Introduction
Gastroesophageal reflux disease (GERD) is one of the most frequent digestive diseases, with a clinical spectrum ranging from heartburn and regurgitation to structural and functional complications of the esophagus and other organs. It affects 10-20% of the population and transcends the digestive system, being associated with clinically relevant esophageal and extra-esophageal complications. Among the former, peptic stricture and Barrett’s esophagus stand out. Respiratory manifestations range from mild, chronic symptoms (cough, dysphonia) to complex conditions such as chronic lung allograft dysfunction (CLAD), bronchiolitis obliterans syndrome (BOS), and aspiration pneumonia. This chapter addresses the pathophysiology, diagnosis, treatment, and follow-up of the main esophageal and respiratory complications: peptic stricture, Barrett’s esophagus, reflux in lung transplantation, aspiration pneumonia, idiopathic pulmonary fibrosis, and refractory asthma. The Lyon 2.0 consensus updates the criteria for a conclusive diagnosis by endoscopy and 24-hour pH-metry: acid exposure time (AET) > 6%, Los Angeles B, C, or D esophagitis, and peptic stricture or Barrett’s esophagus, and proposes the “actionable GERD” approach, which advocates intervention when the presence of GERD and its consequences are objectively demonstrated and also explain the symptoms. Evidence against GERD includes AET < 4% and absence of endoscopic findings, and supportive criteria1. The American College of Gastroenterology (ACG) 2022 and American Gastroenterological Association (AGA) 2023 guidelines emphasize performing objective tests before making therapeutic decisions1–3. It should be considered that, although the existence of pathological reflux is demonstrated through endoscopic findings or 24-hour pH-impedance monitoring, including recording of reflux with proximal extension, these results are not conclusive for establishing a causal relationship with respiratory symptoms. Therefore, interpretation must be made with caution and always in the comprehensive clinical context of the patient, so that clinical judgment prevails over findings from different studies.
Methods (search and criteria)
A targeted search was conducted in PubMed (2015 to 2024) combining the following MeSH terms: “Gastroesophageal Reflux”, “Peptic Stricture”; “Barrett’s Esophagus”; “Extra-esophageal Complications”; “Lung Transplantation”, “Aspiration Pneumonia”, “Idiopathic Pulmonary Fibrosis”, “Refractory Asthma”, “Esophageal pH Monitoring”, “High-Resolution Manometry”, “Antireflux Surgery”, and “Radiofrequency Ablation”. AGA/ACG guidelines and the Lyon 2.0 consensus, systematic reviews, meta-analyses, and cohort studies were prioritized; additionally, narrative reviews and contemporary series focused on lung transplantation and antireflux surgery.
Peptic stricture
Definition and epidemiology
Peptic stricture is an acquired narrowing of the distal esophagus secondary to chronic inflammation from acid exposure. It is the most common cause of benign esophageal stricture, although its prevalence has decreased with the use of proton pump inhibitors (PPIs). Chronic, untreated GERD leads to inflammation, submucosal fibrosis, and the formation of scar tissue in the esophageal wall.
Manifestations
Clinical features suggesting a GERD-related stricture include typical reflux symptoms accompanied by alarm features, such as dysphagia and significant progressive weight loss. Sometimes, patients may deny the previous existence of heartburn and regurgitation.
Diagnosis
The approach begins with endoscopy to classify the type of stricture and rule out malignancy. There are two relevant types of strictures: simple ones, which are those with a short length (< 2 cm), a straight course, without angulations and typically located in the distal third of the esophagus, and complex ones, which are long (> 2 cm), irregular, angulated, and tend to recur. Simple strictures have a better response to treatment. During endoscopy, it is important to consider obtaining tissue in case of suspected malignancy or to rule out any alternative diagnosis, such as eosinophilic esophagitis. If Barrett’s esophagus is suspected in the presence of esophagitis, it is suggested to defer biopsy sampling to another time4.
