Introduction
Gastroesophageal reflux (GER) is a highly prevalent problem. In the following pages, the role of obesity and bariatric procedures in the generation of GER will be analyzed. Evidence of the association between respiratory symptoms and reflux, and its impact on diagnostic approach and treatment, will also be reviewed. Finally, some relevant aspects of the relationship between GER and psychiatric disorders will be mentioned.
Gastroesophageal reflux disease and obesity
Gastroesophageal reflux disease (GERD) is one of the most frequent problems in consultation. Its etiological factors are multiple, but one of the most important is obesity. In Mexico, the prevalence of obesity is among the highest in the world; in a recent survey in adults, a prevalence of obesity of 36.9% and overweight of 38.3% was found1. Obesity was more frequent in women between the fourth and fifth decades of life1. Reflux symptoms are more common in obese patients, and a relationship has been found between body mass index and the presence of reflux symptoms2. Symptoms have been particularly related to patients with central obesity. Central adiposity has also been found as a risk factor for the development of Barrett’s esophagus and adenocarcinoma3. Additionally, lifestyle changes, particularly weight reduction, have a positive effect on reflux symptoms4.
Pathophysiology of GER in obesity
Overweight and obesity significantly increase the risk of GER and its complications; however, there is controversy about the pathophysiology of GER in obese patients, as the mechanisms involved are multiple.
The integrity of the esophagogastric junction (EGJ) and the intra-abdominal location of the lower esophageal sphincter (LES) are extrinsic factors that have a relevant role in anti-reflux mechanisms. The increase in intra-abdominal pressure with overweight increases the transdiaphragmatic gradient, and with it there is a greater possibility of hiatal hernia formation, which in turn is associated with low LES pressure (the intrinsic anti-reflux factor) (Fig. 1). In patients with hiatal hernia, the decrease in the vigor of crural diaphragm contraction on inspiration (extrinsic factor) was found to be the most important factor associated with reflux5. Additionally, obesity increases transient LES relaxations, which are the most important mechanism in reflux6. The role of intra-abdominal fat has also been studied, particularly that located in proximity to the EGJ. In an in vitro study with fat cells obtained during surgeries from obese patients and normal weight patients, it was found that those from obese patients stimulate the generation of inflammatory interleukins that produce deterioration in the integrity of the esophageal mucosa, which manifests as dilation of intercellular spaces7 (Fig. 1).
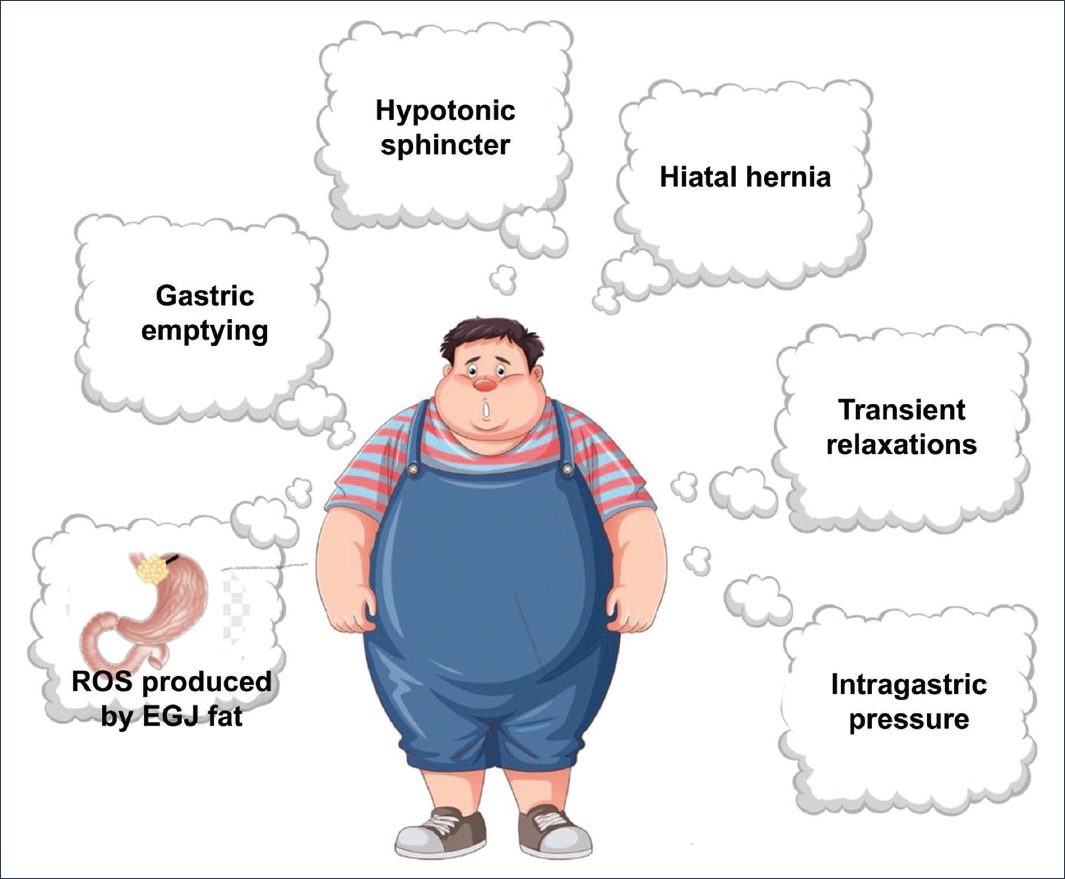
Figure 1. Pathophysiological mechanisms involved in the generation of reflux in the obese patient. ROS: reactive oxygen species; EGJ: esophagogastric junction.
