Introduction
GERD is characterized by the recurrent presence of heartburn and regurgitation, considered bothersome by the patient, as well as by the appearance of specific complications of the disease. When diagnostic studies (endoscopy or pH monitoring) have not been performed to confirm it, it is defined as unproven GERD. In contrast, if there are findings in studies, such as positive endoscopy or pH monitoring, the diagnosis of proven GERD is established. This condition affects approximately 20% of the adult population in high-income countries1. GERD can significantly deteriorate health-related quality of life and has been associated with an increased risk of esophagitis, esophageal stricture, Barrett’s esophagus, and esophageal adenocarcinoma.
From a clinical standpoint, symptoms are grouped into two main categories: typical (esophageal) and atypical (extraesophageal). In many cases, the presence of typical symptoms allows for a presumptive diagnosis of the disease. Patients with classic GERD symptomatology can initiate empirical treatment with proton pump inhibitors (PPIs) or potassium-competitive acid blockers (P-CABs), as long as they do not present alarm features. In those cases where an adequate therapeutic response is not observed (decrease in symptomatology, less than 50% in 2 weeks) or when invasive maneuvers are planned for treatment, it is recommended to perform complementary studies such as upper digestive endoscopy, esophageal manometry, and ambulatory monitoring of esophageal pH.
Although GERD has traditionally been considered a disease caused by gastric acid, up to 50% of patients do not experience significant improvement with pharmacological suppression of acid production2. Those with symptoms suggestive of GERD accompanied by alarm signs that could indicate the appearance of complications or disease, in the presence of symptoms such as progressive dysphagia, unexplained weight loss, or digestive bleeding, as well as patients with relevant risk factors for esophageal adenocarcinoma, such as advanced age, male sex, and obesity, should undergo endoscopic evaluation1. It is essential to understand that GERD can no longer be approached as a single clinical condition, but as a spectrum of phenotypes with different pathophysiological mechanisms and specific diagnostic requirements3.
Methods
This chapter presents a narrative review of the literature on the diagnosis of gastroesophageal reflux disease (GERD). A structured search was conducted in the PubMed database using the following MeSH descriptors: “gastroesophageal reflux”, “esophageal pH monitoring” or “esophageal pH-metry”, “esophageal manometry” and “GERD diagnosis”. Articles published in the last 10 years in English or Spanish were selected. Additionally, literature before 2015 considered of interest in the diagnosis of GERD was integrated. The objective was to prepare a critical, structured, and updated review of the diagnosis of GERD, which integrates recent advances and serves as a basis for better clinical understanding.
Typical symptoms
GERD manifests with a variety of symptoms that can be classified as typical and atypical. Typical symptoms are directly esophageal and include heartburn, defined as a burning sensation below the sternum that travels from the epigastrium to the neck; esophageal chest pain, characterized as retrosternal chest pain clinically indistinguishable from a cardiac etiology (this symptom always warrants cardiology evaluation, and after ruling out a cardiac etiology it can be attributed to an esophageal etiology); and finally regurgitation, understood as the non-forced return of gastric contents to the esophagus that can sometimes reach the mouth4.
Atypical symptoms
Atypical symptoms are less related to the direct pathophysiology of GERD and are extraesophageal. Among these atypical symptoms are those with established association, such as chronic cough and asthma, which are attributed to a bronchial hyperreactive response, in which reflux is a trigger. On some occasions, aspiration of reflux into the airways is associated with diseases such as idiopathic pulmonary fibrosis; it should be noted that the latter does not have an established association4. Another symptom with an established association is laryngitis, and in some cases, dental erosion and cavities may also occur due to repeated exposure to acid, causing damage to tooth enamel5.
Among extraesophageal manifestations without an established association with GERD is pharyngitis3. Similarly, patients may present with globus pharyngeus sensation, also known as globus, commonly described as a lump or tightness in the throat, which does not have an established association with GERD4. Additionally, patients may report burning mouth syndrome, a little-known condition consisting of a persistent burning sensation in the mouth that can affect the tongue, gums, lips, palate, and cheeks6.
The San Diego Consensus recognizes that extraesophageal symptoms of GERD comprise a heterogeneous spectrum of respiratory, otolaryngological, and oral manifestations. These symptoms are divided into two broad spectra: those with consistent evidence and others with weaker or inconclusive associations. Among symptoms with consistent evidence of relationship are chronic cough, uncontrolled asthma, posterior laryngitis, and dental erosion7.
On the other hand, conditions such as chronic pharyngitis, recurrent sinusitis, otitis media, idiopathic pulmonary fibrosis, obstructive sleep apnea syndrome, and globus pharyngeus present weaker or inconclusive associations, so the Consensus emphasizes the need for strict diagnostic criteria and exclusion of differential diagnoses before attributing them to GERD. The document further underscores that clinical response to treatment should not be considered as diagnostic proof by itself, and recommends the use of complementary studies such as multichannel intraluminal impedance-pH monitoring to support the diagnosis in patients with predominantly extraesophageal symptoms7,8.
