Introduction
Gastroesophageal reflux disease (GERD) is one of the most frequent diseases worldwide. The prevalence rate is estimated to range between 13% and 20% of the general population, although there is considerable regional variability1,2. GERD, as defined by the Montreal consensus, is a condition in which symptoms are recognized as esophageal manifestations (further divided into symptoms-only presentations and those with mucosal injury) and extraesophageal manifestations (divided in turn into those with and without established association)2,3 (Fig. 1). Likewise, these syndromes show three phenotypic presentations: non-erosive GERD (60-70% of patients), erosive GERD (around 30%), and Barrett’s esophagus (6-8% of cases)4.
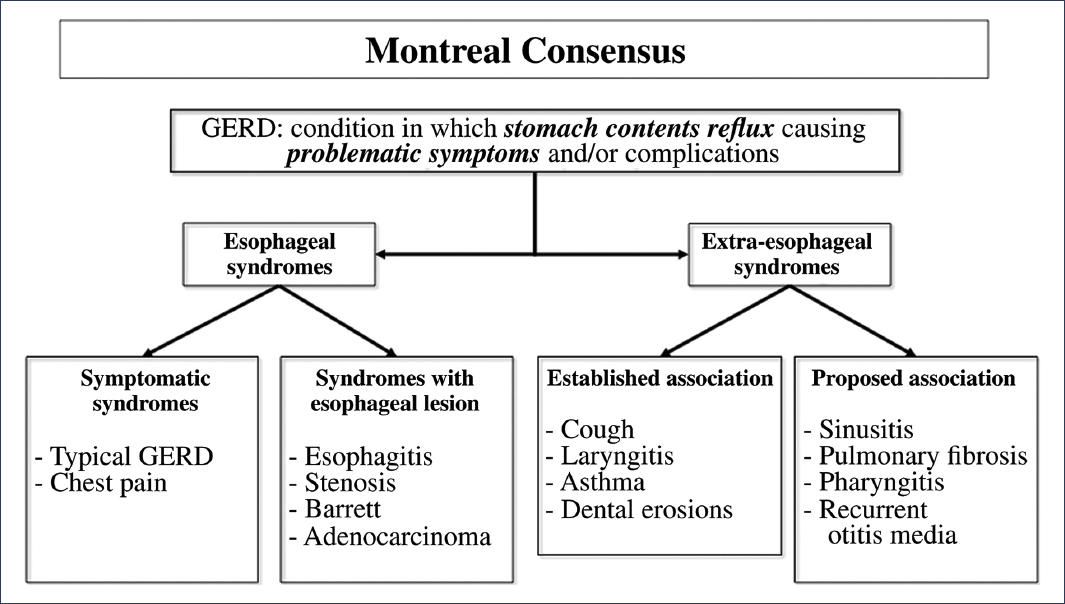
Figure 1. Classification of gastroesophageal reflux disease (GERD) according to the Montreal consensus.
Despite the different modalities and combinations of management that currently exist, it has been reported that up to 40% of patients fail medical treatment due to multiple causes (Table 1). Currently, refractory GERD is defined by the presence of objective evidence of GERD (erosive esophagitis, or abnormal acid exposure, or an elevated number of reflux episodes in ambulatory esophageal pH monitoring) despite optimal pharmacological treatment2.
Table 1. Causes of persistent symptoms of gastroesophageal reflux disease
| Lack of adherence to treatment |
| Inadequate prescription or administration |
| Genetic polymorphisms (PPI metabolism) |
| Poor quality of drugs |
| Esophageal hypersensitivity |
| Hypervigilance, anxiety, somatization |
| Functional heartburn |
| Other esophageal diseases: |
| Eosinophilic esophagitis |
| Pill esophagitis (NSAIDs, potassium, bisphosphonates) |
| Infectious esophagitis |
| Esophageal motility disorders: |
| Achalasia |
| Distal esophageal spasm |
| Hypercontractile esophagus |
| Gastroduodenal disorders: |
| Excessive supragastric belching |
| Rumination syndrome |
| Others: |
| Zollinger-Ellison syndrome |
| Gastroparesis |
|
NSAIDs: nonsteroidal anti-inflammatory drugs; PPI: proton pump inhibitors. Taken from Valdovinos Díaz, et al.2 |
This review will describe the reflux phenotypes found in clinical practice, as well as the definition of truly refractory GERD.
Non-erosive gastroesophageal reflux disease
Definition
Among the GERD phenotypes, non-erosive reflux disease (NERD) is the most frequent in clinical practice, occurring in up to 70% of patients evaluated by endoscopy2,5. Patients may have typical and atypical manifestations of reflux, but endoscopy shows no abnormalities in the esophagus or in the biopsies obtained from it. Figure 2 shows a typical endoscopic image of a patient with NERD.