Other humoral factors in obese patients have been associated with GER. Elevated leptin levels in patients with GER have been associated with greater severity of esophagitis, and on the other hand, an inverse correlation has been found between ghrelin levels and GER. The incidence of esophageal cancer has increased significantly in recent decades. Serum levels of adiponectin, a peptide produced by adipose tissue, have been found elevated in Barrett’s esophagus, which is a precursor lesion of esophageal cancer8. Adiponectin is secreted in three multimeric forms: low molecular weight, medium molecular weight, and high molecular weight. Low molecular weight adiponectin has anti-inflammatory effects, and it has been observed that its elevated levels decrease Barrett’s esophagus in patients with GER9.
Esophageal body motility plays a significant role in acid clearance. There is discrepancy in the results of esophageal manometric studies performed in obese patients. Esophageal dysmotility has been observed in more than half of obese patients; the most frequent finding has been hypomotility of the esophageal body. However, in a more recent series using high-resolution equipment and classified according to the Chicago 3.0 classification, the prevalence of dysmotility was higher in normal weight subjects compared to obese subjects. Furthermore, it was found that esophageal symptoms were more frequent in obese individuals, but these were related to reflux10. In a study of patients with morbid obesity evaluated before bariatric surgery, a high prevalence (34%) of motor alterations was found11. Recently, normative values for esophageal manometry in obese patients have been published, according to which it is suggested that some motor disorders, such as functional obstruction, may be overdiagnosed, since higher values of the integrated relaxation pressure and the distal contractile integral were found in obese patients12. These findings were also demonstrated in another study conducted in obese patients without symptoms and without endoscopic evidence of GER; the values found in high-resolution esophageal manometry did not differ significantly from the normal weight control group, except for higher intragastric pressure and, consequently, an elevated transdiaphragmatic gradient. Likewise, an average integrated relaxation pressure of 17 was documented, higher than the value of 15 in the Chicago 4.0 classification. In this same study, normative values were established for pH and impedance studies, and increased acid exposure time was documented, as well as an increase in the total and postprandial number of reflux episodes, with values that would correspond to borderline reflux according to the Lyon 2.0 consensus13. It can be concluded that esophageal peristalsis in obese patients is not significantly different from that in healthy subjects. On the other hand, high-resolution esophageal manometry study is important in the preoperative evaluation of obese patients for bariatric surgery, since the finding of a hypotonic sphincter demonstrates a predisposition to present GER, or a severe motor alteration of the esophageal body may modify the type of surgery to be performed14. Patients with GER more frequently have gastroparesis, which has been identified in up to 30% of subjects. In obese patients with GER, no significant difference in solid gastric emptying has been documented compared to normal weight subjects15.
