and once in the stomach, it is filled with saline solution or air, occupying space and promoting the sensation of satiety. C: sleeve gastrectomy. Between 75% and 85% of the stomach is removed along the greater curvature, from the antrum to the fundus. D: adjustable gastric band. Limits food intake by placing a constrictor ring that completely surrounds the gastric fundus. E: Roux-en-Y gastric bypass. Combines reduction of stomach size with bypass of the distal stomach and proximal small intestine (50-100 cm), resulting in a Y-shaped intestinal connection. F: biliopancreatic diversion with duodenal switch. Reduces stomach size and bypasses the small intestine (150-200 cm), resulting in a gastrojejunostomy without the Y-shaped enterostomy configuration.
Despite their increasing implementation, important questions persist about their safety, long-term efficacy, real impact on the natural evolution of MASLD, and optimal patient selection. Likewise, a comprehensive preoperative evaluation is essential that considers the presence of advanced fibrosis or cirrhosis, which substantially modifies the risk of perioperative complications and expected metabolic outcomes8.
This chapter aims to critically review the indications for interventional treatment in MASLD, describe endoscopic techniques under investigation or limited use, analyze the role of bariatric surgery in these patients, and discuss key aspects of preoperative evaluation and postoperative complications, according to the most recent scientific evidence, to guide clinical practice and decision-making in a rapidly evolving field.
Therapeutic approach and indications for interventional treatment
The therapeutic approach to MASLD is based on a stepped model, as described below.
Lifestyle modifications
Lifestyle modifications are the cornerstone of the management of MASLD. Current recommendations include:
- – Weight loss: This is the most important factor and is proportionally associated with histological improvement. Weight loss of at least 5% of body weight is recommended to improve hepatic steatosis, > 7% to improve inflammatory activity, and preferably ≥ 10% to induce regression of fibrosis9,10.
- – Diet: Emphasis is placed on caloric restriction and reduction of saturated fats, simple sugars, and especially fructose, since its consumption is associated with fibrosis progression. A Mediterranean-type dietary pattern is recommended, rich in fruits, vegetables, whole grains, and unsaturated fats, due to its benefits on cardiometabolic risk factors and mortality. Consumption of ultra-processed foods should be avoided and fiber intake prioritized9–11.
- – Exercise: Regular physical activity, both aerobic and resistance, reduces hepatic fat and improves insulin sensitivity, independently of weight loss. It is recommended to perform at least 150 minutes per week of moderate-intensity exercise, adapting the prescription to the patient’s abilities and preferences to improve adherence9–12.
- – Alcohol abstinence: Alcohol consumption can accelerate the progression of liver disease and increase the risk of hepatocellular carcinoma. Total abstinence is recommended in patients with significant fibrosis (≥ F2), and alcohol consumption should be avoided in all patients with MASLD9–11.
Pharmacotherapy for overweight and obesity (body mass index [BMI] ≥ 25 kg/m2)
Pharmacotherapy is rapidly evolving, with recent advances and an increasingly personalized approach according to metabolic profile and disease stage. It should be considered in patients with obesity, type 2 diabetes, and MASLD, especially when there is a risk of progression to MASH and fibrosis10,13,15
The introduction of new drugs, particularly glucagon-like peptide-1 (GLP-1) receptor agonists, has improved outcomes in some patients, allowing a greater proportion to achieve weight loss > 10%. However, their use is limited by factors such as cost, insurance coverage, tolerability, and uncertainty about long-term safety and efficacy16. Furthermore, most patients with obesity do not access them or do not maintain these treatments for a prolonged period.
Pharmacological options with evidence in clinical trials to reverse steatohepatitis and improve fibrosis in patients without cirrhosis are listed in table 1.
