Introduction
Gastroesophageal reflux disease (GERD) is a chronic pathology characterized by the return of gastric contents toward the esophagus, which produces bothersome symptoms such as heartburn and regurgitation, affects quality of life, and may lead to complications1–3. Pharmacological treatment is the basis of its management, especially in moderate to severe forms of the disease3,4.
Therapeutic options
Proton pump inhibitors
Proton pump inhibitors (PPIs) constitute the cornerstone in the pharmacological treatment of GERD. They have proven to be the most effective drugs for gastric acid suppression and for the healing of erosive esophagitis.
CLASSIFICATION
PPIs available clinically are classified as:
- ‒ Delayed-release PPIs: they have an enteric coating that dissolves at pH 5.5 in the duodenum. This group includes omeprazole, esomeprazole, lansoprazole, pantoprazole, rabeprazole, dexrabeprazole, and ilaprazole.
- ‒ Dual delayed-release PPIs: they are presented in granules with two enteric coatings; one dissolves at pH 5.5 in the duodenum and the other at pH 6.75 in the terminal ileum. Dexlansoprazole belongs to this group.
- ‒ Immediate-release PPIs: combination of 20 mg of omeprazole and 1100 mg of bicarbonate. In this formulation, the PPI does not have an enteric coating, and the bicarbonate buffers the acid, protecting the omeprazole molecule4–6.
They all have similar mechanisms of action, although they differ slightly in their pharmacokinetics, hepatic metabolism, and relative potency.
MECHANISM OF ACTION
PPIs act by irreversibly inhibiting the H+/K+ ATPase enzyme (proton pump) located in gastric parietal cells, which is responsible for the secretion of hydrochloric acid in the stomach. By covalently binding to this enzyme, PPIs suppress basal and stimulated acid secretion.
PPIs are prodrugs that require two protonations in the acidic environment of the parietal cell to transform into the active drug. They bind only to proton pumps activated by food. Therefore, they should be administered 30 to 60 minutes before breakfast or dinner. Their absorption is rapid, but their plasma half-life is short; in delayed-release PPIs, it is an average of 2 hours. In the case of dexlansoprazole, the dual release allows prolonging the plasma half-life up to 4 hours. Ilaprazole has a half-life of 5 hours.
PPIs are metabolized primarily in the liver by the CYP450 enzyme system, especially CYP2C19 and CYP3A4.
There is significant interindividual variability due to genetic polymorphisms of CYP2C19. In slow metabolizers, they may have higher plasma concentrations and more effect, and in ultrarapid metabolizers, they may have lower efficacy.
Rabeprazole and ilaprazole are the PPIs that have the least metabolism by the 2C19 isoenzyme6.
The parameter to evaluate the clinical efficacy of an antisecretory agent in GERD is the ability to maintain intragastric pH above 4. Dexlansoprazole and esomeprazole maintain pH above 4 for up to 17 and 15 hours, respectively7.
Table 1 lists the main pharmacokinetic differences and standard doses of PPIs.
Table 1. Differences between proton pump inhibitors6
| Drug | Plasma half-life (h) | Duration pH > 4 (h/day) | Usual dose (mg/day) |
|---|---|---|---|
| Omeprazole | 0.5-1 | 10 | 20 |
| Esomeprazole | 1-1.5 | 13 | 20-40 |
| Pantoprazole | 1 | 10 | 40 |
| Lansoprazole | 1-1.5 | 12 | 30 |
| Rabeprazole | 1-1.5 | 12 | 20 |
| Dexrabeprazole | 1-2 | 13 | 10-20 |
| Ilaprazole | 5 | 17 | 10-20 |
| Dexlansoprazole | 1-2 | 17 | 30-60 |
INDICATIONS AND DOSES IN GERD2–5
- ‒ Erosive GERD: in grades A and B, a standard PPI dose is used for 8 weeks. In grades C and D, a double PPI dose is used for 8 weeks. Maintenance treatment can be with the “on-demand” regimen in mild varieties of esophagitis and with continuous treatment with standard dose in severe varieties.
- ‒ Non-erosive reflux disease: a standard PPI dose is recommended for 4 weeks and maintenance treatment with on-demand dosing.
- ‒ Barrett’s esophagus: continuous PPI dose sufficient to keep the patient symptom-free is recommended.
