Introduction
The pathophysiology of gastroesophageal reflux disease (GERD) is complex and multifactorial. Unlike the traditional concept that attributed the disease to acid hypersecretion, it is now recognized that the development of GERD involves a dynamic imbalance between aggressor factors – such as the acidity of gastric contents, the frequency of reflux events, and esophageal sensitivity – and defense mechanisms – such as the integrity of the antireflux barrier, acid clearance, and esophageal mucosal resistance1,2.
Various anatomical and functional elements participate in the genesis of pathological reflux. Among them, transient lower esophageal sphincter (LES) relaxations constitute the most common mechanism in individuals without evident anatomical alterations. On the other hand, the presence of a hiatal hernia represents an anatomical alteration that weakens the effectiveness of the antireflux barrier and facilitates the migration of gastric contents toward the esophagus3–6.
In addition to these structural and functional components, the fundamental role of esophageal hypersensitivity and alterations in the gut-brain axis in the generation and perception of symptoms has been recognized. Psychological factors such as anxiety, depression, and sensory hypervigilance can amplify the symptomatic response to minor stimuli and even induce symptoms in the absence of abnormal acid exposure7,8.
In recent years, a new pathophysiological player has been proposed: the esophageal microbiota. Qualitative changes in the esophageal microbial population could influence mucosal inflammation, epithelial permeability, and progression toward premalignant lesions. Although its exact role is not yet fully elucidated, it constitutes an emerging research line with important clinical and therapeutic implications9.
The objective of this article is to provide a structured and critical review of the main pathophysiological mechanisms involved in GERD, integrating recent evidence on anatomical, neurofunctional, and microbial aspects. A comprehensive understanding of these factors is essential to optimize the diagnostic and therapeutic approach to patients with this condition.
Method
This article constitutes a narrative review based on published scientific literature on the pathophysiological mechanisms of GERD. A structured search was performed in the PubMed database using the MeSH descriptors “Gastroesophageal Reflux,” “Lower Esophageal Sphincter,” “Hiatal Hernia,” “Esophageal Motility Disorders,” “Esophageal Sensitivity,” “Gut-Brain Axis,” and “Esophageal Microbiome.” Articles published between 2015 and 2025, in English and Spanish, were selected, prioritizing systematic reviews, physiological studies, mechanistic studies in humans, and relevant clinical trials.
In addition, older literature published before 2015, that was considered of high historical and conceptual impact in the original description of the pathophysiological mechanisms of GERD was integrated; foundational studies on transient LES relaxations, crural diaphragm function, and the first histopathological characterizations of esophageal mucosa in the presence of acid reflux were included.
The review was organized around five pathophysiological axes: 1) transient LES relaxations, 2) antireflux barrier dysfunction (hiatal hernia), 3) alterations in esophageal defense mechanisms, 4) implications of the esophagus-brain axis, and 5) emerging role of the esophageal microbiota.
Since this work did not involve human or animal subjects, nor handling of sensitive information, approval by an ethics committee or obtaining informed consent was not required.
The objective was to conduct a critical, structured, and updated review of the pathophysiology of GERD, integrating recent advances with classic concepts, which serves as a basis for better clinical understanding and the development of rational therapeutic interventions.
Results
Key pathophysiological mechanisms of GERD
The pathophysiology of GERD is based on an imbalance between aggressor factors (frequency and characteristics of reflux) and defensive factors (antireflux barrier and mucosal integrity). Three pathophysiological pillars have been implicated in the development and progression of the disease: transient LES relaxations, anatomical alterations of the esophagogastric barrier (especially hiatal hernia), and dysfunctions in esophageal defense mechanisms (Table 1).
Table 1. Comparison of reflux mechanisms
| Mechanism | Description | Frequency in GERD | Associated factors |
|---|---|---|---|
| Transient LES relaxations | Prolonged relaxations (>10 s), not associated with swallowing, with crural diaphragm inhibition | 50-90% of episodes | Gastric distension, absence of hiatal hernia |
| Swallow-related relaxations | Brief relaxations (5-10 s) associated with peristalsis | 5-10% of episodes | Hiatal hernia, ineffective peristalsis |
| Hypotensive LES | Basal pressure < 5-10 mmHg, allows free or strain-induced reflux | Uncommon, up to 25% in severe esophagitis | Scleroderma, myotomy, hiatal hernia |
|
LES: lower esophageal sphincter; GERD: gastroesophageal reflux disease. |
|||
ANTIREFLUX BARRIER
The initial defense of the esophagus against acid damage is formed by the antireflux barrier. This barrier is characterized by being a complex anatomical region that includes the intrinsic LES, the crura of the diaphragm, the intra-abdominal position of the LES, the phrenoesophageal ligaments, and the angle of His.