Use of validated questionnaires
Given that GERD is a disease with a complex diagnosis, it is usually established through the combination of various diagnostic methods (endoscopy and pH monitoring). One of the most widely used strategies to avoid invasive procedures is the application of standardized questionnaires. Among them, one of the most employed is the GERD-Q, developed from previous instruments such as the Reflux Disease Questionnaire (RDQ), the Gastrointestinal Symptom Rating Scale (GSRS), and the Gastroesophageal Reflux Disease Impact Scale (GIS). The GERD-Q questionnaire consists of six items, of which four act as positive predictors (heartburn, regurgitation, sleep disturbances due to reflux symptoms, and use of antireflux drugs) and two as negative predictors (epigastric pain and nausea)9. For the diagnosis of GERD, a cutoff score of 8 points has been proposed, which has shown a sensitivity of 64.6% and a specificity of 71.4%10. Studies have been conducted in the Mexican population that have compared the GERD-Q questionnaire with other simultaneous diagnostic methods, such as endoscopy and pH monitoring, and it was found that the questionnaire presented a sensitivity and specificity of 72%, as well as a positive predictive value of 87%, compared to cabinet tests11. However, the role of GERD-Q as a diagnostic tool is limited, since typical symptoms of the disease can occur in individuals who do not have it, while some patients with the disease may be asymptomatic. Additionally, there are other esophageal pathologies that can manifest with symptoms similar to those of GERD (achalasia, rumination, eosinophilic esophagitis). For these reasons, the exclusive use of questionnaires is not sufficient to establish a definitive diagnosis, and should not replace the performance of more specific and objective tests10.
The COuGH Reflux Questionnaire (CGRQ) has also been validated. It aims to evaluate the possible association between chronic cough and GERD, mainly assessing the frequency and intensity of cough and the presence of typical reflux symptoms, as well as extraesophageal manifestations such as dysphonia, throat clearing, or globus sensation. In its validation, a score ≤ 2.5 showed a sensitivity of 49%, a specificity of 88%, a positive predictive value of 89%, and a negative predictive value of 42% for ruling out proven GERD, while a score ≥ 5 achieved a sensitivity of 65%, a specificity of 71%, a positive predictive value of 52%, and a negative predictive value of 82% for confirming proven GERD12.
Diagnostic tests
Therapeutic trial
Most guidelines and consensuses recommend using a therapeutic trial with PPIs as an initial diagnostic strategy in patients with typical symptoms such as heartburn and regurgitation, under the assumption that a favorable response to treatment confirms the diagnosis of GERD. The therapeutic trial consists of the administration of acid blockers (usually PPIs) at a single or double dose for 2-4 weeks or 4-8 weeks. The test is considered positive when there is symptom improvement > 50%. It should be used as a first measure in young patients with typical symptoms without alarm symptoms13,14.
Although this approach is practical and efficient in common clinical scenarios, its diagnostic accuracy is limited. According to a meta-analysis and a prospective study, this strategy presents a combined sensitivity of 78% and a specificity of only 54% when compared to endoscopy and pH monitoring as reference methods15,16. In patients whose predominant symptom is non-cardiac chest pain, the therapeutic trial with PPIs has been shown to be effective17. In contrast, when both endoscopy and pH monitoring are normal, no significant response to PPI treatment has been observed compared to placebo. Furthermore, the presence of chest pain or heartburn does not reliably predict the efficacy of PPIs in these cases16.
Recently, therapeutic trial with P-CABs has been proposed, drugs that have greater potency and speed of action, which could allow for shorter and more precise therapeutic trials, in theory. These data have not yet been confirmed and there are currently several ongoing studies with the purpose of investigating the diagnostic efficacy with these new acid production suppressing molecules.
In case of lack of response to the therapeutic trial, upper digestive endoscopy is indicated. We must remember that those patients with alarm symptoms (dysphagia, persistent vomiting, signs of digestive bleeding, anemia) are candidates for endoscopic study, and a therapeutic trial should not be performed in them.
Esophagogram
The esophagogram (esophagogastroduodenal transit) should not be used as a diagnostic test due to its low sensitivity, specificity, and positive predictive value. Although it may show findings such as the presence of a hiatal hernia or a positive “siphon maneuver”, these are not conclusive4. Its usefulness lies in detecting large structural abnormalities, short esophagus, or, in patients with previous surgery, migration of a fundoplication4.