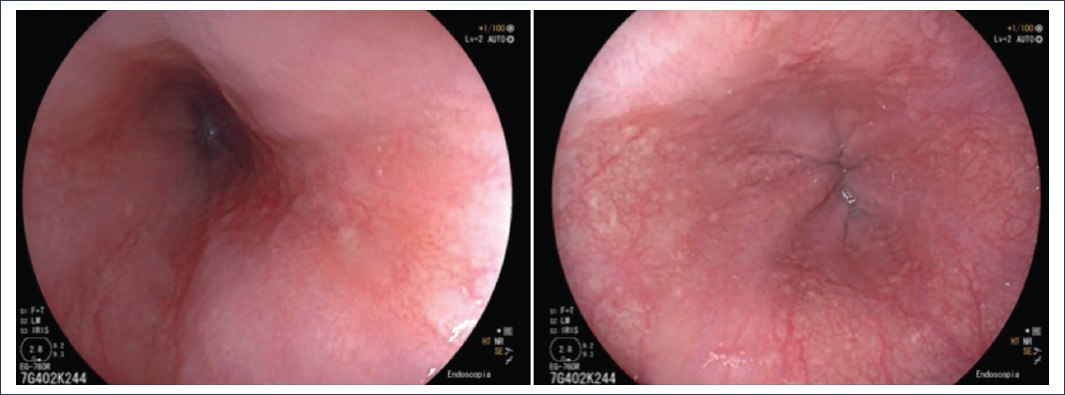
Figure 2. Representative images of an endoscopy without significant findings.
In this group of patients, it is essential to make a precision diagnosis through ambulatory pH-metry, since, as illustrated in Figure 3, only 50% will have “true NERD,” while the rest will suffer from a functional esophageal disorder (functional heartburn and reflux hypersensitivity).
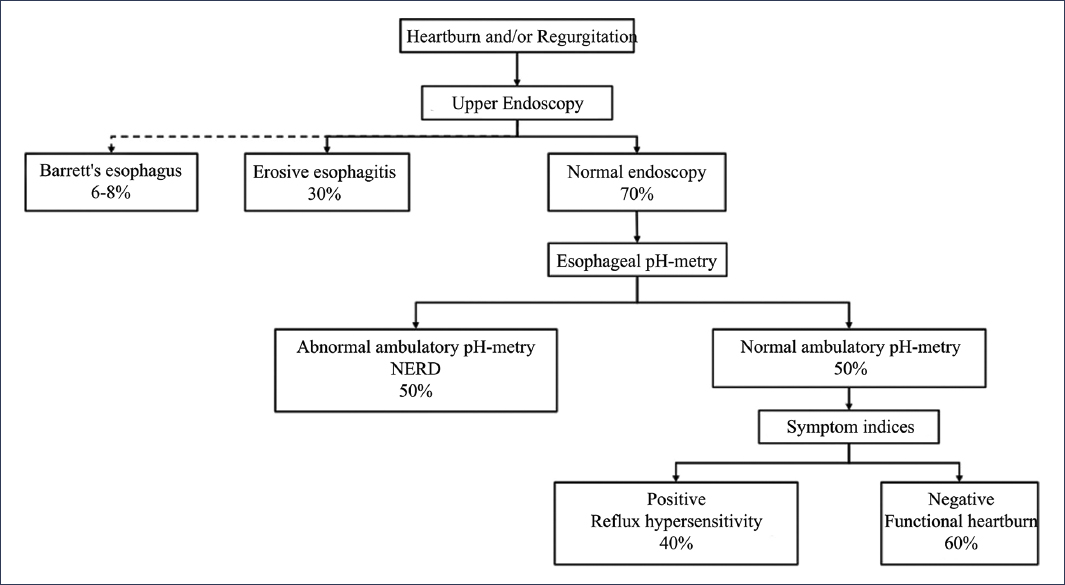
Figure 3. Reflux phenotypes. NERD: non-erosive gastroesophageal reflux disease (adapted and translated from Yamasaki and Fass5).
On many occasions, the diagnosis of this condition is challenging because the clinical presentation and pathophysiology are very heterogeneous and are often indistinguishable without tests that allow a precise diagnosis6. Addressing this problem, the Lyon 2.0 consensus4 (Fig. 4) has provided a series of criteria and tools that help discriminate between patients in this group; thus, conclusive evidence for the diagnosis of GERD is defined by an acid exposure time (AET) greater than 6%. When AET is between 4% and 6%, complementary criteria are needed to have better certainty in the diagnosis, which include the number of reflux episodes, histology, and baseline impedance metrics, among others.