Table 1. Drugs with possible benefits in MASLD
| Drug | Main evidence | Comments |
|---|---|---|
| Pioglitazone | Improves glucidic and lipid metabolism, and has shown resolution of MASH and improvement of fibrosis in patients with and without diabetes, according to meta-analyses and phase 2 trials. The combination of pioglitazone with GLP-1RA may have an additive effect on steatosis reduction and glycemic control | Monitor weight gain, edema, and bone health. The combination pioglitazone + GLP1-RA may be considered for refractory MASH/fibrosis |
| GLP-1RA (liraglutide, semaglutide, tirzepatide) | Have demonstrated significant reduction of hepatic steatosis, improvement of inflammation, and possible reversal of fibrosis in patients with and without diabetes, in addition to cardiovascular and renal benefits | Semaglutide and tirzepatide have shown positive results in phase 3 trials with histological outcomes in MASH |
| Resmetirom (thyroid hormone receptor beta agonist) | Recently approved by the FDA for the treatment of MASH without cirrhosis, with evidence of reduction of hepatic fat and improvement of inflammation and fibrosis in phase 3 trials | Its use outside the United States depends on local regulatory approval |
| Others | PPAR agonists, FXR agonists, ACC and DGAT inhibitors, and FGF21 analogs, with promising results in the reduction of steatosis and improvement of metabolic markers, although not yet approved | Not recommended: vitamin E, pentoxifylline, silymarin due to insufficient evidence or doubtful safety |
ACC: acetyl-CoA carboxylase; DGAT: diacylglycerol acyltransferase; FDA: Food and Drug Administration; FGF21: fibroblast growth factor 21; FXR: farnesoid X nuclear receptor; GLP-1: glucagon-like peptide-1; GLP-1RA: GLP-1 receptor agonists; MASH: metabolic dysfunction-associated steatohepatitis; MASLD: metabolic dysfunction-associated steatotic liver disease; PPAR: peroxisome proliferator-activated receptors.
In patients with type 2 diabetes and MASLD, it is recommended to prioritize drugs with evidence of hepatic and cardiorenal benefit, such as GLP-1 receptor agonists (GLP-1RA) and pioglitazone, over other oral antidiabetic agents that have not demonstrated histological impact on MASLD10,15. Vitamin E may be considered in adults without diabetes with MASH, although its use should be individualized due to the safety profile8. For advanced stages (cirrhosis), there are no effective pharmacological treatments that reverse the disease, and management focuses on prevention of complications and treatment of comorbidity10,15.
Finally, the development of advanced biological therapies (angiotensin receptor and neprilysin inhibitors, cell therapies, monoclonal antibodies) and targeted delivery strategies represents an active line of research, but these are not yet part of clinical practice17.
Current evidence supports that sustained weight loss through conventional medical interventions, such as lifestyle modification and pharmacotherapy, is limited in patients with obesity. Most patients only achieve modest weight loss (around 5% of initial body weight) and, in general, less than 10% of patients maintain significant weight loss (≥ 10%) in the long term2,18,20 Barriers include difficulty maintaining behavioral changes, cost, and lack of sustained adherence.
When the aforementioned measures prove ineffective or the disease shows progression, therapeutic intensification through interventional strategies is evaluated.
Interventional management
Indications for bariatric or metabolic surgery in patients with MASLD follow standard bariatric surgery criteria, but with specific considerations for liver disease8,11.
In patients with compensated cirrhosis secondary to MASLD, interventional management may be considered in centers with experience and multidisciplinary teams, since the risk of hepatic decompensation is similar to that of patients with decompensated cirrhosis. In contrast, in decompensated cirrhosis (presence of variceal hemorrhage, ascites, hepatic encephalopathy, or jaundice), bariatric surgery is contraindicated due to the high risk of complications and lack of safety data8,10,11.
According to the most recent medical literature, bariatric surgery is indicated in adults with:
- – BMI ≥ 40 kg/m2, regardless of comorbidities, given their high risk of progression to MASH, advanced fibrosis, and cirrhosis.
- – BMI ≥ 35 kg/m2 with associated comorbidity. MASLD and MASH are currently recognized as relevant comorbidities along with type 2 diabetes, uncontrolled hypertension, dyslipidemia, osteoarthritis, and urinary incontinence, especially if there is evidence of significant hepatic fibrosis (F ≥ 2).