Upper digestive endoscopy
Endoscopy has been used as a fundamental diagnostic tool, as it allows an objective evaluation of the esophageal mucosa. In the context of GERD diagnosis, erosive esophagitis, Barrett’s esophagus > 3 cm, and peptic stricture are considered specific findings3. For the characterization of erosive esophagitis, the Los Angeles classification constitutes a widely validated and highly useful instrument, as it allows a systematic evaluation of mucosal lesions. This classification divides esophagitis into four grades according to the extent and location of the observed lesions13,14:
- ‒ Grade A: one or more mucosal breaks ≤ 5 mm, not crossing the folds13 (Fig. 1). Does not establish the definitive diagnosis of GERD15.
- ‒ Grade B: one or more mucosal breaks > 5 mm, not crossing the folds13 (Fig. 1).
- ‒ Grade C: one or more mucosal breaks that cross the folds, but involving < 75% of the esophageal circumference13 (Fig. 1).
- ‒ Grade D: one or more mucosal breaks that cross the folds and involve > 75% of the esophageal circumference13 (Fig. 1).
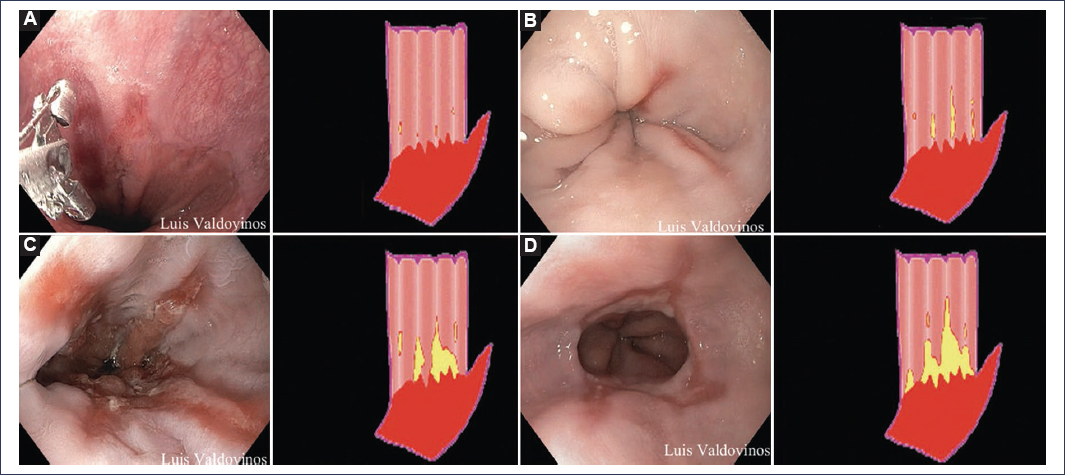
Figure 1. Los Angeles Endoscopic Classification (A-D).
Currently, the presence of Los Angeles grade B, C, or D esophagitis is considered diagnostic of erosive GERD. Grade A esophagitis is no longer considered a diagnostic criterion for GERD because asymptomatic patients may present this finding on endoscopy; however, those with suggestive symptoms, grade A esophagitis, and abnormal esophageal exposure by pH monitoring or pH-impedance monitoring are diagnosed with non-erosive GERD4,13.
For adequate evaluation of esophagitis by endoscopy, it is necessary to discontinue PPIs at least 2 to 4 weeks before the procedure16. If the study is performed ahead of time, there is a risk that esophagitis will manifest at a lower grade or may not even be detectable, which can make accurate representation of the GERD phenotype difficult or cause a false negative on endoscopy4. During the endoscopic procedure, a hiatal hernia may be observed, which although a risk factor for the disease, does not represent a diagnostic criterion (sensitivity of 30-35% and specificity of 90-95%)17,18.
BARRETT’S ESOPHAGUS
Barrett’s esophagus is defined endoscopically by the presence of columnar mucosa (with or without intestinal metaplasia) that extends proximal to the esophagogastric junction. On endoscopy, this mucosa is observed as tongues or as a circumferential band of salmon-pink color that replaces the pale normal squamous epithelium of the distal esophagus3. For adequate characterization, the Prague classification (C & M) is used, which describes:
- ‒ C (circumferential): extent (in centimeters) of columnar mucosa forming a complete circumference (Fig. 2).
- ‒ M (maximum): most proximal extent (in centimeters) of any tongue of columnar mucosa, measured from the esophagogastric junction (EGJ)3 (Fig. 2).