New drugs in the management of obesity and their effect on GERD
Pharmacological treatment of obesity has had a notable evolution in recent years with the identification of glucagon-like peptide-1 (GLP-1) peptides, gastrointestinal hormones derived from the intestine and secreted after meal intake, which possess an insulinotropic effect (incretins). Studies on these peptides led to the development of GLP-1 receptor agonists (GLP-1 RAs), and in 2005 they were approved for the treatment of type 2 diabetes mellitus. With the use of these drugs, satiety and decreased caloric intake were also observed, which has motivated their use in non-diabetic obese patients. In 2014, liraglutide was the first GLP-1 RA approved by the United States Food and Drug Administration for the treatment of obesity, and subsequently, in 2022, semaglutide and tirzepatide were approved. These drugs have a high frequency of gastrointestinal adverse effects (nausea 42.23%, vomiting 21.90%, and diarrhea 21.93%)16 that are related to central and peripheral physiological effects. GLP-1 receptors have been identified in the myenteric plexus, and binding to these receptors inhibits vagal activity, delays gastric emptying, and reduces phase III of the antroduodenal migrating complex; this has been observed to be more pronounced with short-acting GLP-1 RAs17.
In a systematic review and meta-analysis, it was found that liraglutide administration decreases gastric emptying compared to placebo, and this same effect was not observed in studies with semaglutide and placebo. It has been reported that gastric emptying decreases more with the use of short-acting GLP-1 RAs18. Delayed gastric emptying is a recognized predisposing factor for GER, and secondary gastric distension increases transient LES relaxations, which is the most recognized pathophysiological mechanism. Despite the fact that these factors are frequent mechanisms of reflux, GERD symptoms were not among the most frequent adverse events of these drugs19. In a study comparing lixisenatide versus liraglutide (short- and long-acting GLP-1 RAs), no abnormalities were found in esophageal peristalsis, basal pressure, or LES relaxation. Gastric emptying measured by breath test was observed to be significantly delayed with the use of both drugs. In this same work, an ambulatory pH study was performed before and after treatment, and unexpectedly no significant pre- and post-treatment difference was demonstrated with both drugs19. Sandhu et al.20 analyzed whether receiving a GLP-1 RA was a predisposing factor for having abnormal reflux. They studied 457 patients who underwent ambulatory pH study, of which 59 were receiving a GLP-1 RA. These patients did not have more reflux symptoms or more abnormal studies. Therefore, the use of GLP-1 RAs does not appear to be a predictor of abnormal reflux. In a phase 3 study with tirzepatide, the most frequent adverse effects were gastrointestinal (nausea, diarrhea, and constipation), but reflux symptoms occurred in only 6.6% of 250 patients who received tirzepatide, compared to 2.4% of 292 who received placebo21. On the other hand, GERD symptoms were reported more frequently in a study conducted with three doses of liraglutide in diabetic patients, with a significant difference22. Orforglipron is a new non-peptide GLP-1 RA in development for the management of obesity and type 2 diabetes. In a randomized, double-blind phase 2 study in obese patients with four doses of orforglipron, the observed adverse effects were, as with other drugs in this group, predominantly gastrointestinal (nausea, vomiting, constipation, diarrhea, and also GER). As with other GLP-1 RAs, a direct correlation was observed between dose and severity of adverse effects23. On this same drug, a meta-analysis was subsequently performed that found similar results regarding side effects, particularly GER24. Given that some of the GLP-1 RAs increase the possibility of having GER or its exacerbation, and considering the relationship of reflux with esophageal cancer, a meta-analysis was conducted to evaluate the risk of esophageal cancer in patients who have received this type of drug26. The meta-analysis included six studies with a total of 13,391 patients. No increase in the incidence of esophageal neoplasms was found with the use of GLP-1 RAs25. According to publications on different GLP-1 RAs, the use of these drugs is safe, and the most common adverse effects are gastrointestinal: nausea, vomiting, diarrhea, constipation, and less frequently, GER. In patients with data suggestive of gastroparesis and reflux, it is recommended to increase the dosage with longer intervals and use lower doses initially.