- – BMI ≥ 35 kg/m2 when lifestyle changes and pharmacological therapy have failed.
- – Patients with severe MASLD and low surgical risk, in whom bariatric surgery not only improves weight but also induces histological regression.
Selection of the modality depends on factors such as BMI, presence of advanced fibrosis, comorbidity, anesthetic risk, and patient preference, always with a multidisciplinary evaluation (Fig. 2).
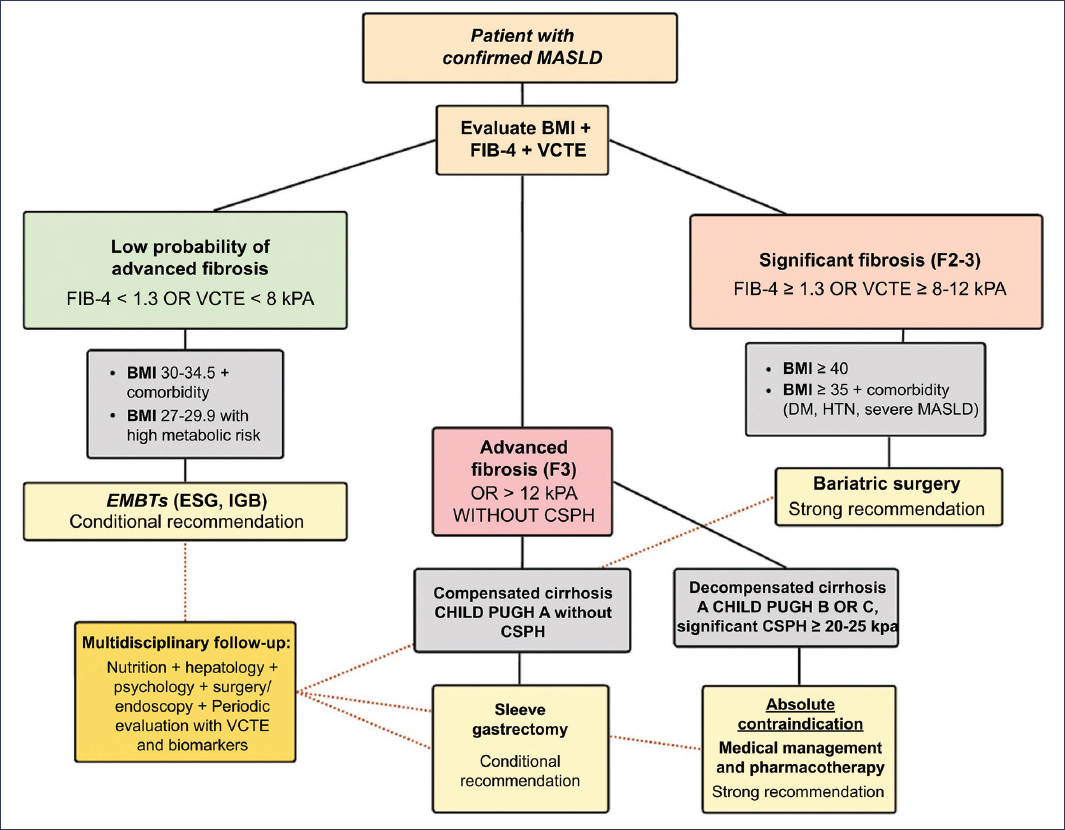
Figure 2. Selection algorithm: endoscopy versus surgery. CSPH: clinically significant portal hypertension; DM: diabetes mellitus; EMBTs: endoscopic bariatric and metabolic therapies; ESG: endoscopic sleeve gastroplasty; FIB-4: Fibrosis-4 Score; HTN: arterial hypertension; IGB: intragastric balloon; BMI: body mass index; MASLD: metabolic dysfunction-associated steatotic liver disease; VCTE: vibration-controlled transient elastography.