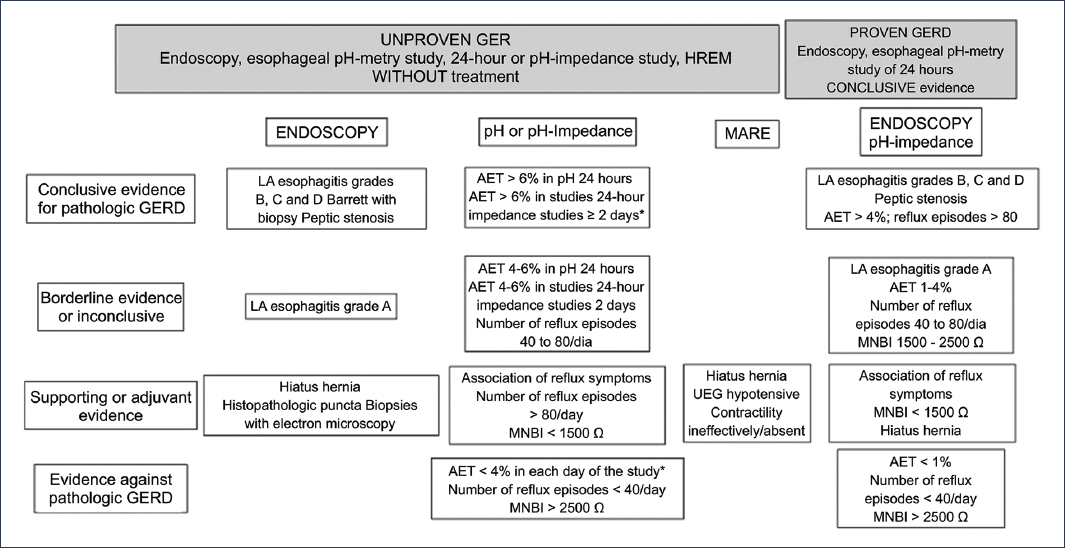
Figure 4. Lyon 2.0 consensus for the diagnosis of gastroesophageal reflux disease (GERD)4,11. LA: Los Angeles; HREM: high-resolution esophageal manometry; MNBI: mean nocturnal baseline impedance; AET: acid exposure time; EGJ: esophagogastric junction. *The ambulatory monitoring value must be met throughout the entire evaluated period.
Risk factors and clinical relevance
As already mentioned, NERD is the most frequent phenotype of GERD, and its complexity lies in the clinically indistinct presence of true NERD versus other differential diagnoses, such as reflux hypersensitivity and functional heartburn.
There is evidence that, although not specifically for this subgroup, the presence of certain factors increases the probability of reflux; these are listed in table 2, which includes the characteristics that can explain the high prevalence of this condition. Given the similarities in terms of risk factors and clinical presentation, a precision diagnosis is essential in this group, since the implications for the management and maintenance of the different conditions included in this spectrum of patients are significant.
Table 2. Risk factors for gastroesophageal reflux disease
| Risk factor | OR |
|---|---|
| Age ≥ 50 years | 1.32 (1.12-1.54)* |
| Smoking | 1.26 (1.04-1.52)* |
| NSAID use | 1.44 (1.10-1.88)* |
| Obesity | 1.73 (1.146-2.06)* |
| Elevated waist/hip ratio | 1.94 (1.12-5.23)† |
| Alcohol consumption | 1.51 (1.13-1.99)† |
| Former smoker | 1.43 (1.13-1.90)† |
|
* Taken from Eusebi, et al.38.† Taken from Sadafi, et al.39. NSAIDs: nonsteroidal anti-inflammatory drugs; OR: odds ratio (95% CI). |
|
Erosive gastroesophageal reflux disease
Definition, risk factors, and clinical relevance
Erosive GERD is the second most frequent phenotype in this pathology. According to descriptive studies, it is estimated that between 10% and 30% of patients will have erosions during endoscopic evaluation2,7. Thus, performing an upper endoscopy is essential not only for the identification of erosions but also to quantify the severity and thereby guide the appropriate treatment according to the risk that this finding represents for the patient8.Although there are numerous scales to standardize endoscopic findings in GERD, the Los Angeles Classification, published in 1996 and subsequently validated, is the one that currently offers the best standardization and correlation with clinical outcomes9,10. Figure 5 shows illustrative images as well as the description of the Los Angeles classification. It is important to note that Los Angeles grade A esophagitis, given its high interobserver variability, is not considered conclusive evidence of reflux and therefore is not included within this group of patients11.
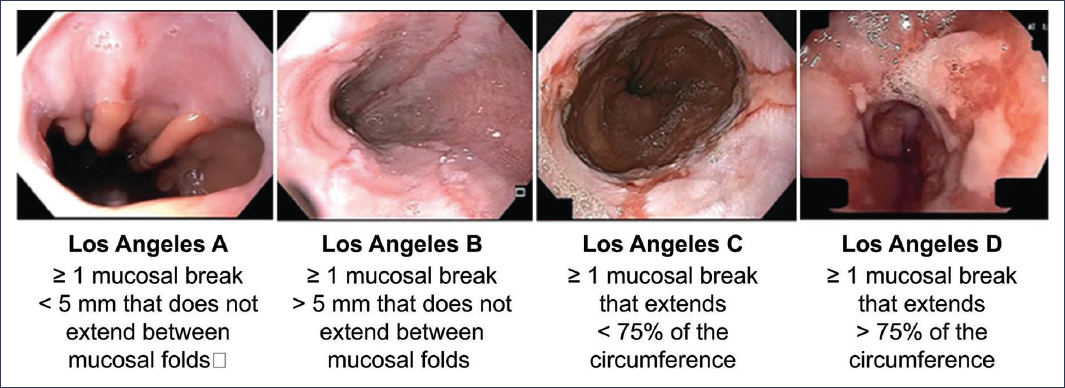
Figure 5. Graphic representation of the Los Angeles classification (taken and translated from Lundell et al.10 and Spechler et al.40).