- ‒ Extraesophageal manifestations of GERD: double PPI dose is recommended for 3 months and continuous maintenance therapy with standard dose.
- ‒ GERD in pregnancy: PPIs can be used in pregnancy. They have a classification B according to the Food and Drug Administration, except for omeprazole, which belongs to group C3.
ADVERSE EFFECTS
PPIs are generally well tolerated, but their prolonged use has been associated with some adverse effects (Fig. 1), such as:
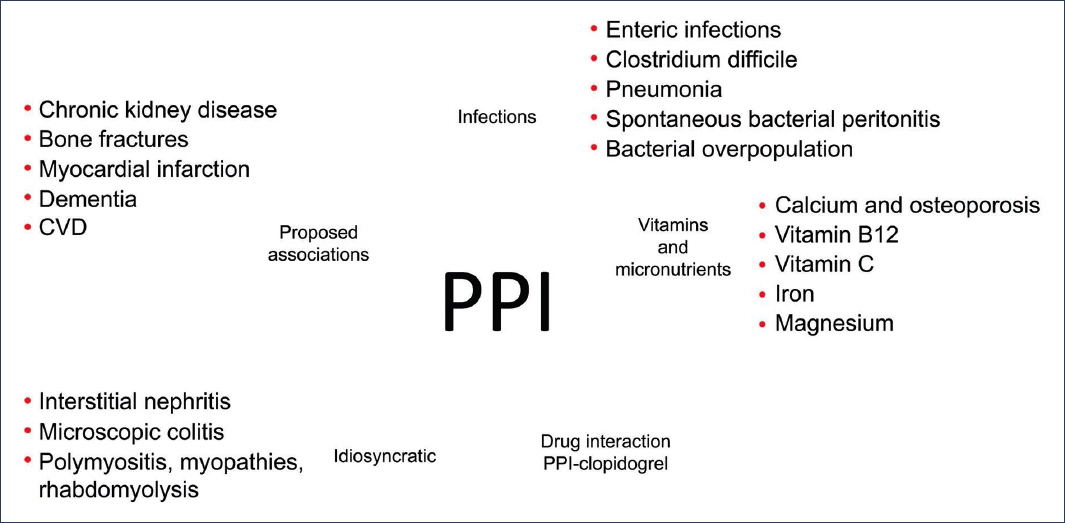
Figure 1. Adverse effects of proton pump inhibitors8.
- ‒ Enteric infections.
- ‒ Increased risk of community-acquired pneumonia.
- ‒ Malabsorption of vitamin B12, magnesium, and calcium.
- ‒ Possible increased risk of bone fractures.
- ‒ Hypergastrinemia with prolonged use.
- ‒ Acute interstitial nephritis (rare, but serious).
Among these adverse effects, enteric infections are the only ones that have a clinically significant odds ratio (> 3)8.
CRITERIA FOR DEPRESCRIBING PPIs
Although they are effective, many patients continue using PPIs unnecessarily. It is recommended to periodically evaluate the need to continue treatment. Some criteria for deprescribing are:
- ‒ Sustained remission of symptoms (> 6 months).
- ‒ Absence of esophagitis upon discontinuation of treatment.
- ‒ Use in unconfirmed GERD that has responded to lifestyle changes.
- ‒ Patients who use them only as “gastric protection,” without concomitant use of nonsteroidal anti-inflammatory drugs.
Withdrawal should be done gradually in patients with prolonged use to avoid a rebound effect of acid hypersecretion. In some cases, switching to an on-demand regimen or using H2 antagonists as bridge therapy may be possible9.
Potassium-competitive acid blockers
Potassium-competitive acid blockers (P-CABs) are a new class of antisecretory drugs used in the treatment of GERD. Their mechanism of action consists of reversibly and competitively inhibiting H+/K+ ATPase in gastric parietal cells, the same therapeutic target as PPIs, but through a different mechanism10–13.
Unlike PPIs, which need to be activated in an acidic environment, they are not prodrugs and exert their effect rapidly and sustainably, with greater consistency in gastric acid suppression10–14.
MECHANISM OF ACTION
P-CABs produce competitive inhibition of potassium at the proton pump H+/K+ ATPase. This prevents the exchange of potassium for hydrogen, which impedes the binding of hydrogen ions with chloride, preventing the formation of hydrochloric acid in the parietal cell10–14.