Treatment
The management of benign strictures secondary to GERD is based primarily on endoscopic dilation accompanied by intensive antisecretory treatment with PPIs or potassium-competitive acid blockers (P-CAB), which has been shown to reduce the number of sessions required and prolong the symptom-free interval. Esophagectomy is reserved for severe, refractory cases or those with chronic fistulas. In addition, there are other emerging therapeutic modalities that will be discussed at the end of this section4.
ENDOSCOPIC DILATION
As already mentioned, endoscopic dilation is the first-line treatment for symptomatic strictures. Different dilators (Savary, Maloney) or balloons through the endoscope channel can be used, depending on the type of stricture and the endoscopist’s experience, as no significant differences in efficacy have been demonstrated5.
Dilators are cylindrical instruments (usually flexible) that achieve mechanical dilation with radial and longitudinal force when introduced blindly over a guidewire (Savary-Gilliard) to cross the stricture. The Maloney-Hurst dilator, not being guided, has a higher risk of perforation, so it is reserved for selected cases6. Fluoroscopic visualization is preferred in complex strictures that have not allowed passage of the endoscope, to visualize the degree of dilation and allow immediate diagnosis of perforations. To prevent perforations, the “rule of 3” is applied, which limits dilation to no more than 3 mm per session, but this practice may not affect reducing the perforation rate4,6,7. On the other hand, balloons offer the advantage of being introduced through the working channel of endoscopes and allow direct visualization, so they are preferred in angulated or very tight strictures; they can also be used under fluoroscopic guidance4 (Fig. 1). In general, a peptic esophageal stricture may require several dilation sessions, depending on its complexity and etiology. Most simple benign strictures respond in one to three sessions, scheduled every 2 weeks. A peptic stricture is considered refractory when a diameter ≥ 14 mm is not achieved after five or more sessions scheduled every 2 weeks4,6.
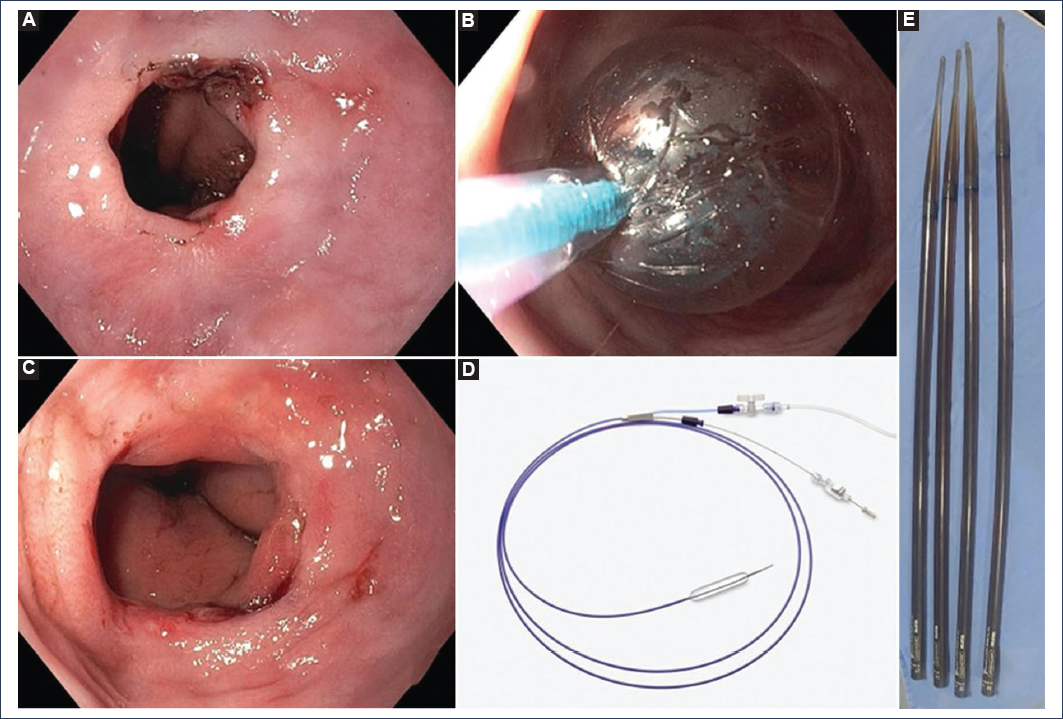
Figure 1. Esophageal stricture of peptic origin. A: endoscopic image of a peptic stricture with ulcerated border, associated with a hiatal hernia. B: dilation of the stricture with a hydrostatic balloon. C: appearance of the stricture at 2 weeks after the second balloon dilation session with an acid inhibitor. D and E: hydrostatic balloon and Savary-Gilliard dilators.