Regarding the rest of the Los Angeles classification groups (esophagitis B, C, and D), they are considered conclusive evidence of GERD2,11 (Fig. 6) and require medical treatment for 8 weeks2. It is notable that the severity of esophagitis correlates with the endoscopic cure rate, as illustrated by a Japanese multicenter study in which patients with grade C and D esophagitis had healing rates of approximately 70% and 60%, respectively12, and with recurrence rates up to 75% under standard treatment13,14.
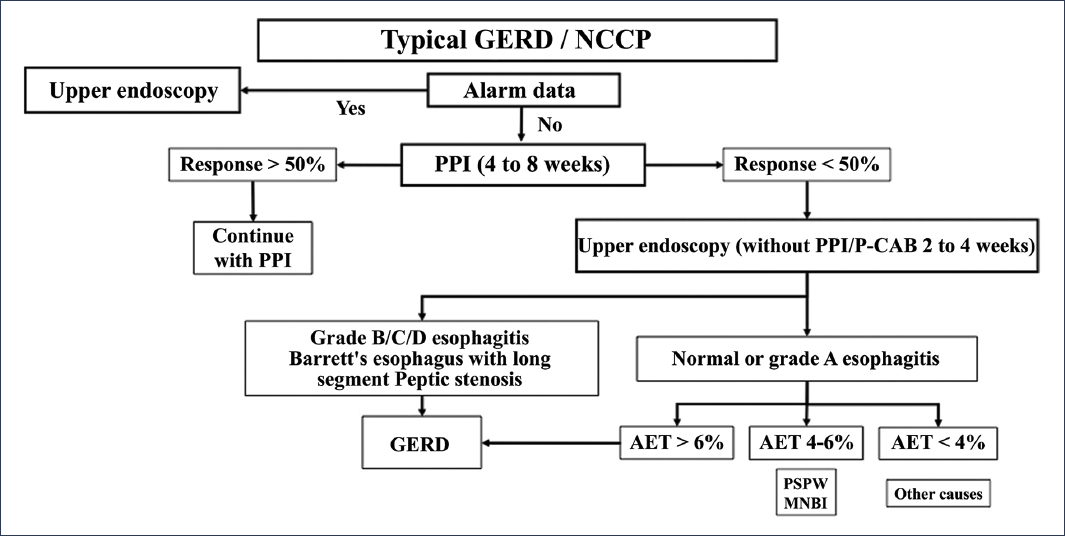
Figure 6. Classification of patients based on diagnostic studies in gastroesophageal reflux disease (GERD). NCCP: non-cardiac chest pain; PPI: proton pump inhibitor; MNBI: mean nocturnal baseline impedance; P-CAB: potassium-competitive acid blocker; PSPW: post-swallow peristaltic wave; AET: acid exposure time (modified from Valdovinos-Díaz et al.2).
Barrett’s esophagus
Definition, risk factors, and clinical relevance
Barrett’s esophagus is a premalignant lesion for the presence of esophageal adenocarcinoma characterized by the replacement of squamous epithelium with intestinal metaplasia with columnar epithelium, alternating with goblet cells15,16. It is considered one of the complications of chronic GERD and constitutes a biological response to persistent injury to the esophageal mucosa.
It is estimated that approximately 6% to 8% of patients with GERD present Barrett’s esophagus2, being more frequent in Caucasian males in the sixth decade of life17. However, some studies suggest that the incidence of Barrett’s esophagus is not different in the Hispanic population18. There are several risk factors for the presence of this disease (Table 3), most of which are also risk factors for esophageal adenocarcinoma19. A meta-analysis that included 49 studies and 307,273 individuals demonstrated that in patients with reflux symptoms and at least one proposed risk factor, the proportion of Barrett’s esophagus was higher than in those without reflux symptoms (12.2% vs. 0.8%). In this same meta-analysis, the presence of certain risk factors was associated with a higher presence of Barrett’s esophagus, such as family history (23.4%), male sex (6.8%), age > 50 years (6.1%), and central obesity (1.9%)20.
Table 3. Proposed risk factors for the presence of Barrett’s esophagus and esophageal adenocarcinoma
| Factor | Risk for Barrett’s esophagus | Risk for esophageal adenocarcinoma |
|---|---|---|
| Age | Yes | Yes |
| White race | Yes | Yes |
| Male sex | Yes | Yes |
| Chronic heartburn | Yes | Yes |
| Age < 30 years at symptom onset | Yes | – |
| Hiatal hernia | Yes | Yes |
| Erosive esophagitis | Yes | Yes |
| Obesity with intra-abdominal distribution | Yes | Yes |
| Metabolic syndrome | Yes | Yes |
| Tobacco use | Yes | Yes |
| Family history of Barrett’s esophagus or esophageal adenocarcinoma | Yes | Yes |
| Obstructive sleep apnea | Yes | – |
| Low birth weight for gestational age | Yes | No |
| Preterm birth | No | Yes |
| Red meat or processed meat consumption | Yes | Yes |
| Human papillomavirus infection | No | Yes |
|
Taken from Spechler and Souza19. |
||
The risk of progression of Barrett’s esophagus has been described in studies to range from 0.1% to 3% per year21,22, and depends on the degree of structural alteration of the epithelium, that is, the degree of dysplasia19. In general, the risk of progression to esophageal adenocarcinoma is estimated at 0.25% per year, but for patients with non-dysplastic Barrett’s esophagus, it is even lower, while in those with low-grade dysplasia, it is estimated at approximately 0.54% per year, and in those with high-grade dysplasia, it is 4% to 8%23–25.