The irreversible binding of P-CABs with H+/K+ ATPase makes it possible for these drugs to inhibit both active and inactive proton pumps in the resting phase of the parietal cell, which allows sustained acid inhibition from the first dose. Additionally, P-CABs are resistant to gastric acid, so they do not have an enteric coating and do not require administration with food10–14.
PHARMACOKINETIC DIFFERENCES WITH PPIs
Table 2 presents the main differences between PPIs and P-CABs. Notably, P-CABs superconcentrate more than 100,000 times in the acidic space of the parietal cell, reversibly inhibit the proton pump, have a longer plasma half-life (up to 9 hours), and completely inhibit acid secretion from the first dose.
Table 2. Pharmacokinetic differences between P-CABs and PPIs11
| P-CAB | PPI |
|---|---|
| Act directly (after their protonation) on the H⁺/K⁺-ATPase pump | Require transformation to the active form, sulfenamide |
| Superconcentrate in the acidic space of the parietal cell (100,000 times more than in plasma) | Concentrate in the acidic space of the parietal cell (1000 times more than in plasma) |
| Competitively bind to the K⁺binding site of the H⁺/K⁺-ATPase pump | Covalent binding of sulfenamide with H⁺/K⁺-ATPase |
| Reversible binding with the proton pump Half-life 9.06 h | Irreversible binding with the proton pump Half-life 0.5-2.1 h |
| Duration of effect related to the drug’s plasma half-life | Duration of effect related to the half-life of the sulfenamide-proton pump complex |
| Complete effect from the first dose | Complete effect after repeated doses |
|
PPI: proton pump inhibitors; P-CAB: potassium-competitive acid blockers. |
|
Additionally, P-CABs are metabolized by isoenzymes different from CYP2C19, thereby avoiding the polymorphisms of rapid or slow metabolizers, and have fewer drug interactions10–14.
Table 3 presents the pharmacokinetic characteristics of different P-CABs14. In Mexico, tegoprazan and fexuprazan are available.
Table 3. Pharmacology of P-CABs and PPIs
| Pharmacokinetics and pharmacodynamics of different P-CABs | ||||||
|---|---|---|---|---|---|---|
| Drug (Maximum dose) | Revaprazan (200 mg daily) | Vonoprazan (40 mg daily) | Tegoprazan (200 mg daily) | Fexuprazan (160 mg daily) | Keverprazan (40 mg daily) | Zastaprazan (20 mg daily) |
| Structure | Pyrimidine | Sulfonyl pyrrole | Benzimidazole | Pyrrole | Sulfonyl pyrrole | Imidazopyridine |
| pKa | 6.68 | 9.06 | 5.2 | 8.40 | 9.12 | 6.02 |
| TMax, h | 2.1 ± 1.3 | 1.5 (0.75-3.0) | 1.8 (1.0-4.0) | 2.75 (1.5-5.0) | 1.40 (1.0-3.0) | 1.2 (0.75-2.0) |
| Half-life, h | 2.4 ± 0.2 | 6.1 ± 1.1 | 7.1 ± 2.2 | 7.5 ± 0.8 | 6.5 ± 1.1 | 10.28 ± 3.87 |
| 24-h pH, U – Day 1 | 2.2 | NA | 5.8 ± 0.4 | 5.8 ± 0.3 | NA | 4.95 ± 0.49 |
| 24-h pH, U – Day 7 | 2.5 | NA | 6.4 ± 0.3 | 6.5 ± 0.3 | NA | 5.47 ± 0.45 |
| 24-h pH > 4 Holding Time, % – Day 1 | 28.1 | 85.3 ± 8.3 | 76.8 ± 10.4 | 91.3 ± 4.1 | 85.8 ± 16.9 | 69.8 ± 15.9 |
| 24-h pH > 4 Duration % – Day 7 | 34.2 | 100.0 ± 0.0 | 94.6 ± 3.5 | 99.2 ± 1.9 | 100.0 ± 0.0 | 85.2 ± 11.4 |
| Nocturnal acid breakthrough | Yes | No | No | No | No | No |
|
PPI: proton pump inhibitors; P-CAB: potassium-competitive acid blockers. Modified from Scarpignato and Hunt11. |
||||||
CLINICAL EFFICACY
Clinical trials have demonstrated that P-CABs are as effective as PPIs in the healing of erosive esophagitis and are more effective in cases of severe esophagitis. This superiority has also been observed in maintenance therapy in severe esophagitis. It has also been observed that they achieve a higher percentage of symptom-free patients in the first weeks of treatment. In erosive GERD, a standard P-CAB dose is used for 8 weeks. For maintenance treatment, standard doses or half the dose are used continuously3–14.