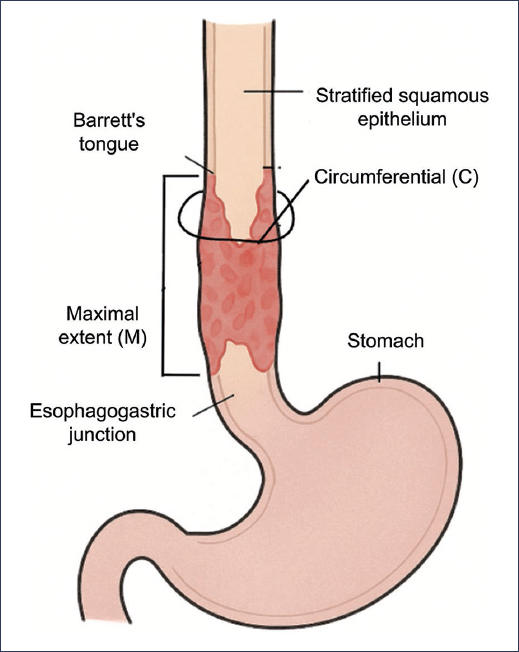
Figure 2. Prague Classification for Barrett’s esophagus. Didactic illustration showing an esophagus with squamous epithelium, the esophagogastric junction, and how circumferential extension and maximum extension are measured.
PEPTIC STRICTURE
Peptic stricture is a chronic complication of GERD, characterized by the formation of a ring or fibrous narrowing in the distal esophagus secondary to repeated inflammation and scarring of the mucosa. Endoscopically, it is observed as a concentric and rigid reduction of the esophageal lumen, often accompanied by whitish or scarred mucosa, and occasionally with signs of active esophagitis. Endoscopy is not only essential for its diagnosis, allowing assessment of the length and degree of stenosis, but is also fundamental for treatment, as it facilitates esophageal dilations using balloons or bougies. Additionally, it is essential to obtain biopsies of all strictures, even those with benign appearance, to rule out underlying malignant lesions, such as esophageal adenocarcinoma19,20.
Histopathology
The histopathological diagnosis is established by non-pathognomonic microscopic changes: tissue eosinophilia (69% in erosive esophagitis, 34% in non-erosive reflux disease), papillary elongation, basal cell proliferation, dilated cellular spaces, neutrophil infiltration (in erosive esophagitis), and specialized intestinal metaplasia (the only diagnostic finding)21,22.Although histology alone is not diagnostic of GERD, scoring systems have been developed that quantify histological changes to support the diagnosis and differentiate it, such as eosinophilic esophagitis21. Some examples are:
- ‒ ERD-HR (Erosive Reflux Disease-Histology Reflux score): evaluates the presence and degree of papillary elongation, basal layer hyperplasia, dilation of intercellular spaces, inflammation (eosinophils/neutrophils), and epithelial damage23,24.
- ‒ HSS (Histology Scoring System) by Savarino: quantifies five key parameters, which are basal hyperplasia, papillary elongation, intercellular dilation, inflammatory infiltrate, and epithelial damage, on a severity scale23.
These scales have shown good correlation with acid exposure parameters measured by pH monitoring and help standardize the interpretation of biopsies23.
Likewise, electron microscopy allows an ultrastructural evaluation of the esophageal mucosa to be performed and changes that are not always visible on conventional optical microscopy to be identified. In GERD, marked dilation of intercellular spaces (between keratinocytes of the squamous epithelium), alterations in intercellular junctions, especially in desmosomes and tight junctions, and changes in mitochondria, intracellular edema, and cytoplasmic vacuolization have been described. These findings are considered early markers of acid-induced epithelial damage, even in the absence of endoscopically visible erosions24.
Finally, dilation of intercellular spaces is one of the earliest histological changes in GERD. It is believed that increased paracellular permeability allows the diffusion of protons and other noxious solutes to the deeper layers of the epithelium, activating nerve receptors and producing symptoms (e.g., heartburn). It can be measured semiquantitatively with hematoxylin-eosin staining, but electron microscopy is the reference technique for its detection. Although not exclusive to GERD (it is also seen in eosinophilic esophagitis and drug-induced damage), its presence, together with other histological findings, reinforces the diagnosis25.
24-hour esophageal pH monitoring
For many years, esophageal pH monitoring was considered the reference method for diagnosing non-erosive GERD (NERD). However, in the most recent clinical guideline from the American College of Gastroenterology (ACG), it is established that there is no single and definitive diagnostic method for GERD3. Consequently, the diagnosis should be based on an integration of clinical factors, including symptomatic presentation, endoscopic evaluation of the esophageal mucosa, reflux monitoring, and response to treatment3.
The wireless telemetry capsule (Bravo™ Reflux Capsule, Medtronic, Minneapolis, MN, USA), which is attached to the esophageal mucosa during endoscopy, and transnasal catheter monitoring, are methods that present advantages and limitations26. During monitoring, multiple variables are analyzed, including acid exposure time (AET), which is the most important parameter, as well as the number of reflux episodes and their correlation with symptoms. Regarding monitoring time duration with a transnasal catheter, whether only for pH monitoring or for pH-impedance monitoring, it is generally 24 hours, whereas the wireless capsule allows extended recording for between 48 and 96 hours.