Faced with this problem, it is essential to use standardized reporting systems for endoscopic and histological characteristics15. Currently, the Prague classification reporting system is recommended, which includes the description of circumferential involvement and the maximum extent of columnar epithelium26, and has been shown to have an excellent reliability coefficient for both parameters (0.95 for circumferential and 0.94 for maximum extent)27. Additionally, to perform biopsies, it is recommended to use the Seattle protocol, which indicates taking them in four quadrants in segments spanning from 1 to 2 cm circumferentially28,29. Use of this protocol has been shown to increase dysplasia detection in Barrett’s esophagus up to 13-fold30 (Fig. 7). Finally, one of the aspects that can substantially contribute to the correct classification of the degree of dysplasia is minimizing interobserver variability in the pathological interpretation of biopsies31. Although there is currently no consensus on the required experience of the pathologist, current guidelines recommend interpretation by two pathologists with experience in interpreting esophageal pathology, especially in cases of high-grade dysplasia27.
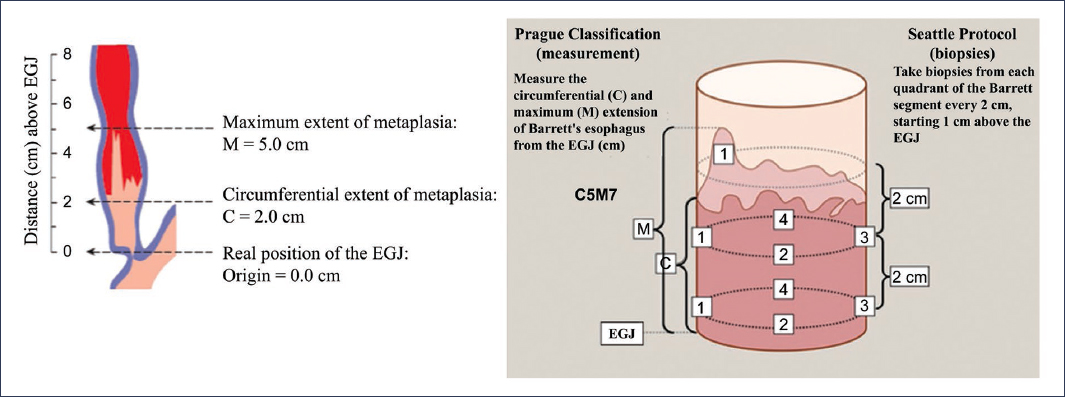
Figure 7. Prague classification reporting system and Seattle protocol in Barrett’s esophagus26,28–30. EGJ: esophagogastric junction.
Refractory gastroesophageal reflux disease
Definition, risk factors, and clinical relevance
The persistence of GERD symptoms occurs in 30% to 40% of patients despite appropriate medical treatment32,33. This frequent scenario does not necessarily imply the presence of refractory GERD, since there are numerous conditions that can explain the persistence of GERD symptoms despite optimal treatment (Table 1). Thus, it is essential to take a systematic approach to this group of patients, depending on the presence or absence of objective evidence of reflux (erosive esophagitis, Barrett’s esophagus, and abnormal AET)2, recognizing two subgroups of patients: those without objective evidence of GERD and those with objective evidence of GERD. In the first group, an appropriate diagnostic approach should be considered, which implicitly recommends studying patients before attempting a new treatment regimen (Figs. 1 and 8). In this group of patients, a large proportion will have a non-GERD-related diagnosis to explain the symptoms, including functional esophageal disorders34,35. On the other hand, in the group with objective evidence of GERD and persistence of symptoms, before considering an evaluation, it is recommended to verify adherence to and appropriate use of medical treatment, and subsequently consider performing diagnostic evaluation under medical treatment2 (Fig. 9). It is important to clarify that, in view of what was previously discussed, the definition of true refractory GERD requires objective evidence of GERD (erosive esophagitis, Barrett’s esophagus, or abnormal acid exposure) despite optimal medical treatment for at least 8 weeks32,36.