Endoscopic techniques under investigation or limited use
Endoscopic bariatric and metabolic therapies have been incorporated into the joint 2024 guidelines of the American Society for Gastrointestinal Endoscopy (ASGE) and the European Society of Gastrointestinal Endoscopy (ESGE), using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system to support their recommendations18, where they are classified as strong (“we recommend”) or conditional (“we suggest”), according to the certainty of evidence and the balance between benefits and risks (Table 2).
Table 2. Recommendations of the American Society for Gastrointestinal Endoscopy (ASGE) and the European Society of Gastrointestinal Endoscopy (ESGE) on the use of endoscopic bariatric and metabolic therapies18
| Recommendations | GRADE system |
|---|---|
| In adults with overweight or obesity, suggest the use of endoscopic bariatric and metabolic therapies + lifestyle modifications in patients with BMI ≥ 30 kg/m2 or BMI 27-29 kg/m2 with at least one obesity-associated disease. | Conditional recommendation, very low level of evidence |
| In adults with obesity, suggest the use of an intragastric balloon + lifestyle modifications, versus lifestyle modifications alone | Conditional recommendation, moderate level of evidence |
| In adults with obesity, suggest treatment with endoscopic gastric remodeling + lifestyle modifications, versus lifestyle modifications alone | Conditional recommendation, moderate level of evidence |
| In adults with obesity, suggest treatment with aspiration therapy + lifestyle modifications, versus lifestyle modifications alone, depending on the available device | Conditional recommendation, low level of evidence |
Recommended endoscopic bariatric and metabolic therapies include endoscopic sleeve gastroplasty and the intragastric balloon, both approved by the Food and Drug Administration (FDA) of the United States of America and CE marked in Europe. These techniques have demonstrated improvements in metabolic comorbidity, including reduction of glycosylated hemoglobin (HbA1c), blood pressure, and hepatic function parameters. Endoscopic sleeve gastroplasty, in particular, is associated with a significant reduction in alanine aminotransferase (Δ 7.4 U/l at 5 years), an improvement in hepatic steatosis, and a reduction in hepatic fibrosis (standardized mean difference: 0.7; 95% confidence interval: 0.1-1.3)21,22. Meta-analysis shows improvement in NAFLD activity score and insulin resistance after endoscopic bariatric and metabolic therapies22.
They are fewer invasive alternatives, with short- and medium-term hepatic benefits, but with less sustained efficacy than conventional bariatric surgery. They may be considered in patients with high surgical risk or contraindication for major surgery, and should be performed in centers with multidisciplinary experience, after careful evaluation of the risk-benefit ratio. In the presence of decompensated cirrhosis or significant portal hypertension, endoscopic techniques are contraindicated due to the high risk of complications10,11,23.
Regarding endoscopic metabolic procedures (such as the intragastric balloon and endoscopic gastroplasty), although they are less invasive and have shown benefits in weight reduction and MASLD improvement, the long-term evidence on their safety and efficacy is insufficient to make firm recommendations in the context of MASLD; their use should be considered on an individualized basis until more robust data exist10,24.
Guidelines suggest the use of endoscopic bariatric and metabolic therapies in adults with obesity, specifically in patients with BMI ≥ 30 kg/m2, or with BMI 27-29.9 kg/m2 and at least one obesity-associated disease. They are always recommended in combination with lifestyle modifications18.
The field of endoscopic bariatrics has advanced considerably in the last decade. The most relevant techniques are presented below.
ENDOSCOPIC SLEEVE GASTROPLASTY
It uses endoscopic suturing systems to reduce gastric capacity without anatomical resection. This technique has shown promising results in weight reduction and improvement of steatosis and hepatic fibrosis in the short and medium term. It may be a viable option in patients with MASLD who are not candidates for conventional bariatric surgery, although experience in patients with cirrhosis is very limited and should be considered with extreme caution7,11,23. Clinical trials and prospective studies show a total body weight loss of approximately 13-14% at 12 months, with maintenance of 11-12% at 5 years21,25. Compared to the intragastric balloon, endoscopic sleeve gastroplasty achieves greater sustained weight loss and a better metabolic profile26.