Complications of endoscopic treatment of strictures are mainly perforation, with a risk of 0.1% to 0.4%, which is higher when a bougie-type dilator is used, in complex and post-radiation strictures. This complication must be treated immediately with clips or surgery. Bleeding is usually uncommon and generally self-limited. Rarely, mild chest pain may occur. It is recommended to assess the risk of bleeding, monitor the patient for at least 2 hours after the procedure, and watch for warning signs such as fever, persistent chest pain, or severe odynophagia6.
INTRALESIONAL STEROIDS
For the treatment of refractory peptic strictures, intralesional steroids may be injected to promote tissue remodeling. In a systematic review that included four clinical trials, two cohorts, and six case series with 341 patients, it was described that steroid injections added to dilation reduced the need for further dilations in peptic, radiation, and caustic strictures, without consistent effects in anastomotic strictures. Data were insufficient for eosinophilic esophagitis, sclerotherapy, and drug-induced strictures. Few non-severe complications were reported, including esophageal candidiasis. The most used steroid was triamcinolone at doses of 20 to 80 mg injected in the four quadrants of the esophageal wall before or after dilation. Although currently there is no consensus on the dose, timing, or optimal frequency of injection, a maximum of three sessions with 20 mg of triamcinolone in each quadrant before dilation is generally recommended. Specifically for peptic stricture, solid evidence was found in favor of intralesional steroid use in refractory or recurrent cases, limited evidence in radiation, caustic, and anastomotic strictures, and lack of evidence in eosinophilic esophagitis strictures, so it is not recommended. These data have the limitation of high heterogeneity, limited sample size, and the need for randomized controlled trials5.
ESOPHAGEAL STENTS
Another option is the use of fully covered metallic stents, which manage to keep the lumen open continuously to induce remodeling. Success rates up to 45% have been reported, but with a significant migration percentage of 25% to 35% and adverse effects of 20% to 25%. There is another type of biodegradable stent, but they have lower radial force and require additional dilation4.
Currently, new modalities are available for the treatment of benign esophageal strictures, such as the dilating cap, endoscopic incisions, and lumen-apposing metal stents.
The dilating cap is a cone-shaped device that is placed at the tip of the endoscope and allows dilation under direct vision; an effectiveness of 96% has been reported. Incisions are reserved for short strictures or webs; they consist of making radial cuts with a needle or scissors to subsequently perform balloon dilation. Lumen-apposing metal stents have proven useful for short strictures (< 1 cm), as they keep the lumen open, but have a high migration rate of up to 40%4.
Barrett’s esophagus
Definition and epidemiology
Barrett’s esophagus is a columnar metaplasia that replaces the normal squamous epithelium, due to chronic acid exposure, affecting approximately 5% of the population in the United States and 1% of the world population. It is considered a premalignant lesion, with a risk of developing esophageal adenocarcinoma between 0.12% and 0.5% annually8.
The main risk factors for developing Barrett’s esophagus are recognized to be chronic GERD, male sex, age over 50 years, smoking, obesity, white race, and a diet rich in meats and processed foods8.