P-CABs are superior to placebo in relieving symptoms in non-erosive reflux disease. They are used at a standard dose for 4 weeks. In maintenance treatment, the on-demand regimen can be used3–14.
ADVERSE EFFECTS
Although generally well tolerated, P-CABs can cause some side effects, such as abdominal pain, diarrhea or constipation, nausea, headache, elevation of liver enzymes, and hypergastrinemia. So far, frequent serious adverse effects have not been reported, but their long-term use is still under evaluation in long-term follow-up studies14.
H2 receptor antagonists
H2 receptor antagonists were for many years the main pharmacological treatment for GERD, before the development of PPIs. Although they have been largely displaced by the latter, they still have a place in the management of certain patients with gastroesophageal reflux15.
The most common drugs in this class are ranitidine (withdrawn in many countries due to possibly carcinogenic impurities), famotidine (available in Mexico), nizatidine, and cimetidine.
MECHANISM OF ACTION
They act by reversibly blocking histamine H2 receptors located in gastric parietal cells. This reduces gastric acid production, especially basal secretion (not related to food intake). They inhibit the action of histamine on acid secretion, mainly decrease nocturnal acid secretion, and their effect is faster than that of PPIs, but less potent and of shorter duration3.
INDICATIONS IN GERD
- ‒ Patients who do not tolerate PPIs.
- ‒ Combination therapy with PPIs for control of nocturnal symptoms.
- ‒ Alternative in deprescription or on-demand management regimens.
They are used for short periods of time (2 to 3 weeks) due to the phenomenon of tachyphylaxis.
ADVERSE EFFECTS
They are generally well tolerated, but some adverse effects may occur, more frequently with cimetidine, such as headache, dizziness or fatigue, diarrhea or constipation, mental confusion (in elderly patients or with renal insufficiency), gynecomastia and sexual dysfunction (especially with cimetidine), and drug interactions (cimetidine inhibits several hepatic enzymes)2,3.
Antacids
They act by neutralizing already secreted gastric acid. They offer rapid symptomatic relief, but of short duration. They contain compounds such as magnesium hydroxide, aluminum, and calcium carbonate (Table 4).
Table 4. Main non-absorbable antacids
| Magnesium hydroxide |
| Mg salts are osmotically active: diarrhea; 15-20% renal excretion |
| Aluminum hydroxide |
| Al reacts with intestinal mucosa proteins: constipation |
| Impairs phosphate absorption: hypomagnesemia; 17-30% renal excretion |
| Aluminum and magnesium salts |
| Magaldrate (Mg and Al hydroxysulfate) |
| Almagate (hydrated Al and Mg hydroxycarbonate) |
| Melox®: Al and Mg hydroxide |
| Calcium carbonate |
| CaCO3 |
| Constipation |
| Combinations |
| Melox® night: calcium carbonate/magnesium alginate/magnesium carbonate/aluminum hydroxide |
Their use in GERD is restricted to the relief of occasional symptoms. They should not be used as long-term treatment due to their poor efficacy. Prolonged use may cause adverse events such as diarrhea, constipation, and hypomagnesemia.
Table 4 details the main antacids and their adverse effects²,³.
Alginates
Alginates are compounds derived from brown algae (such as Laminaria) that are frequently used in the symptomatic treatment of GERD, especially in mild forms or as a complement to other therapies2,3.
MECHANISM OF ACTION
Unlike drugs that act by reducing gastric acid production, alginates form a physical barrier that prevents the reflux of gastric contents toward the esophagus. Upon contact with gastric acid, alginates react, forming a thick, viscous, low-density gel. This gel floats on the gastric contents like a raft, acting as a mechanical barrier that prevents acid ascent toward the esophagus.