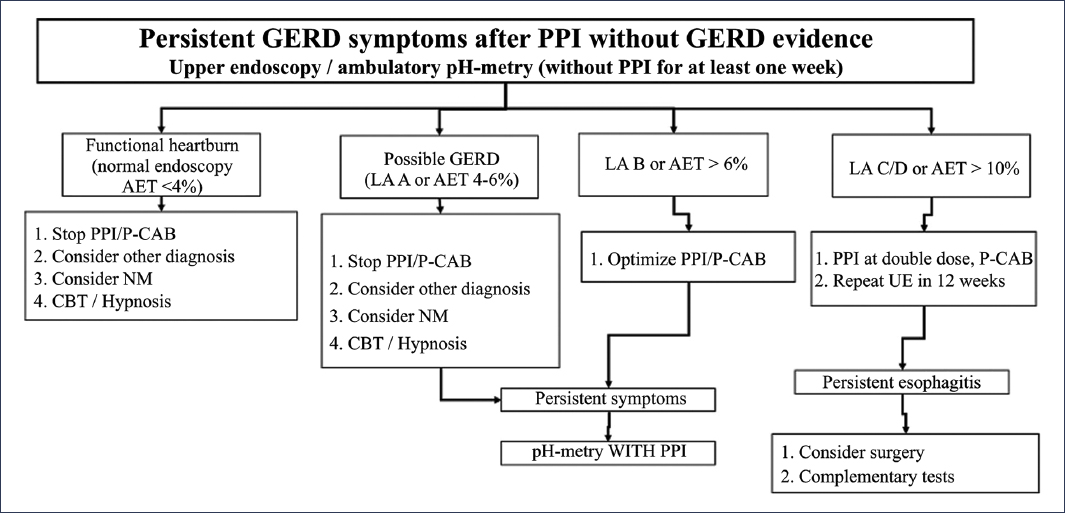
Figure 8. Diagnostic and therapeutic algorithm for patients with persistent reflux symptoms without previous objective evidence of gastroesophageal reflux disease (GERD)2. UE: upper endoscopy; PPI: proton pump inhibitor; LA: Los Angeles; NM: neuromodulator; P-CAB: potassium-competitive acid blocker; CBT: cognitive-behavioral therapy; AET: acid exposure time.
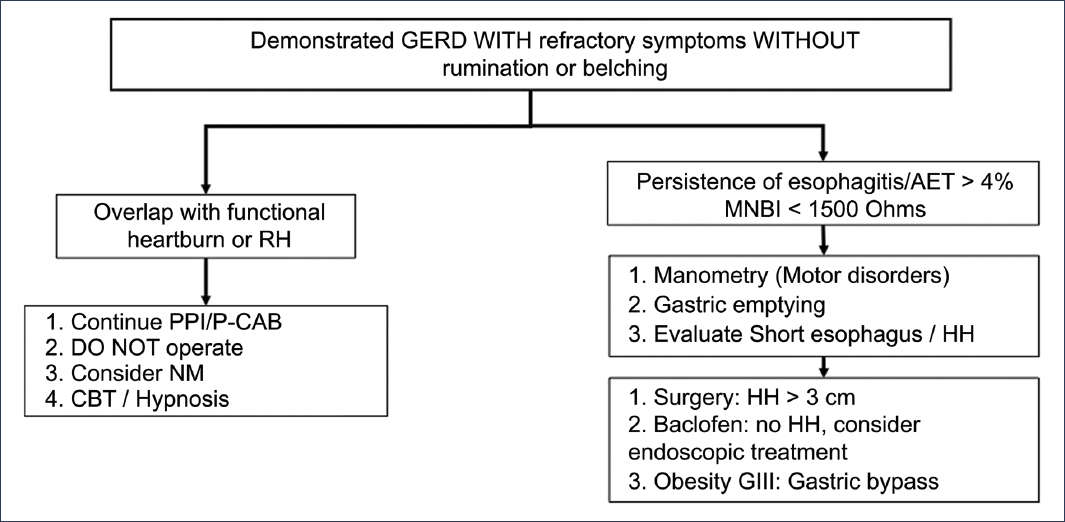
Figure 9. Diagnostic and therapeutic algorithm for patients with persistent reflux symptoms with previous objective evidence of gastroesophageal reflux disease (GERD)2. HH: hiatal hernia; RH: reflux hypersensitivity; PPI: proton pump inhibitor; MNBI: mean nocturnal baseline impedance; NM: neuromodulator; P-CAB: potassium-competitive acid blocker; CBT: cognitive-behavioral therapy; AET: acid exposure time.
According to this definition, the criteria for pH measurement and impedance under treatment are considered different from those included in Lyon 2.011, with thresholds of 0.5% for AET and 40 reflux episodes used as criteria to predict response to escalated GERD treatment37.
Correct clinical classification (rather than phenotypic) is crucial due to its implications for appropriate patient management and its proper sequencing. Patients with true refractory GERD may benefit from more potent alternatives or high doses of drugs, and in selected cases, surgical intervention. In contrast, patients with functional disorders (esophageal hypersensitivity or functional heartburn, among others) benefit more from neuromodulators or psychological interventions2.
Conclusion
In the study and treatment of GERD, the fundamental concept is appropriate diagnosis within the clinical spectrum of this condition. Patients can benefit from different therapeutic modalities applied objectively and tailored to each patient.
Funding
The author declares that no funding was received for this study.
Conflicts of interest
The author declares no conflicts of interest.