Among the endoscopic bariatric and metabolic therapies evaluated, it carries the lowest risk of adverse events, with a moderate event rate < 1% and no reported mortality in cohorts up to 5 years. Events include transient abdominal pain and mild nausea21,26.
INTRAGASTRIC BALLOONS
These consist of silicone balloons filled with saline solution that are placed endoscopically and removed after 6 months. They have been shown to induce significant weight loss in the short term and improvements in steatosis and hepatic fibrosis markers in patients with MASLD. However, evidence suggests that the magnitudes of weight loss and hepatic improvement are lower compared to conventional bariatric surgery, and there is a tendency for weight regain after device removal. Furthermore, the presence of hepatic insufficiency or cirrhosis, especially with clinically significant portal hypertension or esophagogastric varices, constitutes a contraindication for the use of the intragastric balloon due to the risk of serious complications, such as gastric perforation. In the United States of America, devices approved by the FDA (Orbera, ReShape, Obalon) have specific contraindications related to advanced liver disease and the presence of varices20,23.
The intragastric balloon produces a total body weight loss of 10.2% at 6 months, with partial recovery after device removal (7.6% at 12 months)25,27. The effect is less sustained than that of endoscopic sleeve gastroplasty but exceeds lifestyle intervention alone. The most frequent adverse effects are nausea or vomiting (20%) and abdominal pain (7%). Serious events, such as migration or perforation, have a low incidence25,27.
ASPIRATION THERAPY
The AspireAssist® system allows postprandial gastric content to be drained through a percutaneous tube. It is currently in disuse due to concerns about quality of life and complications18.
NEW TECHNIQUES
Duodenal mucosa ablation (resurfacing) and endoscopic duodenojejunal bypass have shown encouraging preliminary results in improving metabolic and hepatic parameters, but still require further validation in controlled and long-term studies7.
Duodenal resurfacing consists of controlled thermal ablation of the duodenal mucosa using a special catheter that administers hot water after submucosal elevation. The objective is to induce remodeling of duodenal enteroendocrine cells, which may restore altered hormonal signaling in type 2 diabetes and improve glycemic control, with a modest impact on weight loss. Controlled studies have demonstrated that it can reduce HbA1c in patients with type 2 diabetes, with an acceptable safety profile and a low rate of serious adverse events18,28,31 However, this technique has not yet received FDA approval, and its use is limited to research protocols or specialized centers32.
Endoscopic duodenojejunal bypass is performed primarily through two techniques: the duodenojejunal bypass liner (EndoBarrier) and magnetic compression anastomosis systems without incision. This procedure is not approved by the FDA for the treatment of obesity or type 2 diabetes, according to the most recent guidelines and reviews from American and European societies. Its use remains experimental, and its regulatory status is for clinical research, without authorization for commercial use18,30.
EndoBarrier is an impermeable polymeric sleeve that is anchored in the duodenal bulb and extends to the proximal jejunum, creating a physical barrier that diverts food from contact with the duodenal and proximal jejunal mucosa, delaying mixing with pancreatobiliary secretions30,33,36 This simulates the mechanism of a surgical bypass, promoting hormonal changes beneficial for glycemic control and weight loss. Clinical trials have shown an additional excess weight loss of approximately 9-10% compared to controls at 12 months, along with significant improvement in HbA1c18,30,37.
Regarding its safety, although the procedure is minimally invasive, up to 74% of patients may experience some adverse event35, so its use should be restricted to research protocols and under strict clinical surveillance.
In a systematic review of 1056 patients, serious adverse events accounted for 3.7%, with hepatic abscess being one of the most significant. The main mechanism appears to be related to the device anchoring system38. In the U.S. registry study (ENDO trial), the incidence of hepatic abscess was 2.4% (5 of 212 patients)18,30. In various meta-analyses and systematic reviews, the rate of hepatic abscess varies between 0.13% and 3.5%, depending on the population and study design25,38. Other adverse effects have been reported, such as intolerance (up to 10.9%), gastrointestinal bleeding (up to 4-6%), device obstruction (3-4%), pancreatitis (1-2%), intestinal perforation, and ulceration18,34,35.