Role of bariatric surgery in GERD control
Given the high prevalence of obesity and GER in this population, the frequency of this combination is increasingly common. The role of anti-reflux surgery in the treatment of GERD in obese patients has been controversial. In laparoscopic fundoplication, greater technical difficulty has been reported in performing fundoplication and high symptomatic recurrence in overweight and obese patients (9.5%), compared with normal weight patient group (3.04%), but the incidence of postoperative complications is similar in obese and non-obese patients27. In a study with longer-term follow-up, in which an ambulatory pH study was additionally performed, greater recurrence of symptoms and objective evidence of reflux by pH study were documented in obese patients who underwent laparoscopic fundoplication compared with non-obese patients26 (Table 1). It is speculated that the increase in intra-abdominal pressure in obese patients affects the fundoplication and leads to dismantling of the anti-reflux mechanism. Because of this, obesity is considered a relative contraindication for anti-reflux surgery. For this reason, bariatric surgery is the most accepted surgical option by surgeons.
Table 1. Recurrence of gastroesophageal reflux following laparoscopic fundoplication in long-term follow-up of 11 years in overweight and obese patients versus normal weight controls
| Recurrence | All (n = 201) | Normal weight (n = 43) | Overweight (n = 89) | Obesity (n = 69) |
|---|---|---|---|---|
| > 5.8% acid exposure | 27 | 1 | 7 | 19 |
| Erosive esophagitis | 55 (27.3%) | 5 (11.6%) | 19 (21.3%) | 31 (44.9%) |
| Reoperation | 7 | 0 | 1 | 6 |
Taken and modified from Schietroma et al.27.
The most commonly performed bariatric surgery is sleeve gastrectomy; however, this surgery has been associated with exacerbation of reflux in patients who already had it previously or with the appearance of de novo reflux in those who did not have it. In a meta-analysis of four studies conducted by Qumseya et al.28, the appearance of de novo reflux was observed in 40% of patients. Yeung et al., in another meta-analysis, found de novo reflux in 23% and exacerbation of previous reflux in 19%. Numerous mechanisms have been proposed to explain this situation. The decrease in gastric capacity and loss of the fundus favor an increase in intragastric pressure. The formation of a narrowing zone at the angular incisure, pyloric spasm, and potential damage to the vagus nerve produce delayed gastric emptying. Also, the anatomical changes produced in the EGJ, with loss of the angle of His and section of the muscle fibers that form the LES, weaken the anti-reflux mechanism of the EGJ (Fig. 2).
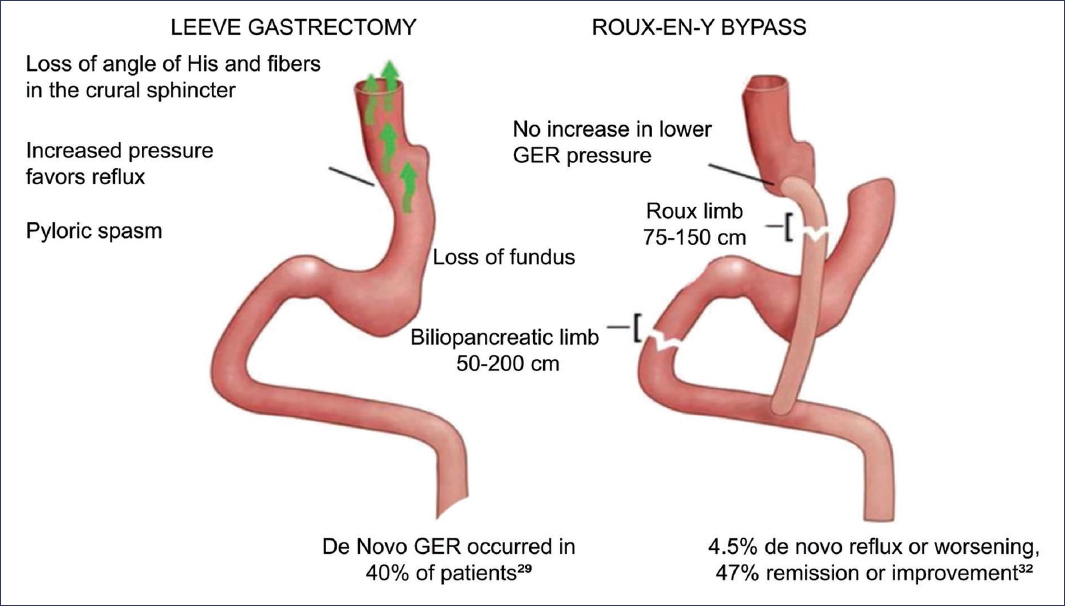
Figure 2. Gastroesophageal reflux (GER) postoperative after sleeve gastrectomy and Roux-en-Y bypass, pathophysiological mechanism and prevalence.