Ethical considerations
Protection of human and animal subjects. The author declares that no experiments were performed on humans or animals for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve patient personal data or require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. The author declares that no generative artificial intelligence was used in writing this manuscript.
References
1. Richter JE, Rubenstein JH. Presentation and epidemiology of gastroesophageal reflux disease. Gastroenterology. 2018;154:267-76.
2. Valdovinos Díaz MA, Amieva-Balmori M, Carmona-Sánchez R, Coss-Adame E, Gómez-Escudero O, González-Martínez M, et al. Good clinical practice recommendations for the diagnosis and treatment of gastroesophageal reflux disease. An expert review from the Asociacion Mexicana de Gastroenterologia. Rev Gastroenterol Mex (Engl Ed). 2024;89:121-43.
3. Vakil N, van Zanten SV, Kahrilas P, Dent J, Jones R;Global Consensus Group. The Montreal definition and classification of gastroesophageal reflux disease:a global evidence-based consensus. Am J Gastroenterol. 2006;101:1900-20;quiz 1943.
4. Fass R, Boeckxstaens GE, El-Serag H, Rosen R, Sifrim D, Vaezi MF. Gastro-oesophageal reflux disease. Nat Rev Dis Primers 2021;7:55.
5. Yamasaki T, Fass R. Reflux hypersensitivity:a new functional esophageal disorder. J Neurogastroenterol Motil. 2017;23:495-503.
6. Savarino E, Zentilin P, Savarino V. NERD:an umbrella term including heterogeneous subpopulations. Nat Rev Gastroenterol Hepatol. 2013;10:371-80.
7. Savarino V, Marabotto E, Zentilin P, Demarzo MG, Pellegatta G, Frazzoni M, et al. Esophageal reflux hypersensitivity:non-GERD or still GERD?Dig Liver Dis. 2020;52:1413-20.
8. Dent J. Endoscopic grading of reflux oesophagitis:the past, present and future. Best Pract Res Clin Gastroenterol. 2008;22:585-99.
9. Armstrong D, Bennett JR, Blum AL, Dent J, De Dombal FT, Galmiche JP, et al. The endoscopic assessment of esophagitis:a progress report on observer agreement. Gastroenterology. 1996;111:85-92.
10. Lundell LR, Dent J, Bennett JR, Blum AL, Armstrong D, Galmiche JP, et al. Endoscopic assessment of oesophagitis:clinical and functional correlates and further validation of the Los Angeles classification. Gut. 1999;45:172-80.
11. Gyawali CP, Yadlapati R, Fass R, Katzka D, Pandolfino J, Savarino E, et al. Updates to the modern diagnosis of GERD:Lyon consensus 2.0. Gut. 2024;73:361-71.
12. Mizuno H, Matsuhashi N, Sakaguchi M, Inoue S, Nakada K, Higuchi K, et al. Recent effectiveness of proton pump inhibitors for severe reflux esophagitis:the first multicenter prospective study in Japan. J Clin Biochem Nutr. 2015;57:233-8.
13. Armstrong D. Systematic review:persistence and severity in gastro-oesophageal reflux disease. Aliment Pharmacol Ther. 2008;28:841-53.
14. Mizuno H, Nishino M, Yamada K, Kamiyamamoto S, Hinoue Y. Efficacy of Vonoprazan for 48-Week Maintenance Therapy of Patients with Healed Reflux Esophagitis. Digestion. 2020;101:411-21.
15. ASGE Standards of Practice Committee;Qumseya B, Sultan S, Bain P, Jamil L, Jacobson B, Anandasabapathy S, et al. ASGE guideline on screening and surveillance of Barrett's esophagus. Gastrointest Endosc. 2019;90:335-59e2.
16. Standards of Practice Committee;Wani S, Qumseya B, Sultan S, Agrawal D, Chandrasekhara V, Harnke B, et al. Endoscopic eradication therapy for patients with Barrett's esophagus-associated dysplasia and intramucosal cancer. Gastrointest Endosc. 2018;87:907-31e9.
17. Ward EM, Wolfsen HC, Achem SR, Loeb DS, Krishna M, Hemminger LL, et al. Barrett's esophagus is common in older men and women undergoing screening colonoscopy regardless of reflux symptoms. Am J Gastroenterol. 2006;101:12-7.
18. Keyashian K, Hua V, Narsinh K, Kline M, Chandrasoma PT, Kim JJ. Barrett's esophagus in Latinos undergoing endoscopy for gastroesophageal reflux disease symptoms. Dis Esophagus. 2013;26:44-9.
19. Spechler SJ, Souza RF. Barrett's esophagus. N Engl J Med. 2014;371:836-45.
20. Qumseya BJ, Bukannan A, Gendy S, Ahemd Y, Sultan S, Bain P, et al. Systematic review and meta-analysis of prevalence and risk factors for Barrett›s esophagus. Gastrointest Endosc. 2019;90:707-17e1.
21. Sikkema M, de Jonge PJ, Steyerberg EW, Kuipers EJ. Risk of esophageal adenocarcinoma and mortality in patients with Barrett›s esophagus:a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2010;8:235-44;quiz e32.