The capsule has the advantages of eliminating the physical discomfort and social awkwardness that the transnasal catheter can generate, which favors the patient maintaining daily activities during the evaluation26,27. However, monitoring through probes allows pH monitoring with multichannel impedance to be performed, which, unlike conventional pH monitoring, is capable of identifying weakly acidic or non-acidic reflux episodes, evaluating bolus clearance, and determining the proximal extent of reflux3. Multichannel impedance pH monitoring is useful if the AET result is in the gray zone, as it allows evaluation of additional parameters that support the diagnosis of NERD18. There is currently no capsule system that allows impedance measurements to be made.
Dual pH sensor catheters and pharyngeal probes are also available to document acid reflux that reaches the proximal esophagus and oropharynx; however, their clinical utility is limited due to high variability in results reported by various studies3.
Among all the variables evaluated, both with wireless capsule and transnasal catheter, the most consistently reliable are AET and the DeMeester composite score3. According to the Lyon Consensus 2.0, the most relevant datum for GERD diagnosis during classic pH monitoring is AET: an AET > 6% in 24 hours establishes an objective diagnosis of NERD, and when this value is positive, no additional evaluation is required. An AET < 4% is considered normal, and when AET is 4-6% it is considered a gray area4.
In patients in whom the endoscopic capsule is used with the purpose of identifying NERD, measurements should be made every 24 hours and AET calculated (remember that the study can last up to 96 hours). For interpretation, the worst day over a 24-hour period is taken into account; this means that, although evaluations of 48 or 92 hours are performed, the total acid exposure time should not be taken, but rather divided into 2, 3, or 4 days depending on the duration of the study, and if on one of these days the AET is > 6%, it is considered diagnostic of NERD. This is one of the main reasons why the capsule has greater sensitivity, since a longer study manages to avoid as much as possible the daily variability of the disease, a strategy known as “worst day evaluation”4 (Fig. 3).
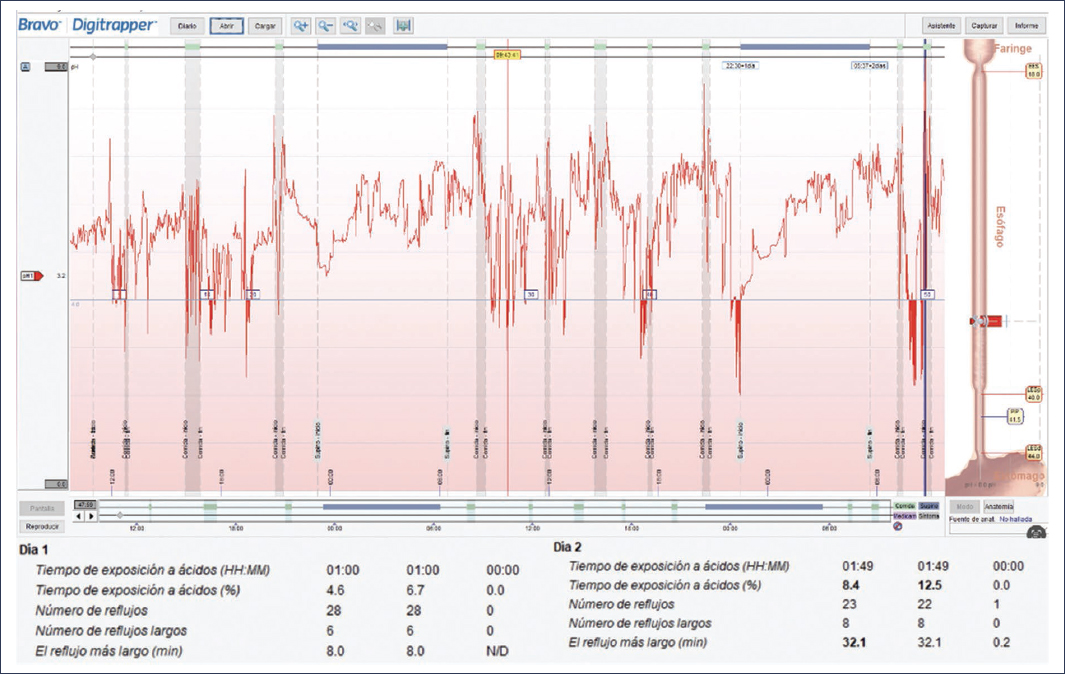
Figure 3. pH monitoring.
Symptom indices can be evaluated with all pH monitoring systems. This measure is useful for establishing symptomatic association, for predicting clinical response to treatment, and for diagnosing reflux hypersensitivity. The symptom index (SI) and symptom association probability (SAP) are considered positive when they are > 50% and > 95%, respectively3,4. The SI is calculated by dividing the total number of reflux episodes associated with symptoms by the total number of symptoms reported during monitoring. To calculate SAP, the 24-hour monitoring time is divided into 720 2-minute intervals, in each interval the presence or absence of reflux and symptoms is recorded, and Fisher’s exact test is applied to obtain a p value. The clinical validity of both indices has been questioned and one has not been shown to be superior to the other for diagnostic purposes3,4.