The LES comprises the distal 3-4 cm of the esophagus and is tonically contracted at baseline10. It constitutes the main component of the antireflux barrier, with the ability to prevent reflux even when completely displaced from the diaphragmatic crura due to a hiatal hernia11. The proximal portion of the LES is normally located between 1.5 and 2 cm above the squamocolumnar junction, while the distal segment, approximately 2 cm in length, is situated within the abdominal cavity. Anatomical studies attribute to this part of the antireflux barrier a function similar to that of a flap valve, related to the sling and clasp muscle fibers of the gastric cardia. This location allows maintenance of gastroesophageal competence during increases in intra-abdominal pressure.
The basal pressure of the LES ranges between 10 and 30 mmHg, with considerable reserve capacity, as only a pressure of between 5 and 10 mmHg is required to prevent gastroesophageal reflux4. This high-pressure zone is maintained by the intrinsic tone of the LES muscle and the activity of excitatory cholinergic neurons. The basal pressure of the LES exhibits significant diurnal variations: after meals and reaches its highest point during the night; it also increases markedly during phase III of the migrating motor complex. It is also modulated by circulating peptides and hormones, foods (especially those rich in fat), and various drugs.
The LES is located within the diaphragmatic hiatus, formed by the right pillar of the diaphragm, and anchored by the phrenoesophageal ligaments, which insert at the level of the squamocolumnar junction. The hiatus is a teardrop-shaped channel, approximately 2 cm in its major axis. From an embryological development standpoint, the diaphragmatic pillar derives from the dorsal mesentery of the esophagus and has innervation distinct from that of the costal diaphragm. This structure is inhibited in response to esophageal distension, during vomiting, and during transient LES relaxations, but not during swallowing. The crural diaphragm provides extrinsic compression to the intrinsic LES, contributing to resting pressure during inspiration and increasing LES pressure during episodes of elevated abdominal pressure, as occurs with coughing, sneezing, or bending12. Crural pillar contractions generate rhythmic pressure increases on the LES, on the order of 5 to 10 mmHg. During deep inspirations or episodes of intense abdominal strain, these increases can reach values between 50 and 150 mmHg13.
The oblique entry of the esophagus into the stomach creates an acute angle on the greater curvature aspect of the esophagogastric junction (EGJ), known as the angle of His. Studies performed in cadavers have demonstrated that this angle produces a flap valve effect; however, its specific contribution to EGJ competence remains unclear14.
MECHANISMS OF REFLUX
Reflux usually occurs through four main mechanisms: transient lower esophageal sphincter relaxations, low LES pressure, swallow-related LES relaxation, and strain during periods of decreased LES pressure (Table 2).
Table 2. Protective and aggressor factors in gastroesophageal reflux disease
| Protective factors | Aggressor factors | Impact on GERD |
|---|---|---|
| Antireflux barrier (LES, crural diaphragm, angle of His) | Transient LES relaxations, hiatal hernia | Increases reflux exposure |
| Acid clearance (peristalsis, saliva) | Ineffective motility, hyposalivation | Prolongs acid exposure |
| Mucosal integrity | Dilation of intercellular spaces, increased permeability | Esophageal hypersensitivity |
| Gut-brain axis modulation | Anxiety, depression, hypervigilance | Amplifies symptom perception |
|
LES: lower esophageal sphincter; GERD: gastroesophageal reflux disease. |
||
- − Transient LES relaxations
- They constitute the predominant reflux mechanism in both healthy individuals and patients with GERD, particularly in those with normal LES pressures and without hiatal hernia. These relaxations are spontaneous, prolonged episodes (> 10 seconds), not related to swallowing, that are not accompanied by esophageal peristalsis and are associated with active inhibition of the crural diaphragm. Their primary physiological stimulus is distension of the proximal stomach – especially the fundus – by food or gas, which is consistent with their physiological function as a belching mechanism3,5,6,15.