Diagnosis
The diagnosis of Barrett’s esophagus is made by endoscopy with the finding of columnar mucosa (intestinal metaplasia) in the distal esophagus extending beyond 1 cm from the termination of the gastric folds, confirmed by histopathology. Biopsy sampling in lengths < 1 cm is discouraged because the cancer risk is extremely low (0.007% in follow-up > 4 years)8,9. During endoscopy, the circumferential length and the longest segment of metaplasia should be measured in a standardized manner using the Prague criteria, which allows classification as short-segment Barrett’s esophagus when the extension is < 3 cm and long-segment when it is ≥ 3 cm. The importance of this classification lies in the fact that long-segment Barrett’s esophagus has a higher risk of dysplasia and cancer, but the risk of progression to adenocarcinoma also depends on the presence and grade of dysplasia. In the absence of dysplasia, the risk is 0.12% to 0.5% annually, for low-grade dysplasia the risk increases to 0.5% to 0.7% annually, and if high-grade dysplasia is present, the risk ranges from 5% to 7% annually8. Follow-up recommendations are based on the histopathological result and are summarized in Table 1.
Table 1. Recommendations for Barrett’s esophagus follow-up according to histopathological result
| Guideline | Negative dysplasia | Indeterminate result | Low-grade dysplasia | High-grade dysplasia |
|---|---|---|---|---|
| AGA, 2011 | Every 3-5 years | – | Every 6-12 months | Eradication treatment + follow-up every 3 months |
| ASGE, 2012 | Every 3-5 years | Endoscopy after acid suppression | Repeat at 6 months, then annually | Consider treatment; if not a candidate, follow up every 3 months |
| BSG, 2014 | < 3 cm: no surveillance 3 cm: every 2-3 years | Repeat at 6 months | Every 6 months | Endoscopic treatment |
| ACG, 2016 | Every 3-5 years | Repeat at 3-6 months | Every 12 months | Endoscopic treatment |
| AMEG, 2017 | < 3 cm: every 3-5 years 3 cm: every 2-3 years | Acid suppression for 3 months and repeat endoscopy | Every 6-12 months | Endoscopic treatment |
| ESGE, 2017 | 1-3 cm: every 5 years 3-10 cm: every 3 years 10 cm: refer to expert | Repeat at 6 months | Repeat at 6 months; if it persists, ablation | Ablation. If there is a lesion: EMR; if no dysplasia: repeat in 3 months |
|
ACG: American College of Gastroenterology; AGA: American Gastroenterological Association; ASGE: American Society for Gastrointestinal Endoscopy; BSG: British Society of Gastroenterology; ESGE: European Society of Gastrointestinal Endoscopy; EMR: endoscopic mucosal resection. |
||||
Regarding biopsy sampling, the Seattle protocol is traditionally used, which consists of taking biopsies from the four quadrants every 1-2 cm of the circumferential length of the mucosa with Barrett’s esophagus, and one biopsy per centimeter in the tongues9,10. Detection of dysplasia depends on endoscopic surveillance, sampling, inter-pathologist variability, and the patchy distribution of dysplasia. It has been reported that up to 75% of initial diagnoses of low-grade dysplasia can be reclassified as absence of dysplasia when another expert pathologist reviews them, and that in 50% of new cases of adenocarcinoma, there was no previous diagnosis of Barrett’s esophagus10.
Treatment
The standard management of Barrett’s esophagus includes medical treatment and endoscopic treatment, with endoscopic surveillance. The basis of medical treatment is acid suppression, and although aspirin has been shown to have a role in chemoprevention, it is not systematically recommended due to its adverse effects. Endoscopic surveillance for mucosal evaluation with biopsy sampling in short-segment Barrett’s esophagus should be performed every 5 years, while for long-segment the time is shortened to every 3 years. When patients present low-grade dysplasia, it is indicated every 6 to 12 months, and in case of high-grade dysplasia, immediate endoscopic treatment must be performed8. It is important to note that the detection of dysplasia depends on the quality of the endoscopic procedure, so the use of high-definition white light, proper cleaning, and adequate inspection time should be emphasized. Neoplasia detection rates increase with inspections of at least 1 minute per centimeter of Barrett’s esophagus. To increase diagnostic yield, new image-enhancement technologies that allow targeted biopsy sampling can be used, such as NBI (narrow band imaging), LCI (linked color imaging), or artificial intelligence tools in endoscopy11.
Endoscopic treatment includes resection of visible lesions, either by endoscopic mucosal resection or by endoscopic submucosal dissection, as well as radiofrequency ablation to eradicate the entire surface of residual metaplasia.