22. Yousef F, Cardwell C, Cantwell MM, Galway K, Johnston BT, Murray L. The incidence of esophageal cancer and high-grade dysplasia in Barrett›s esophagus:a systematic review and meta-analysis. Am J Epidemiol. 2008;168:237-49.
23. Singh S, Manickam P, Amin AV, Samala N, Schouten LJ, Iyer PG, et al. Incidence of esophageal adenocarcinoma in Barrett's esophagus with low-grade dysplasia:a systematic review and meta-analysis. Gastrointest Endosc. 2014;79:897-909.e4;quiz 983.e1, 983.e3.
24. Wani S, Falk GW, Post J, Yerian L, Hall M, Wang A, et al. Risk factors for progression of low-grade dysplasia in patients with Barrett's esophagus. Gastroenterology. 2011;141:1179-86, 1186.e1.
25. Krishnamoorthi R, Mohan BP, Jayaraj M, Wang KK, Katzka DA, Ross A, et al. Risk of progression in Barrett›s esophagus indefinite for dysplasia:a systematic review and meta-analysis. Gastrointest Endosc. 2020;91:3-10.e3.
26. Sharma P, Dent J, Armstrong D, Bergman JJ, Gossner L, Hoshihara Y, et al. The development and validation of an endoscopic grading system for Barrett's esophagus:the Prague C &M criteria. Gastroenterology. 2006;131:1392-9.
27. Shaheen NJ, Falk GW, Iyer PG, Souza RF, Yadlapati RH, Sauer BG, et al. Diagnosis and Management of Barrett›s Esophagus:An Updated ACG Guideline. Am J Gastroenterol. 2022;117:559-87.
28. Levine DS, Haggitt RC, Blount PL, Rabinovitch PS, Rusch VW, Reid BJ. An endoscopic biopsy protocol can differentiate high-grade dysplasia from early adenocarcinoma in Barrett's esophagus. Gastroenterology. 1993;105:40-50.
29. Januszewicz W, Fitzgerald RC. Barrett's oesophagus and oesophageal adenocarcinoma. Medicine (Abingdon). 2019;47:275-85.
30. Abela JE, Going JJ, Mackenzie JF, McKernan M, O›Mahoney S, Stuart RC. Systematic four-quadrant biopsy detects Barrett›s dysplasia in more patients than nonsystematic biopsy. Am J Gastroenterol. 2008;103:850-5.
31. Muthusamy VR, Wani S, Gyawali CP, Komanduri S;CGIT Barrett's Esophagus Consensus Conference Participants. AGA Clinical Practice Update on New Technology and Innovation for Surveillance and Screening in Barrett's Esophagus:Expert Review. Clin Gastroenterol Hepatol. 2022;20:2696-706.e1.
32. Zerbib F, Bredenoord AJ, Fass R, Kahrilas PJ, Roman S, Savarino E, et al. ESNM/ANMS consensus paper:Diagnosis and management of refractory gastro-esophageal reflux disease. Neurogastroenterol Motil. 2021;33:e14075.
33. Fass R, Sifrim D. Management of heartburn not responding to proton pump inhibitors. Gut. 2009;58:295-309.
34. Patel D, Fass R, Vaezi M. Untangling nonerosive reflux disease from functional heartburn. Clin Gastroenterol Hepatol. 2021;19:1314-26.
35. Davis TA, Gyawali CP. Refractory gastroesophageal reflux disease:diagnosis and management. J Neurogastroenterol Motil. 2024;30:17-28.
36. Yadlapati R, Vaezi MF, Vela MF, Spechler SJ, Shaheen NJ, Richter J, et al. Management options for patients with GERD and persistent symptoms on proton pump inhibitors:recommendations from an expert panel. Am J Gastroenterol. 2018;113:980-6.
37. Gyawali CP, Tutuian R, Zerbib F, Rogers BD, Frazzoni M, Roman S, et al. Value of pH impedance monitoring while on twice-daily proton pump inhibitor therapy to identify need for escalation of reflux management. Gastroenterology. 2021;161:1412-22.
38. Eusebi LH, Ratnakumaran R, Yuan Y, Solaymani-Dodaran M, Bazzoli F, Ford AC. Global prevalence of, and risk factors for, gastro-oesophageal reflux symptoms:a meta-analysis. Gut. 2018;67:430-40.
39. Sadafi S, Azizi A, Pasdar Y, Shakiba E, Darbandi M. Risk factors for gastroesophageal reflux disease:a population-based study. BMC Gastroenterol. 2024;24:64.
40. Spechler SJ, Laine L, DeVault KR, Nabulsi A, Hunt B, Katz P. Comparison of Los Angeles grades of erosive esophagitis scored by local investigators vs central adjudicators in a clinical trial. Clin Gastroenterol Hepatol. 2024;22:2526-8.e1.
41. Naini BV, Souza RF, Odze RD. Barrett's esophagus:a comprehensive and contemporary review for pathologists. Am J Surg Pathol. 2016;40:e45-66.