In pH-impedance monitoring, some other additional measurements can be made that are useful for the diagnosis of NERD, especially when AET is in the gray zone. Baseline nocturnal impedance is a surrogate for mucosal integrity; it has been observed that inflamed mucosa, as in patients with reflux, has decreased baseline nocturnal impedance. Measurements are made during sleep, between 2 and 5 a.m., when there is greater impedance stability. The digital sensors are between 3 and 5 cm from the lower esophageal sphincter, three 10-minute periods are taken, and an arithmetic mean is calculated; a value > 1500 ohms is considered normal4. The number of reflux episodes by impedance is interpreted as follows: abnormal more than 80 episodes, in gray zone between 40 and 80 episodes, and normal less than 40 episodes. These additional data from pH-impedance monitoring are useful, especially if the AET result is in the gray zone (that is, 4-6% AET). For example, the presence of more than 80 reflux episodes in 24 hours by impedance, with positive SI and baseline nocturnal impedance < 1500 ohms, in a patient with AET result in gray zone, suggests NERD in the presence of symptoms. If AET is < 4%, the study is negative for GERD, and according to SI, the patient can be classified as reflux hypersensitivity (positive SI) or functional heartburn (negative SI)4.The parameters for the interpretation of pH monitoring with antisecretory treatment are different, and the presence of AET > 4% or more than 80 reflux episodes by impedance is considered abnormal; when these criteria are met, refractory GERD is considered. These findings in pH-impedance monitoring with antisecretory treatment have a sensitivity of 90% for predicting response to surgical management3,4,21,22.
Esophageal manometry and Milan score
According to the Lyon Consensus 2.0, GERD is diagnosed by endoscopy and pH monitoring findings, with esophageal manometry being a study used only secondarily, which has the purpose of locating the lower esophageal sphincter, for the correct placement of GERD monitoring catheters and ruling out disorders such as achalasia and rumination, which simulate GERD. Despite its secondary role in diagnosis, esophageal manometry is widely requested and has been proposed as a useful complement in patients with reflux23.
Understanding of the anatomical and functional factors that favor GERD has advanced considerably thanks to high-resolution manometry, which allows detailed evaluation of the EGJ. High-resolution esophageal manometry has the purpose of ruling out diseases with manifestations similar to those of GERD (achalasia, rumination), and evaluating esophageal function and peristaltic reserve through different metrics, as well as the type, pressure, and relaxation of the EGJ. Some phenotypes, such as ineffective esophageal motility, aperistalsis, and poor peristaltic reserve during multiple rapid swallow sequences, have been associated with postoperative dysphagia23,28.
In version 4.0 of the Chicago Classification, special emphasis has been placed on two key metrics: the separation between the lower esophageal sphincter and the crural diaphragm, and the EGJ contractile integral (EGJ-CI). Both parameters have been shown to be closely related to an alteration in the EGJ barrier function and are associated with greater AET, measured with ambulatory pH monitoring. On the other hand, the clinical relevance of ineffective esophageal motility has been reinforced, which usually coexists with GERD, especially in patients with refractory symptoms23,29.
As a complement to standard metrics, the straight leg raise maneuver has been proposed as a useful dynamic tool to evaluate EGJ competence in situations of physiological stress. This maneuver generates a transient increase in intra-abdominal pressure, and in patients with an incompetent antireflux barrier, this pressure can be transmitted retrograde and be indicative of a dysfunctional EGJ. A multicenter study demonstrated that an increase of at least 11 mmHg in intraesophageal pressure during the maneuver is associated with pathological esophageal acid exposure (AET > 6%), showing a sensitivity of 79% and a specificity of 85% in patients with persistent symptoms suspicious of GERD30.
In this context, the Milan score has been developed as a comprehensive proposal to improve the evaluation of GERD in patients undergoing high-resolution manometry, which includes EGJ morphology, EGJ-IC value, presence of ineffective esophageal motility, and response to the straight leg raise maneuver, being useful for stratifying the risk and severity of GERD25,28. The Milan score is a construct that takes into account manometry data that can predict GERD: ineffective esophageal motility, leg raise maneuver, and its effect on the transmission of abdominal pressure to the thorax, EGJ-IC and EGJ type (esophagogastric junction) to determine the presence and size of a hiatal hernia. It assigns a score to each of these findings and predicts the possibility of presenting GERD with an area under the curve of 0.8830.