Bariatric surgery in MASLD
Bariatric surgery plays an important role in MASLD, especially in patients with obesity and metabolic comorbidities such as type 2 diabetes, hypertension, and obstructive sleep apnea10,16,39. Current evidence demonstrates that bariatric surgery induces significant and sustained weight loss, with average reductions of 25-30% of body weight, which translates into substantial improvements in hepatic steatosis, inflammation, and hepatic fibrosis40, as well as significant improvement in glycemic control (HbA1c reduction around 24.4%), type 2 diabetes remission in approximately half of patients, and reduction of comorbidity such as hypertension and nephropathy11,41,42. In patients with compensated cirrhosis, mean weight loss at one year can reach 43 kg, with a BMI reduction of approximately 10.8 kg/m2 and an excess weight loss of 62.8%42.
Evidence demonstrates resolution of MASH in 80-84% of patients at 1 and 5 years after surgery, without worsening of fibrosis11,43. Fibrosis regression occurs in 70% of cases, and disappears completely in up to 56% of patients, although reversal is less frequent in cases of advanced fibrosis and may persist in about 47% of patients in the long term, especially if weight loss is lower or persistent comorbidity exists. Fibrosis reduction is progressive and is associated with greater weight loss and metabolic improvement23,44.
In terms of clinical events, bariatric surgery reduces the risk of major adverse hepatic outcomes (progression to cirrhosis, hepatocellular carcinoma, liver transplant, or liver-related death) at 10 years, with a cumulative incidence of 2.3% versus 9.6% in non-surgical controls (adjusted hazard ratio: 0.12; p = 0.01)45. In addition, it decreases the incidence of hepatocellular carcinoma (0.05% vs. 0.34% in controls) and reduces portal pressure in patients with cirrhosis and obesity23. It also decreases the risk of cardiovascular events3,8,10,11,46. In controlled and observational studies, resolution of MASH without worsening of fibrosis is achieved in 56-70% of patients undergoing Roux-en-Y gastric bypass or sleeve gastrectomy, compared to much lower rates with lifestyle intervention alone8,10.
The benefits of bariatric surgery on MASLD are not limited to caloric restriction, but include changes in hepatic signaling, intestinal microbiota, and immune modulation, which contribute to histological and metabolic improvement3,47. However, each year, less than 2% of eligible patients receive this surgery, due to factors such as access, cost, perception of invasiveness, and the need for social support and long-term follow-up18,39.
SLEEVE GASTRECTOMY
It consists of resection of approximately 80% of the stomach, leaving a narrow gastric tube. It is the most frequently performed procedure in patients with metabolic liver disease, including those with compensated cirrhosis and in the post-liver transplant setting, due to its safety profile, lower risk of malabsorption, and preservation of endoscopic access to the upper gastrointestinal tract and biliary tract. It has demonstrated significant improvements in steatosis, inflammation, and hepatic fibrosis, as well as in metabolic and hepatic function parameters.
ROUX-EN-Y GASTRIC BYPASS
This procedure creates a small gastric reservoir that connects to the small intestine, excluding the duodenum and part of the jejunum. It has shown substantial benefits in the resolution of steatosis and hepatic fibrosis, with robust metabolic effects. However, in patients with significant hepatic dysfunction or cirrhosis, it may be associated with greater risk of nutritional complications and difficulty with endoscopic access to the biliary tract, so it is usually reserved for selected cases10,48.
ADJUSTABLE GASTRIC BANDING
Although historically it was an option, its use has decreased considerably due to its lower long-term efficacy and complications such as erosion or band displacement. Hepatic benefits are more modest compared to sleeve gastrectomy and Roux-en-Y gastric bypass7,10,11.
BILIOPANCREATIC DIVERSION WITH DUODENAL SWITCH
It is a more aggressive technique, with profound metabolic effects, but with a greater risk of nutritional complications. Its use is limited and is reserved for very selected cases7,10,11.