- This distension activates a complex vagal reflex arc, which begins in mechanoreceptors of the intraganglionic laminar nerve endings of vagal afferents, projecting to the brainstem through the nucleus of the solitary tract, toward the dorsal motor nucleus of the vagus. From there, signals are transmitted to inhibitory neurons located in the myenteric plexus of the distal esophagus. This circuit produces an integrated motor response that includes LES relaxation, contraction of the longitudinal musculature with esophageal shortening, decreased obstruction at the EGJ, crural diaphragm inhibition, and costal diaphragm contraction16.
- Studies with high-resolution manometry and impedance have demonstrated that transient LES relaxations explain virtually all reflux episodes in healthy individuals and between 50% and 90% of events in patients with GERD, depending on the presence of hiatal hernia and the severity of associated esophagitis. In healthy individuals, between 40% and 60% of transient LES relaxations are accompanied by reflux, while in patients with GERD this percentage rises to 60-70%. Some factors that could influence the occurrence of reflux during a transient LES relaxation are abdominal strain, the presence of hiatal hernia, the degree of esophageal shortening, and the duration of the relaxation episode.
- Although physiological in most cases, in GERD these relaxations become more frequent, prolonged, and with a greater degree of LES opening, resulting in greater exposure of the esophagus to gastric acid, bile, and pepsin3,5,6,15.
- Pharmacological inhibition of transient LES relaxations has been extensively studied. Various compounds – including CCK-A receptor antagonists, anticholinergic drugs, morphine, somatostatin, nitric oxide inhibitors, 5-HT3 receptor antagonists, and GABAβ receptor agonists – have shown the ability to reduce the frequency of these events; however, to date, no targeted therapy that is both effective and safe for widespread clinical use is available17.
- − Swallow-related LES relaxations
- Approximately 5% to 10% of reflux episodes occur during swallow-induced LES relaxations. Most of these episodes are associated with defective or incomplete peristalsis. During a swallow-induced LES relaxation, under normal conditions, reflux is uncommon because the crural diaphragm does not relax, the duration of LES relaxation is relatively brief (5 to 10 seconds), and the approaching peristaltic wave acts as a barrier, preventing reflux18.
- Reflux during swallow-induced relaxations is more frequent in the presence of hiatal hernia. This could be explained by lower distensibility of the EGJ in patients with hernia, which allows its opening at pressures equal to or even lower than intragastric pressure, thus facilitating reflux of gastric contents accumulated within the hernial sac.
- − Hypotensive LES
- Gastroesophageal reflux can occur in the context of LES hypotension, either as strain-induced reflux or as free reflux. Strain-induced reflux occurs when the LES with relatively low pressure is overcome by a sudden increase in intra-abdominal pressure, as occurs with coughing, straining, or bending. This type of reflux is unlikely when LES pressure is > 10 mmHg and no hiatal hernia exists19.
Free reflux is characterized by a drop in esophageal pH without an identifiable change in intragastric pressure, and usually occurs when LES pressure is < 5 mmHg. Reflux due to absent or severely reduced LES pressure is uncommon; it is typically observed in patients with advanced scleroderma and in those who have undergone myotomy for achalasia. In general, it occurs in patients with severe esophagitis, in whom it may represent up to 25% of reflux episodes, and rarely in patients without esophagitis4.
These last three reflux mechanisms are observed almost exclusively in the presence of hiatal hernia. These observations reinforce the concept that the functional integrity of the EGJ depends on both the intrinsic LES and the extrinsic sphincteric component provided by the diaphragmatic hiatus. In essence, reflux requires two “hits” on the EGJ. In patients with an anatomically normal EGJ, simultaneous inhibition of the LES and crural diaphragm is needed for reflux to occur (i.e., through transient LES relaxations). In contrast, when a hiatal hernia that compromises crural sphincter function exists, reflux can occur solely with LES relaxation, during periods of sphincteric hypotension, swallow-induced relaxation, or strain maneuvers.