Lyon score
In an article recently published in the American Journal of Gastroenterology31, the Lyon score is described for the first time, a new scoring system for GERD based on the criteria of the Lyon Consensus 2.0. This score was designed with the purpose of predicting response to antireflux treatment. It takes into account endoscopic findings (erosive esophagitis), multichannel impedance pH monitoring (esophageal acid exposure, number of reflux episodes, baseline nocturnal impedance), and manometric findings (transmission of abdominal pressure to the thorax during sustained leg raise and hiatal hernia > 3 cm). A score > 5 is positive for GERD, and a score > 10 is considered severe reflux32.
This score was designed to increase diagnostic certainty in GERD when endoscopy is negative and to predict clinical response to antireflux therapies (medical or surgical), defined as a≥ 50% reduction in global symptoms30. The area under the curve includes the following values: AET, mean baseline nocturnal impedance, number of reflux episodes, combined symptom score, and presence or absence of hiatal hernia27. The optimal test threshold is 6.25 points, with a sensitivity of 81.2% and a specificity of 73.4%. Also important is that the Lyon score showed greater sensitivity than the classic DeMeester composite score for predicting therapeutic response, both in the development cohort and in one from Asia32.
Lyon Consensus 2.0 and actionable GERD
The Lyon Consensus established conclusive diagnostic criteria both for and against GERD, as well as complementary metrics that help confirm or rule out the disease when primary criteria are borderline or inconclusive. In 2023, an update of this consensus was published under the name Lyon Consensus 2.04. This new version offers a contemporary definition of what is considered clinically relevant GERD, presented as actionable GERD, which could be translated as actionable or intervention-susceptible GERD, that is, one in which findings from esophageal studies justify modifying, intensifying, or personalizing the therapeutic approach of the symptomatic patient, whether long-term medical treatment, endoscopic or surgical4.
Among the main changes from the original consensus are the recognition of Los Angeles grade B esophagitis as conclusive diagnostic evidence of GERD, the standardization of parameters and thresholds to be used in extended monitoring with wireless pH capsule, and useful diagnostic criteria for identifying refractory GERD when studies are performed under antisecretory treatment in patients with already confirmed diagnosis; finally, all those metrics that have not demonstrated diagnostic utility in the context of clinically relevant GERD are eliminated31,33,34 (Fig. 4).
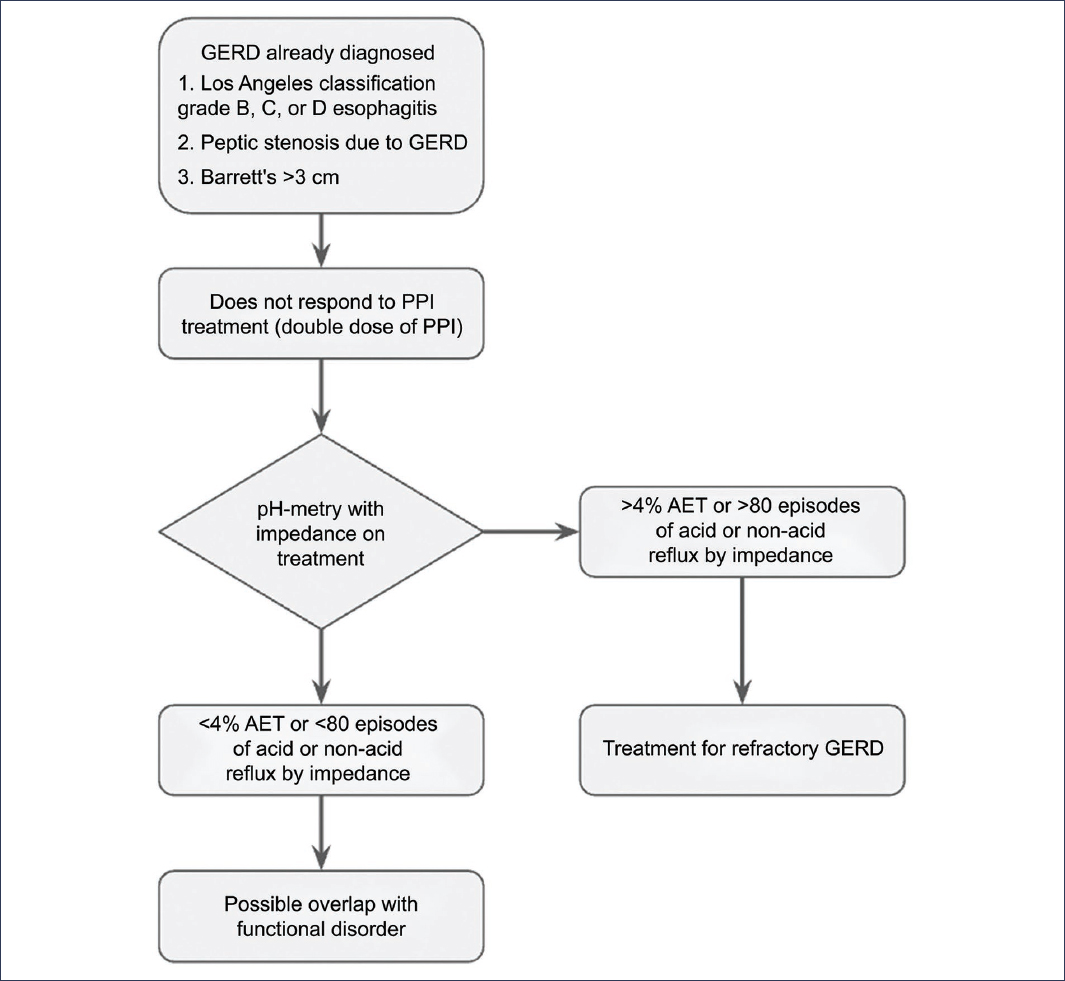
Figure 4. Flow diagram for the diagnosis of refractory gastroesophageal reflux disease (GERD). PPI: proton pump inhibitors; AET: acid exposure time.