Preoperative evaluation in patients with metabolic dysfunction-associated steatotic liver disease
Before considering surgery in patients with MASLD, it is essential to perform an exhaustive and multidisciplinary preoperative evaluation (Table 3). This process not only determines surgical viability but also allows optimization of the factors that most influence perioperative prognosis: hepatic reserve, presence of portal hypertension, cardiometabolic comorbidity, and nutritional status6.
Table 3. Preoperative evaluation
| Area | What is evaluated? | Reason for its importance |
|---|---|---|
| Classification and hepatic reserve | Child-Pugh, MELD-Na, albumin, INR, PTT, bilirubin | Defines if the patient is compensated (CP A) – candidate – or decompensated (CP B/C) – requires stabilization or contraindicates surgery – ; guides prognosis and anesthetic risk |
| Portal hypertension | Varices (endoscopy), portal Doppler, splenomegaly | The presence of clinically significant portal hypertension increases perioperative bleeding and dehiscence; if there are varices, ligation or beta-blockade before the procedure |
| Renal function and electrolytes | Creatinine, urea, sodium, potassium; rule out hepatorenal syndrome | Renal deterioration elevates MELD and complicates the choice of drugs, fluids, and radiological contrast |
| Nutritional status and sarcopenia | Body composition, albumin, vitamins A, D, B1 and B12, iron, zinc, proteins | Occult malnutrition and micronutrient deficiency worsen healing and encephalopathy |
| Metabolic and cardiovascular comorbidity | HbA1c, blood pressure, lipid profile, electrocardiogram, echocardiogram, stress test, OSA | Type 2 diabetes, hypertension, and cardiovascular disease multiply morbidity and mortality; must be optimized before intervention |
| Hemostasis and platelets | Platelets, fibrinogen, thromboelastographic rotation | Platelets < 50,000 and coagulopathy generate high bleeding risk |
| Abdominal imaging | Ultrasound, computed tomography or magnetic resonance imaging: portal thrombosis, HCC, biliary pathology | Detects anatomical contraindications and plans the type of procedure |
| Medication and toxins | Alcohol, NSAIDs, anticoagulants, herbal medicines | Remove hepatotoxins, adjust hypoglycemic agents and anticoagulation, optimize abstinence |
| Psychological assessment | Depression, anxiety, eating disorders, substance abuse, expectations, support network | Success after intervention depends on adherence and capacity for behavioral change; early detection allows intervention and follow-up |
| Anesthetic risk | ASA-PS, difficult airway, pulmonary function, OSA | Obesity + cirrhosis = ventilatory and hemodynamic risk. Defines the type of induction, invasive monitoring, and care in ICU |
| Center and surgical team | High-volume hospital with liver transplant and bariatric units, specialized ICU | Reduces mortality (< 1%), facilitates rescue in case of decompensation or need for bridge transplant |
| Multidisciplinary approach | Hepatology, bariatric surgery, anesthesia, nutrition, endocrinology, cardiology, psychology | Comprehensive consensus on surgical timing and peri- and postoperative strategy; longitudinal follow-up |
NSAIDs: nonsteroidal anti-inflammatory drugs; ASA-PS: American Society of Anesthesiologists Physical status classification system; HbA1c: glycosylated hemoglobin; HCC: hepatocellular carcinoma; INR: International Normalized Ratio; MELD-Na: model for end-stage liver disease-sodium score; OSA: obstructive sleep apnea; PTT: partial thromboplastin time; ICU: intensive care unit.
Only patients with compensated cirrhosis, and after optimization of all the above variables, should proceed to the surgical procedure, ideally in centers with combined experience in hepatology and bariatrics.
Complications and postoperative considerations
Complications
Complications and adverse effects after bariatric surgery in patients with MASLD can be classified as early (first 30-90 days) and late (months to years after the intervention) events, and their frequency and severity vary according to the type of procedure and patient comorbidity.