When reflux occurs, what reaches the esophagus is this gel, not the acidic content, reducing esophageal injury. Some formulations combine alginates with antacids (for example, sodium bicarbonate and calcium carbonate), which improves both mechanical protection and acid neutralization.
EFFICACY
They are especially useful in patients with postprandial symptoms or acid regurgitation. They can be used as on-demand treatment or as an adjuvant together with PPIs in patients with persistent symptoms. They do not heal erosive esophagitis, but significantly improve symptomatic control16.
ADVERSE EFFECTS
Alginates are well tolerated. Their adverse effects, infrequent and mild, include abdominal distension, occasional nausea, constipation, or diarrhea. Due to their safety profile, they can be used in pregnant women and pediatric patients under medical supervision16.
Bioadhesive barriers
Esoxx One is a medical device containing hyaluronic acid, chondroitin sulfate, and bioadhesive polymers (poloxamers), which is used in the adjuvant treatment of GERD.
MECHANISM OF ACTION
In vitro studies have shown that hyaluronic acid repairs and regenerates the mucosa, and chondroitin sulfate protects against acid-pepsin-bile damage. Both compounds, combined with poloxamer, form a mucoadhesive barrier that coats the esophagus with a protective film over the esophageal mucosa17.
INDICATIONS IN GERD
In controlled clinical trials, Esoxx One in adjuvant therapy with PPIs has been shown to be superior to PPIs alone for symptom control in non-erosive reflux disease. It is also used in combination with PPIs to accelerate the healing of erosive esophagitis. It can be used in pregnancy and lactation17.
ADVERSE EFFECTS
In general, Esoxx One is well tolerated. Mild nausea or hypersensitivity to any of the components has been occasionally described17.
Treatment of refractory GERD
It is important to know how to differentiate patients who have persistent GERD symptoms from those who have refractory GERD. The former are patients who have already been studied with endoscopy or esophageal pH monitoring, have objective evidence of GERD, and after treatment with double-dose PPIs for 8 weeks, continue with symptoms. Patients with refractory GERD are those who have demonstrated evidence of GERD and have not responded to treatment with double-dose PPIs for 8 weeks, and who, in a new diagnostic evaluation under PPI treatment, persist with esophagitis, with abnormal acid exposure, or with nocturnal baseline impedance < 1500 ohms or the number of reflux episodes > 80 in 24 hours18.
Treatment of persistent GERD symptoms includes the following options:
- ‒ Insist on lifestyle modifications (weight loss, head-of-bed elevation, early dinner, etc.) and adequate PPI dosing, 30 to 60 minutes before breakfast and dinner.
- ‒ Optimization of PPI treatment, which consists of using a different drug from the one initially used, preferably one that is not metabolized by CYP2C19, such as rabeprazole or ilaprazole, to circumvent the possibility of accelerated PPI metabolism.
- ‒ Use of adjuvant treatments, such as antacids, alginates, mucosal protectants, or a nocturnal dose of famotidine for short periods of 2 to 3 weeks.
- ‒ Change PPI treatment to a P-CAB. There is evidence that P-CABs can be effective in cases of PPI-refractory GERD symptoms.
Treatment of refractory GERD involves optimization of antisecretory agents, as noted, and the use of advanced treatments such as fundoplication in cases of large hiatal hernias, gastrojejunal bypass in patients with GERD and morbid obesity, and in selected patients endoscopic treatments, such as gastroplication, performed by endoscopists trained in these procedures18.
Conclusions
The treatment of GERD has evolved with the development of faster, more effective drugs with greater antisecretory potency. The appropriate use of P-CABs and PPIs represents the cornerstone in the medical treatment of this condition.
Knowledge of the pharmacology, pharmacodynamics, therapeutic efficacy, and safety of P-CABs and PPIs is indispensable for their correct prescription. Phenotyping of the patient with GERD through available diagnostic tests allows precision medicine and guarantees greater therapeutic success.
Funding
The author declares not having received funding for this study.
Conflicts of interest
The author declares having been a speaker for Carnot, Chinoin, and M8.
Ethical considerations
Protection of people and animals. The author declares that no experiments were performed on humans or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data or require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The author declares that no type of generative artificial intelligence was used for the writing of this manuscript.