ANATOMICAL ALTERATIONS: HIATAL HERNIA
The presence of hiatal hernia does not imply the presence of GERD, as many subjects have no evidence of GERD despite having hiatal hernia.
However, hiatal hernia is the most frequent anatomical abnormality associated with GERD, and is significantly more common in patients with erosive esophagitis compared to healthy individuals20. This condition involves axial displacement of the LES and gastric fundus toward the mediastinum, separating the functional overlap of the LES with the crural diaphragm and eliminating the angle of His. As a result, closing pressure at the EGJ decreases and exposure to acid reflux increases both in the supine position and during strain or swallowing maneuvers.
The severity of reflux shows a direct correlation with the size of the hiatal hernia. Patients with hernias > 3 cm have a higher risk of erosive esophagitis, Barrett’s esophagus, and complications such as strictures. The hernia also contributes to a phenomenon called “re-reflux,” in which content trapped in the hernial sac repeatedly returns to the esophagus after each swallow, hampering esophageal clearance21.
In addition, large hernias impair the ability of the crural diaphragm to exert its extrinsic sphincteric function and modify the geometry of the antireflux valve, which are key elements in the competence of the antireflux barrier.
DYSFUNCTION OF ESOPHAGEAL DEFENSE MECHANISMS
Once reflux occurs, the extent of damage depends largely on the effectiveness of esophageal defense mechanisms, which include acid clearance and the integrity of the esophageal mucosa.
Esophageal acid clearance
This process has two components: a mechanical one, mediated by primary and secondary peristalsis, and a chemical one, provided by neutralization of residual acid through bicarbonate-rich saliva. In patients with GERD, esophageal motility is commonly ineffective, defined by peristaltic contractions of low amplitude (< 450 mmHg·cm/s) that contribute to slow and prolonged clearance. Hyposalivation, commonly observed during sleep, and in smokers, also delays residual acid neutralization22–24.
Mucosal integrity
Under normal conditions, the esophageal mucosa acts as a barrier that prevents the diffusion of irritant substances toward the submucosa. In GERD, a decrease in basal esophageal impedance and dilation of intercellular spaces have been observed, which allow the activation of subepithelial nerve endings even with subthreshold acid exposure. These findings explain the appearance of symptoms in patients with non-erosive reflux or with normal pH-metry, and reinforce the notion that epithelial damage does not necessarily require evident esophagitis7,8,25.
The perception of reflux is the result not only of peripheral stimuli but also of their processing in the central nervous system (Fig. 1). In this context, the esophagus-brain axis has emerged as a central component in the pathophysiology of GERD, especially in subgroups with refractory symptoms or in the absence of clinical-endoscopic correlation8.