In the short term (first 30 days), the most frequent adverse effects include postoperative sepsis, gastrointestinal leaks, pulmonary events, hemorrhage, venous thromboembolism, acute renal failure, intestinal obstruction, and cardiac events. Surgical mortality in the first year is low but not negligible, and has been reported mainly after gastric bypass, associated with complications such as gastrointestinal leaks and respiratory failure6,48,50
In the long term, the most frequent complications are intestinal obstruction, persistent abdominal pain, marginal ulcer, anastomotic stenosis, cholelithiasis, incisional hernia, gastrogastric fistula, dumping syndrome, and gastroesophageal reflux, depending on the type of surgery49,51. Nutritional and vitamin deficiencies (iron, vitamin B12, vitamin D, calcium, proteins) are common and can lead to anemia, hypocalcemia, hypoalbuminemia, osteoporosis, and, in severe cases, protein malnutrition, especially after malabsorptive procedures such as Roux-en-Y gastric bypass48,51,52. Postprandial hypoglycemia and dumping syndrome may also occur, particularly in patients with type 2 diabetes48,52.
In the psychiatric realm, an increased risk of de novo depression and substance use (opioids, alcohol) has been observed, as well as cases of suicide in long-term follow-up, which underscores the importance of multidisciplinary evaluation and follow-up51,52. Chronic abdominal pain and the need for additional abdominal surgeries are more frequent in surgically treated patients compared to medical treatment51.
In summary, bariatric surgery in patients with MASLD carries perioperative risks and long-term complications that require close surveillance, lifelong nutritional supplementation, and multidisciplinary follow-up, with proper patient selection and performance of the procedure in high-experience centers being fundamental48,51,52.
Postoperative considerations
Postoperative evaluation of patients with MASLD undergoing bariatric surgery should be comprehensive, using non-invasive methods and, in selected cases, liver biopsy. Recent literature supports significant and rapid improvement in hepatic steatosis, inflammation, and hepatic stiffness after bariatric surgery, observable from the first 3 months and sustained in the medium and long term5,53,56
Recommended non-invasive tools include transient hepatic elastography by controlled vibration to assess stiffness or fibrosis, and the controlled attenuation parameter to quantify it5,54; biochemical indices, such as the Fatty Liver Index and the NAFLD score, also show utility for follow-up44. Additionally, some advanced serum tests, such as lipidomic panels (e.g., OWLiver®), may provide added value in monitoring and predicting disease remission53.
In medium-term follow-up (1-3 years), a significant reduction in the proportion of patients with steatohepatitis and an increase in those with a healthy liver is observed, although complete reversal of advanced fibrosis may be limited, persisting in a relevant percentage of patients despite resolution of inflammation and significant weight loss44. Advanced age and the presence of type 2 diabetes are associated with a lower probability of liver disease remission53,54. The magnitude of weight loss is a key predictor of histological improvement, both in the resolution of MASH and in the regression of fibrosis44,57.
Regarding the choice of procedure, both Roux-en-Y gastric bypass and vertical gastrectomy show comparable benefits in improving steatosis and fibrosis, but more aggressive procedures may be associated with greater nutritional risk7. Bariatric surgery also contributes to the resolution of metabolic comorbidity, independently of baseline hepatic status58.
Conclusion
MASLD is one of the main causes of hepatic and cardiovascular morbidity worldwide, intimately related to the obesity and metabolic syndrome epidemic. Bariatric surgery is an effective tool for modifying the natural evolution of MASLD in patients with obesity and metabolic comorbidity, improving both hepatic and cardiovascular outcomes, provided that candidates are properly selected and it is performed in centers with experience. It constitutes the most effective interventional treatment for achieving sustained weight loss, metabolic improvement, and regression of hepatic damage, while endoscopic techniques emerge as promising alternatives for patients with lower BMI or high surgical risk. However, its implementation requires meticulous preoperative evaluation, careful candidate selection, and long-term multidisciplinary follow-up. Future research should focus on controlled studies with prolonged follow-up to determine their impact on fibrosis progression, cardiovascular complications, and survival.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical considerations
Protection of people and animals. The authors declare that no experiments have been performed on human beings or animals for this research.
Confidentiality, informed consent and ethical approval. The study does not involve patient personal data or require ethical approval. The SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.