In the preoperative evaluation of obese patients who are candidates for sleeve gastrectomy, it is recommended to perform an upper endoscopic study. Patients with esophagitis on endoscopy have a higher probability of developing reflux and reflux complications, so the alternative of gastric bypass should be considered in these cases. Postoperative endoscopy is recommended to detect reflux complications, such as Barrett’s esophagus and consequently esophageal adenocarcinoma. In a systematic review and meta-analysis, Qumseya et al.28 observed the appearance of de novo Barrett’s esophagus in 11.6% of patients, with and without reflux symptoms, confirming the need for periodic follow-up. They also found that esophagitis developed with an increase of 13% each year28. There is no consensus regarding the need to perform high-resolution esophageal manometry and ambulatory pH studies prior to bariatric surgery, although evidence from several publications suggests that the findings of these diagnostic tests can help select the best method in each case and avoid late postoperative complications29.
Roux-en-Y gastric bypass is an effective surgery in the treatment of obesity. It is considered the procedure of choice in obese patients with concomitant GER and is the surgery recommended by most surgeons for this group of patients, with resolution of GER symptoms, although its de novo appearance is also reported. In one study, weight gain in the postoperative period was found as a predisposing factor for the persistence or appearance of reflux symptoms30. In a systematic study and meta-analysis on patients who underwent bypass, remission of reflux symptoms was demonstrated in 47%, with 39.5% showing no change. Several mechanisms have been proposed for the lack of improvement or de novo appearance of GER symptoms, such as anastomotic stenosis, a large new stomach, the presence of gastro-gastric fistula, and retained antrum syndrome with the generation of hypergastrinemia, and consequently stimulation of acid secretion31. According to the American Gastroenterological Association (AGA) guidelines, weight loss plays an important role in the treatment of GER in obese patients, together with other postural and dietary recommendations. Regarding surgical treatment, the recommendation of gastric bypass as the preferred surgery in obese patients is not a completely resolved issue, and patients should therefore be informed about permanent anatomical changes and the risks of bariatric surgery.
Gastroesophageal reflux disease and respiratory disorders
Asthma
Asthma is one of the most common respiratory problems in clinical practice, and GER is one of the conditions most frequently associated with asthma and can influence the course of the disease. Asthma, chronic laryngitis, and dental alterations are part of the extra-esophageal symptoms of reflux. The association of GER and asthma has been extensively studied. Havemann et al.32, in a systematic review of articles published from 1966 to 2006, found an average prevalence of 59% of GER symptoms in asthmatic patients, 51% of abnormal pH studies, and 37% of presence of erosive esophagitis (Table 2). On the other hand, an average prevalence of 3.9% of asthma was found in subjects with a diagnosis of GER. A correlation was also observed between the severity of asthma and the severity of esophagitis on endoscopy32 (Fig. 3). GER-induced asthma should be suspected in patients who initiate it in adulthood, in cases of difficult-to-control asthma, and when symptoms suggestive of reflux precede an asthma exacerbation, with ingestion of large meals, when lying down, or with alcohol intake.