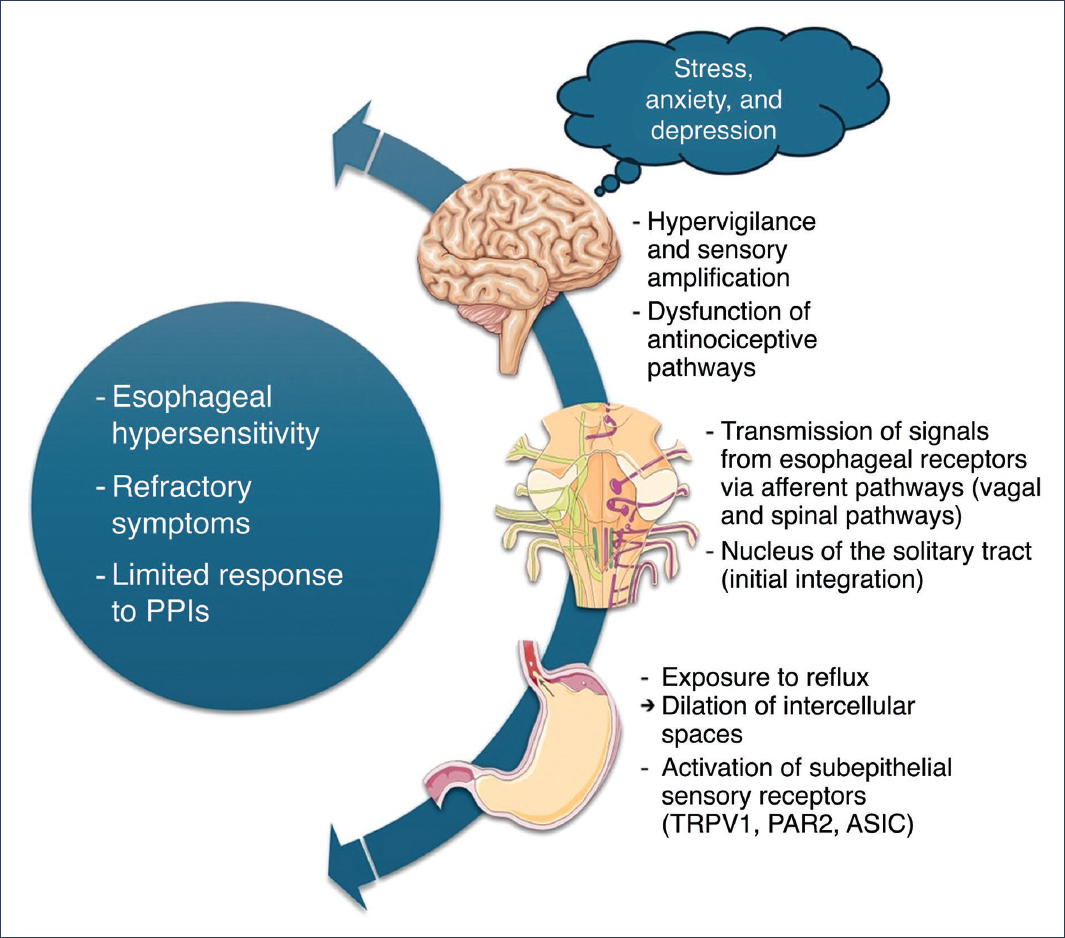
Figure 1. Esophagus-brain axis interactions in the pathophysiology of gastroesophageal reflux disease (GERD). The diagram illustrates the bidirectional relationship between the esophagus and the central nervous system in the perception and modulation of GERD symptoms. In the esophagus, exposure to reflux causes dilation of intercellular spaces and activation of subepithelial sensory receptors (TRPV1, PAR2, ASIC), initiating the transmission of afferent signals via vagal and spinal pathways to the nucleus of the solitary tract for integration. In the brain, hypervigilance, sensory amplification, and dysfunction of antinociceptive pathways contribute to esophageal hypersensitivity, while stress, anxiety, and depression modulate symptom perception, resulting in refractory symptoms and limited response to proton pump inhibitors (PPIs) (image created using graphic elements provided by Servier Medical Art, under Creative Commons Attribution 4.0 International license).
Studies have demonstrated that patients with GERD exhibit greater sensitivity to mechanical, thermal, chemical, and electrical stimuli applied to the esophagus, a phenomenon known as esophageal hypersensitivity. This is due to both peripheral mechanisms (increase in density or expression of TRPV1, PAR2, or ASIC receptors) and central mechanisms (hypervigilance, sensory amplification, and dysfunction of descending antinociceptive pathways)26.
Stress, anxiety, and depression modulate the activity of the esophagus-brain axis, increasing the perception of reflux and even inducing changes in epithelial permeability. Exposure to acute stress, for example, in animal studies has been associated with dilation of mucosal intercellular spaces, suggesting a direct link between the emotional and the physiological27,28.
These mechanisms explain why some patients present severe symptoms with minimal structural alterations, and why the response to proton pump inhibitors is limited in certain individuals. Treatment in these cases may include neuromodulators, cognitive-behavioral psychotherapy, or relaxation techniques.
Esophageal microbiota
The esophageal microbiota has been traditionally understudied, but analyses based on 16S rRNA sequencing techniques have revealed that a diverse bacterial community exists in the esophagus, whose profile can vary in different clinical conditions29.
In patients with GERD and Barrett’s esophagus, a change in microbial composition has been observed, with a relative increase in gram-negative anaerobic microorganisms, such as Prevotella, Fusobacterium, and Veillonella, compared to the predominance of Streptococcus in healthy individuals30.
It is postulated that these changes could induce or perpetuate inflammatory processes through activation of Toll-like receptors, proinflammatory cytokines, and epithelial barrier disruption31. An interaction of the altered microbiota, intraluminal acidity, and susceptibility to esophageal metaplasia has also been suggested, although these mechanisms are not yet fully understood32.