Alternative techniques exist, such as cryotherapy and argon plasma coagulation, for the eradication of residual metaplasia8.
Radiofrequency ablation has proven effective in eradicating Barrett’s esophagus with or without dysplasia. In the case of high-grade dysplasia, an eradication rate of 81% has been demonstrated, compared with 19% in the control group12. Similarly, in low-grade dysplasia, progression after radiofrequency was reported in 15%, compared with 26% with surveillance alone13. Although ablation can eradicate Barrett’s esophagus without dysplasia, this approach has not been shown to reduce mortality, and in this scenario, it is recommended to identify patients at higher risk of progressing to benefit from ablation (e.g., with risk factors or p53 gene mutations)10.
Gastroesophageal reflux disease in lung transplantation
Epidemiology and relevance
The prevalence of pathological reflux (acid and non-acid) after lung transplantation ranges between 50% and 70%, with a high frequency of esophagogastric dysmotility14–16. Microaspirations contribute to airway inflammation and are associated with CLAD and BOS. Biomarkers such as pepsin and bile acids in bronchoalveolar lavage support the occurrence of gastric microaspirations15.
Pathophysiology
Contributing factors include anatomical changes, denervation, alteration of lower esophageal sphincter tone, esophageal hypomotility, gastroparesis, and alteration of post-surgical esophageal clearance. Non-acid postprandial reflux and proximal exposure can be particularly harmful to the graft14–16.
Diagnosis
The Lyon 2.0 consensus defines the conclusive criteria for “actionable GERD” by endoscopy and 24-hour pH-metry: AET > 6%, Los Angeles B, C, or D esophagitis, and stricture or Barrett’s esophagus1. Supporting evidence includes > 80 reflux episodes in 24 hours, hiatal hernia, and positive symptomatic association with a symptom index (SI) > 50% or a symptom association probability (SAP) > 95%. High-resolution manometry identifies hypomotility or aperistalsis and guides the feasibility of fundoplication. Evidence against GERD consists of AET < 4% and absence of endoscopic findings1–3,14–16.
Treatment
In recent clinical series, early anti-reflux surgery (fundoplication) is associated with lower incidence of CLAD and BOS, and with better preservation of graft function when performed before establishment of CLAD17,18. However, evidence derives from cohorts and meta-analyses with heterogeneity. Patient selection requires objectively demonstrated reflux, clinico-physiological correlation, and multidisciplinary assessment. PPIs reduce acidity but do not prevent non-acid reflux or aspiration. Optimization of gastric emptying with the use of prokinetics, postural measures, and eating hygiene (small and frequent meals, avoid lying down before 3 hours after eating) are adjuvants2,3,15–18.
Aspiration pneumonia
Epidemiology and clinical features
Aspiration pneumonia represents 5-15% of community-acquired pneumonias and affects patients with neurological disorders, oropharyngeal dysphagia, or severe GERD.
Clinically it may present with chronic cough, dyspnea, wheezing, or chemical pneumonitis. Typical radiological findings include consolidations in susceptible lobes, especially the right lower lobe19.
Pathophysiology and biomarkers
Microaspirations of acid, pepsin, and bile cause epithelial injury and inflammation. The finding of pepsin or bile acids in bronchoalveolar lavage supports the gastric origin15,19. pH-metry with impedance allows correlation of reflux events (acid, mixed, or non-acid) with cough or desaturation19. One study has shown that genetic predisposition to GERD is associated with increased risk of chronic obstructive pulmonary disease (COPD) and pneumonia20.
Diagnosis and treatment
It is recommended to document pathological GERD (Lyon criteria) before proposing anti-reflux surgery. Esophageal and gastric imaging and emptying studies are supportive. Use of double-dose PPIs, swallowing therapy, and postural measures constitute the first line of treatment. In children with neurological damage, fundoplication reduces, but does not eliminate, the risk of aspiration pneumonia (odds ratio [OR]: ≈ 0.44)21. In adults, surgery should be individualized after demonstrating relevant reflux (AET > 6%)1–3,19,21.