Table 2. Prevalence of gastroesophageal reflux in patients with asthma with ambulatory pH study
| Reference | Country | Type of study | Population | Abnormal reflux |
|---|---|---|---|---|
| Sontag et al., 1990 | United States | Cross-sectional | Secondary care, asthma clinic | 81% |
| Susiki et al., 1997 | Japan | Cross-sectional | Secondary care | 72% |
| Vincent et al., 1997 | France | Cross-sectional | Secondary care | 31.9% |
| Kiljander et al., 1999 | Finland | Cross-sectional | Secondary care, asthma clinic | 53.3% |
| Carmona-Sánchez et al., 1999 | Mexico | Cross-sectional | Secondary care, asthma clinic | 75% |
| Compte et al., 2000 | Spain | Cross-sectional | Secondary care, asthma clinic | 14.8% |
| Al-Asoom et al., 2003 | Saudi Arabia | Cross-sectional | Secondary care, asthma clinic | 44% |
| Kiljander, 2004 | Finland | Cross-sectional | Secondary care | 35.6% |
| Legget et al., 2005 | United Kingdom | Cross-sectional | Secondary care | 55.8% |
Taken from Havemann et al.32.
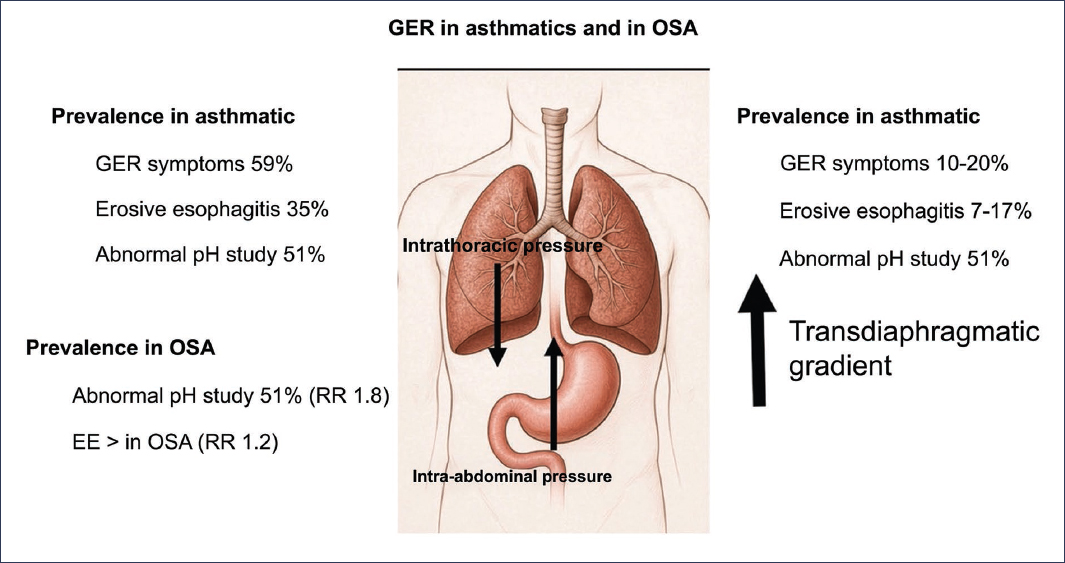
Figure 3. Prevalence of gastroesophageal reflux (GER) in asthmatic patients and those with obstructive sleep apnea (OSA), and predisposing factors for reflux33,42.
There are two proposed mechanisms to explain how asthma is generated in patients with GER: 1) direct reflux into the airway, which produces inflammation and bronchospasm, and 2) reflex mechanism, with the generation of bronchospasm by stimulation of esophageal receptors mediated by a vagal reflex32,33.
Asthma is a predisposing factor for reflux. The increase in negative intrathoracic pressure, and with it the increase in transdiaphragmatic gradient, favor the ascent of gastric contents toward the esophagus. Additionally, bronchodilator drugs relax smooth muscle and, therefore, the LES.
In asthmatic patients with typical GER symptoms, empirical treatment with double-dose proton pump inhibitor (PPI) for 8 to 12 weeks is recommended. However, the response of respiratory symptoms and pulmonary function tests has been conflicting and discrepant in the various published series, so according to the review by Broers et al.34, there is insufficient evidence to recommend empirical use of PPIs in the treatment of asthma, although they mention that it may be useful in patients with nocturnal symptoms. On the other hand, in a meta-analysis it was found that PPI use can induce the appearance of asthma in patients who use them, and exacerbation of asthma when they are discontinued; it is speculated that the mechanism is the generation of dysbiosis and acid hypersecretion when discontinuing them, respectively.