The role of the microbiota is an emerging area of research with therapeutic potential, which opens the door to future interventions such as the use of probiotics, targeted antibiotics, or microbiome modulators in the personalized management of GERD.
Discussion
The pathophysiology of GERD represents a complex, dynamic, and multifactorial phenomenon. Although gastric acid secretion plays an undeniable role, in recent decades it has become clear that the mere presence of acid is not sufficient to explain the clinical diversity or severity of the disease. This article has integrated functional, anatomical, sensory, and microbiological findings that allow a more complete understanding of the pathophysiological process.
Transient LES relaxations emerge as the main reflux mechanism in individuals without hiatal hernia. This has been demonstrated in studies with high-resolution manometry and impedance, in which most reflux episodes are not accompanied by basal LES hypotonia, but rather by repetitive and prolonged transient LES relaxations. This observation has led to considering transient LES relaxations, rather than as a pathological alteration per se, as an exaggerated or dysfunctional response to normal gastric stimuli.
Hiatal hernia, on the other hand, represents a critical anatomical component. Its contribution to GERD is not limited to altering the anatomy of the LES, as it also compromises crural diaphragm function and the antireflux valve, decreasing closing pressure and increasing susceptibility to postprandial and nocturnal reflux. These findings support surgical intervention (such as fundoplication) in patients with significant hiatal hernia and refractory reflux.
Regarding esophageal defense mechanisms, current evidence highlights that ineffective esophageal motility and hyposalivation contribute to prolongation of acid exposure time, exacerbating mucosal damage. However, the most notable finding is that even patients with “physiological” reflux can experience severe symptoms, due to alterations in epithelial integrity (such as dilation of intercellular spaces), which reinforces the concept of esophageal hypersensitivity as a key aspect in the pathophysiology of non-erosive reflux.
The esophagus-brain axis has gained importance as a critical modulator of reflux perception. Numerous studies have demonstrated that factors such as anxiety, depression, and sensory hypervigilance amplify the symptomatic experience, independently of the degree of acid exposure. This neuropsychological dimension not only explains why some patients present severe symptoms with minimal structural alterations but also why the response to proton pump inhibitors is limited in certain individuals. Treatment in these cases may include neuromodulators, cognitive-behavioral psychotherapy, or relaxation techniques.
Finally, the role of the esophageal microbiota constitutes an emerging frontier in the pathophysiology of GERD. Although current data are still limited, preliminary evidence suggests that an altered microbiome could contribute to chronic inflammation, epithelial permeability, and progression toward Barrett’s esophagus. This introduces a new ecological and therapeutic dimension, in which bacterial modulation strategies could be incorporated into the comprehensive management of the disease.
Conclusions
GERD cannot be understood exclusively as an alteration in gastric acid secretion. Its pathophysiology reflects the complex interaction of anatomical, physiological, sensory, and neuroimmune dysfunctions.
Transient LES relaxations constitute the primary reflux mechanism in patients without evident anatomical abnormalities, while the presence of hiatal hernia disrupts the antireflux barrier, exacerbating the severity of reflux and its mucosal injury. In turn, alterations in acid clearance and mucosal integrity explain the persistence of epithelial damage even in situations of moderate reflux.
The esophagus-brain axis is a key dimension for understanding symptomatic variability, clinical-endoscopic dissociation, and therapeutic refractoriness. Psychiatric comorbidity and central mechanisms of sensory amplification modify the experience of reflux, demanding more comprehensive diagnostic and therapeutic approaches.
Finally, recent findings on the esophageal microbiota suggest that its composition may influence inflammation, epithelial permeability, and potentially carcinogenesis, opening new research and treatment perspectives.
A multidimensional understanding of the GERD pathophysiology is essential for precision medicine. Future research should integrate functional, psychological, and microbial biomarkers, as well as validate personalized interventions beyond simple acid suppression.
Funding
The author declares having received no funding for this study.
Conflicts of interest
The author declares having no conflicts of interest.
Ethical considerations
Protection of human and animal subjects. The author declares that no experiments were performed on humans or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data nor require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The author declares not having used any type of generative artificial intelligence for the writing of this manuscript.