Idiopathic pulmonary fibrosis
Definition and disease burden
Idiopathic pulmonary fibrosis is a chronic fibrosing interstitial pneumonia of unknown cause, limited to the lung and characterized by a radiological/histological pattern of usual interstitial pneumonia. It has a progressive course, with a median survival of less than 3 years and a worse prognosis in males and at advanced ages, with accelerated loss of pulmonary function. Risk factors include smoking, environmental exposures, genetic predisposition, and GERD22,23.
Association with GERD and proposed pathophysiological mechanisms
Various studies have shown a high prevalence of GERD in idiopathic pulmonary fibrosis, even in patients without digestive symptoms, with predominance of non-acid, proximal reflux, and presence of hiatal hernia. Chronic microaspiration of acid, pepsin, and bile can induce epithelial injury and profibrotic signaling (transforming growth factor beta, nuclear factor kappa B, farnesoid X receptor), demonstrated in animal models and in bronchoalveolar lavage. Bidirectional mechanics is also relevant: low pulmonary compliance favors transient lower esophageal sphincter relaxations and proximal reflux. Although a role for Helicobacter pylori has been proposed, the evidence is inconsistent. Genetic data suggest an effect of GERD on idiopathic pulmonary fibrosis risk, without evidence of inverse causality22–24.
Diagnosis
Application of the Lyon 2.0 criteria is recommended to avoid overtreatment. pH-metry with impedance should be performed with usual meals and analysis of total episodes, proximal exposure, and symptomatic association (SI/SAP). An AET > 6% is conclusive of GERD, an AET of 4-6% is considered a gray zone (supported with > 80 reflux episodes, endoscopic findings, and positive SI/SAP), and an AET < 4% is against GERD. However, as already mentioned, it must be remembered that a pH-metry positive for GERD does not necessarily explain respiratory symptoms, even with evidence of proximal extension, so it must be integrated with clinical features, functionality, and multidisciplinary discussion1,22–24.
Treatment
Treatment with PPIs offers contradictory results and carries the risk of respiratory infections. Anti-reflux surgery can be considered in cases with documented aspiration and demonstrated pathological reflux. P-CABs lack specific evidence, although they are promising due to their sustained acid suppression1–3,23,24.
Refractory asthma
Epidemiology
Population cohorts have shown a bidirectional association between GERD and asthma: GERD increases the risk of asthma (OR: ≈1.46) and asthma increases the risk of GERD (OR: ≈1.36)25. Systematic reviews highlight coexistence with difficult-to-control asthma and COPD26,27.
Mechanisms
There are two main pathways: 1) microaspiration of gastric contents with airway inflammation and 2) esophago-bronchial vagal reflexes that induce bronchoconstriction. Nocturnal and postprandial worsening supports a reflux-related component. Esophageal hypersensitivity may contribute to chronic cough26,27.
Diagnosis and treatment
In refractory asthma, causality should not be assumed; documentation of pathological reflux or symptomatic association (SI > 50%, SAP > 95%) on pH-metry with impedance is required. PPIs show modest benefit in some patients and do not substitute for standard asthma treatment. Anti-reflux surgery is exceptional and is reserved for patients with proven GERD and with a highly probable clinical association1–3,26,27.
Discussion
Complications of GERD comprise a spectrum ranging from structural esophageal lesions, such as peptic stricture and Barrett’s esophagus, to extra-esophageal respiratory manifestations. All require a comprehensive diagnostic evaluation based on endoscopy, esophageal physiology tests, and objective documentation of pathological reflux by pH-metry with impedance and high-resolution manometry.
Peptic stricture is a sequela of chronic, untreated acid reflux that induces inflammation, fibrosis, and scar tissue formation. Its approach should focus on progressive endoscopic dilation, supported by intensive antisecretory therapy with PPIs or P-CABs, to reduce recurrences and favor healing. In refractory cases, intralesional steroid injections or fully covered stents may be considered, while esophagectomy is reserved for complex or recurrent strictures.