Patients without GER symptoms should be evaluated to document reflux35. Anti-reflux surgery for the management of extra-esophageal manifestations has a less favorable response than for typical symptoms. Recent clinical guidelines from the American College of Gastroenterology (ACG) recommend that anti-reflux surgery in asthmatic patients or with other extra-esophageal manifestations should only be performed when abnormal reflux has been objectively demonstrated (acid exposure time > 12% in 24 h) or a hiatal hernia > 4 cm36. Chronic cough is frequently attributed to GER and is part of the symptoms considered in the San Diego Consensus for laryngoesophageal reflux, along with dysphonia, throat clearing, excess mucus, and sore throat37. This consensus distinguishes between laryngopharyngeal symptoms and laryngopharyngeal reflux disease, which requires objective demonstration of abnormal reflux, either by endoscopy or through an abnormal ambulatory pH study, or with a positive response to a therapeutic trial in patients with laryngoesophageal symptoms (LeS) and typical symptoms. Establishing this difference is relevant, as the diagnostic and therapeutic approach is different.
At present, there is no diagnostic method that is conclusive to establish that GER is the cause of extra-esophageal symptoms. Laryngoscopy and endoscopy are of limited utility, and ambulatory pH studies identify the magnitude of GER but do not establish correlation with symptoms38. Significant improvement of extra-esophageal symptoms should not be taken as confirmation that reflux is the cause of these. The diagnosis of GER-associated cough requires clinical evaluation, endoscopic findings, response to treatment, and pH study results38. Management of GER-associated cough, as with typical manifestations, consists of lifestyle modifications and pharmacological treatment. Management with PPIs has not shown the same efficacy as for typical symptoms. In a meta-analysis of five PPI studies controlled with placebo for the management of chronic cough, no significant difference was found. Other therapies, such as alginates, neuromodulators, cognitive therapy, and hypnotherapy may have a role in the management of GER-associated cough. Regarding surgical treatment of cough and laryngeal symptoms, the response is variable according to a systematic review that included 27 articles. A good response to PPI use is associated with a satisfactory response to anti-reflux surgery. According to the American Gastroenterological Association (AGA) clinical guideline on surgical treatment of extra-esophageal manifestations of GER, chronic cough and laryngeal symptoms improve in highly selected populations, such as patients with concomitant typical symptoms, those who present a favorable response with PPIs, and those with significantly abnormal reflux (> 12% acid exposure in 24 h)39.
Obstructive sleep apnea (OSA)
It is a disorder characterized by repetitive sleep-induced laryngeal collapse, with cessation of airflow. Obstructive sleep apnea (OSA) is part of the frequent comorbidity associated with GER. The prevalence of OSA in the United States is approximately 15% in men and 5% in women. OSA is particularly associated with nocturnal reflux, according to a recent study that analyzed the frequency of nocturnal GER in two cohorts, one of 822 subjects diagnosed with moderate to severe OSA and another of 738 subjects from the general population40. The frequency of GER in patients with OSA ranges between 54% and 76%. Wang et al.41, in a retrospective study of a national database, found that patients with OSA not only had a higher frequency of GER (32% vs. 15% in patients without OSA) but also a higher risk of Barrett’s esophagus (64.6 per 10,000 in patients with OSA vs. 18.8 per 10,000 in those without OSA). Likewise, esophageal stricture was observed more in patients with OSA41. The association with overweight and obesity is frequent in patients with OSA, with an increase in intra-abdominal pressure. In OSA there is also an increase in negative intrathoracic pressure, which adds to the increase in intra-abdominal pressure and, therefore, increases the transdiaphragmatic gradient and reflux. This interrelationship of GER and OSA has been demonstrated in studies where treatment with continuous positive airway pressure particularly reduces nocturnal reflux42,43, as well as in studies where patients with GER treated with omeprazole show improvement in OSA symptoms.
The pathophysiological mechanism by which patients with OSA have more GER is unknown. It is proposed that OSA can generate gastric distension, delayed gastric emptying, transient LES relaxations, and elevated transdiaphragmatic gradient. OSA is related, epidemiologically and pathophysiologically, to asthma and rhinosinusitis, and together with GER they constitute what is called CORE syndrome. Therefore, these conditions should not be treated separately. Treatment with continuous positive airway pressure improves the evolution of these patients44.