Barrett’s esophagus is an acquired intestinal metaplasia secondary to chronic acid exposure, with a risk of progression to adenocarcinoma estimated between 0.12% and 0.5% annually. Systematic endoscopic surveillance (Seattle protocol), use of high-definition endoscopy, and ablation therapies – especially radiofrequency – have modified the prognosis, allowing effective eradication of dysplasia and reduction of progression risk. Integration of biomarkers and artificial intelligence tools promises to optimize detection and characterization of dysplastic lesions in the future.
Regarding extra-esophageal respiratory manifestations, evidence indicates that GERD contributes to the pathogenesis of disorders such as CLAD and BOS following lung transplantation, aspiration pneumonia, idiopathic pulmonary fibrosis, and refractory asthma. However, direct causality is only established when pathological reflux is objectively demonstrated, and there is a temporal correlation with symptoms. Table 2 shows the level of evidence for the association of GERD with respiratory complications.
Table 2. Summary of clinical evidence for respiratory complications
| Complication | Clinical evidence | Level of certainty |
|---|---|---|
| Lung transplant | Association with CLAD and BOS; ARS may improve FEV1 and delay graft dysfunction | Moderate |
| Aspiration pneumonia | Higher risk in frail patients with dysphagia; prevention with PPI or ARS and rehabilitation | High |
| Pulmonary fibrosis | Observational association, without confirmed causality; the benefit of antacids is uncertain | Low |
| Refractory asthma | Frequent association, but trials with PPIs show variable results | Low |
|
ARS: antireflux surgery; BOS: bronchiolitis obliterans; CLAD: chronic lung allograft dysfunction; FEV1: forced expiratory volume in the first second; PPI: proton pump inhibitors. |
||
The Lyon 2.0 consensus proposes the concept of “actionable GERD,” focused on integrating objective metrics (AET, number of reflux episodes, symptomatic association, motility, and therapeutic response) before indicating treatment. Early anti-reflux surgery shows the most consistent benefit in the context of lung transplantation, by reducing progression to CLAD or BOS. In idiopathic pulmonary fibrosis and refractory asthma, the relationship is less robust, and indiscriminate use of PPIs should be avoided. A multidisciplinary approach and precision medicine, based on physiology and biomarkers, are recommended, reserving surgery for patients with a demonstrated correlation between reflux and symptoms.
Conclusions
- ‒ Esophageal complications (peptic stricture and Barrett’s esophagus) and respiratory complications are part of the clinical spectrum of GERD, and their management requires a comprehensive approach based on physiology and effective acid control.
- ‒ In peptic stricture, endoscopic dilation associated with potent antisecretion is the pillar of treatment; in refractory cases, intralesional steroids or stents may be used.
- ‒ In Barrett’s esophagus, the combination of structured endoscopic surveillance and radiofrequency ablation reduces progression to dysplasia or cancer.
- ‒ Not all respiratory manifestations are due to GERD; causality must be confirmed with objective metrics (pH-metry with impedance and manometry).
- ‒ In lung transplantation, early evaluation should be performed and fundoplication considered in selected cases.
- ‒ In aspiration pneumonia, swallowing rehabilitation, postural measures, and PPIs will be prioritized; surgery reduces risk but does not eliminate it.
- ‒ In idiopathic pulmonary fibrosis and refractory asthma, evidence suggests an association, but therapeutic benefit is variable; management should be individualized and overtreatment should be avoided.
- ‒ Lyon 2.0, AGA 2023, and ACG 2022 guidelines should be applied, promoting collaboration between gastroenterologists, pulmonologists, and thoracic surgeons.
- ‒ PPIs remain the main antisecretory drugs in these GERD complication scenarios, but P-CABs are a promising alternative for acid control in refractory cases and should be evaluated in future studies.
Acknowledgments
The authors thank the invitation from the Mexican Association of Gastroenterology and the president in office, Dr. J.M. Remes-Troche.
Funding
The authors declare that they have not received funding for this study.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical considerations
Protection of people and animals. The authors declare that no experiments on humans or animals have been performed for this research.
Confidentiality, informed consent, and ethical approval. The study does not involve personal patient data or require ethical approval. SAGER guidelines do not apply.
Statement on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for writing this manuscript.