GERD and psychiatric disorders
Psychological factors significantly impact symptom perception in chronic diseases. In esophageal diseases, a discrepancy has been observed between symptom severity and alterations found in pH and manometry studies. Anxiety has been associated with greater symptom severity. In a study published by Kessing et al.45, patients with higher anxiety scores reported greater intensity of symptoms (chest pain and heartburn), but not a greater number of symptoms. Anxiety levels were found to be higher in subjects with functional heartburn. In this same study, the effect of depression on symptoms was also analyzed, and it was not found to influence their perception or severity45. In a retrospective and cross-sectional study of a series of patients in Korea who underwent upper endoscopy, the presence of anxiety and depression was analyzed in three groups of patients with reflux: 1) patients with erosive esophagitis, 2) patients with non-erosive reflux, and 3) asymptomatic patients with erosive esophagitis. Anxiety and depression were observed more frequently in patients with reflux than in the control group, and particularly in patients with non-erosive reflux. Interestingly, asymptomatic patients with erosive esophagitis did not have significant psychological alterations46. In a more recent study, the correlation of GER with anxiety and depression was analyzed using logistic regression48. A significant correlation was demonstrated between the degree of anxiety and depression with ambulatory pH study values. Greater acidity, acid exposure time, and number of reflux episodes were observed in patients with higher anxiety levels47,48, suggesting that greater chronic acid exposure generates a higher degree of anxiety. On the other hand, Losa et al.48 demonstrated higher levels of anxiety and depression in subjects with hypersensitive esophagus, functional heartburn, and non-erosive reflux, and at the same time found greater perception of painful stimuli48.
In 2018, the Northwestern group developed a 15-item questionnaire, the Esophageal Hypervigilance-Anxiety Score (EHAS), to measure the degree of hypervigilance and anxiety in patients with esophageal diseases49. This tool has been translated and validated in several languages. Guadagnoli et al.50, in a retrospective study of patients who underwent wireless pH study, used EHAS to evaluate hypervigilance at different degrees of acid exposure and symptomatic correlation. They found no difference in hypervigilance state between patients with physiological reflux and those with abnormal exposure, nor did they observe a difference in those who had a significant symptom index compared to those who had a negative index. The study confirms that hypervigilance is a psychological process present throughout the entire spectrum of patients with reflux, and not only in those with hypersensitivity or functional heartburn50. More recently, this same group established and validated a shorter questionnaire of seven items (EHAS 7), to make its use easier in clinical practice51. The hypervigilance state is a psychological condition that impacts the perception and exacerbation of symptoms in esophageal diseases. Stimuli perceived by vagal afferent nerve endings are processed in the central nervous system. Hypervigilance and anxiety contribute to hypersensitivity and decrease quality of life.
Treatment of patients with GERD and anxiety should contemplate the coexistence of these two conditions. In patients with functional esophageal disorders, such as functional heartburn and functional chest pain, the use of neuromodulators, tricyclic antidepressants (TCAs), and selective serotonin reuptake inhibitors (SSRIs) is suggested. In a meta-analysis, the utility of TCAs and serotonin and norepinephrine reuptake inhibitors was demonstrated in relieving chest pain, and of TCAs and SSRIs in globus, but without utility in functional dysphagia. The authors suggest using neuromodulators selectively according to the symptom, starting with low doses and gradually escalating doses until achieving optimal therapeutic effect. Regarding treatment duration, it is suggested to be 8 to 12 weeks as an initial phase and with extension up to 6 to 12 months or more.
Conclusions
GERD is one of the most common reasons for consultation, and it is possible that in the future we will see it even more frequently due to the increase in predisposing factors, such as obesity, diabetes, OSA, the use of new drugs that cause gastroparesis, and surgical procedures that deteriorate the anti-reflux mechanism. We must learn to recognize those patients who will benefit most from the use of neuromodulators and not from escalating anti-reflux treatment.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical considerations
Protection of people and animals. The authors declare that no experiments were conducted on human beings or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data or require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.