In this update, clinically relevant GERD is defined as those scenarios in which management requires long-term acid suppression, intensification of medical treatment, or consideration of interventionist options. In these cases, it is essential to have a GERD diagnosis with high certainty. On the other hand, contrary to what was stated in the original Lyon Consensus, well-characterized Los Angeles grade B esophagitis constitutes conclusive diagnostic evidence of GERD and does not require additional confirmation through reflux monitoring before starting treatment4. Another important recommendation is that endoscopy should be performed between 2 and 4 weeks after discontinuation of antisecretory therapy in patients with an unconfirmed diagnosis of GERD. This is because mucosal healing with PPIs occurs in approximately 80-90% of cases, so the probability of detecting significant esophagitis decreases considerably if endoscopy is performed after more than 8 weeks of PPI treatment; this means that endoscopy could yield false negative results if performed with antisecretory treatment4.
Another significant improvement established in this update was extended pH monitoring using a wireless capsule, based on the fact that up to 70% of symptomatic patients present with normal esophageal mucosa on initial endoscopy. During this reference endoscopy, it is possible to place a wireless probe to monitor AET for up to 96 hours, which allows considering the daily variability of AET4. Additionally, as already mentioned, this method offers better patient tolerance and reduces delay in diagnosis.
The prognostic performance of wireless reflux monitoring is significantly superior when extended from 72 to 96 hours, compared to the first 48 hours of recording. The third and fourth days of monitoring with the wireless capsule provide crucial information for distinguishing a predominant physiological pattern of acid exposure from a pathological one, even allowing discontinuation of PPIs when a predominant physiological pattern is identified. Therefore, pH monitoring for 96 hours better predicts the need to discontinue PPI treatment or its continuation, compared to shorter monitoring periods4,31.
Differential diagnoses
Non-cardiac chest pain refers to retrosternal pain that is not associated with demonstrable cardiovascular disease and can have multiple etiologies, including esophageal conditions, as well as rheumatological or pulmonary causes. Among patients with non-cardiac chest pain of esophageal origin, a subgroup with so-called functional chest pain is identified, characterized by pain not attributable to GERD (which is established through normal endoscopy and multichannel impedance pH monitoring) nor to spastic motor disorders (ruled out through high-resolution esophageal manometry without alterations)4,31.
In patients with typical symptoms suggestive of GERD, but with normal esophageal acid exposure, two conditions are distinguished: reflux hypersensitivity, in which there is a positive symptomatic association between reflux episodes and reported symptoms (positive SI), and functional heartburn, in which there is no correlation between reflux episodes and symptom occurrence (negative SI). These latter conditions represent functional subtypes within the spectrum of esophageal non-cardiac chest pain4,30,31,33,35,36.
Other differential diagnoses to consider are functional heartburn, rumination, and primary esophageal motor disorders, such as achalasia. Functional heartburn is defined as the recurrent sensation of retrosternal burning, without evidence of GERD, structural alterations, or esophageal motility disorders, according to Rome IV criteria.
Its diagnosis is clinical and is established by exclusion of GERD and other causes, generally supported by a normal esophageal impedance pH study and lack of response to PPIs37.
Rumination is a disorder characterized by repeated regurgitation of recently ingested food, which is re-chewed and swallowed again or expelled, without nausea or retching, due to voluntary or involuntary contractions of the abdominal muscles. The diagnosis is based on anamnesis, direct observation of episodes, and occasionally high-resolution manometry with impedance to confirm the typical pattern of abdominal contraction and lower esophageal sphincter relaxation38,39.
Finally, achalasia is a primary motor disorder of the esophagus characterized by the absence of esophageal peristalsis and incomplete or absent relaxation of the lower esophageal sphincter, which causes progressive dysphagia and regurgitation. The diagnostic test of choice is high-resolution esophageal manometry, which allows classification of achalasia subtypes according to the Chicago Classification40 (Fig. 5).
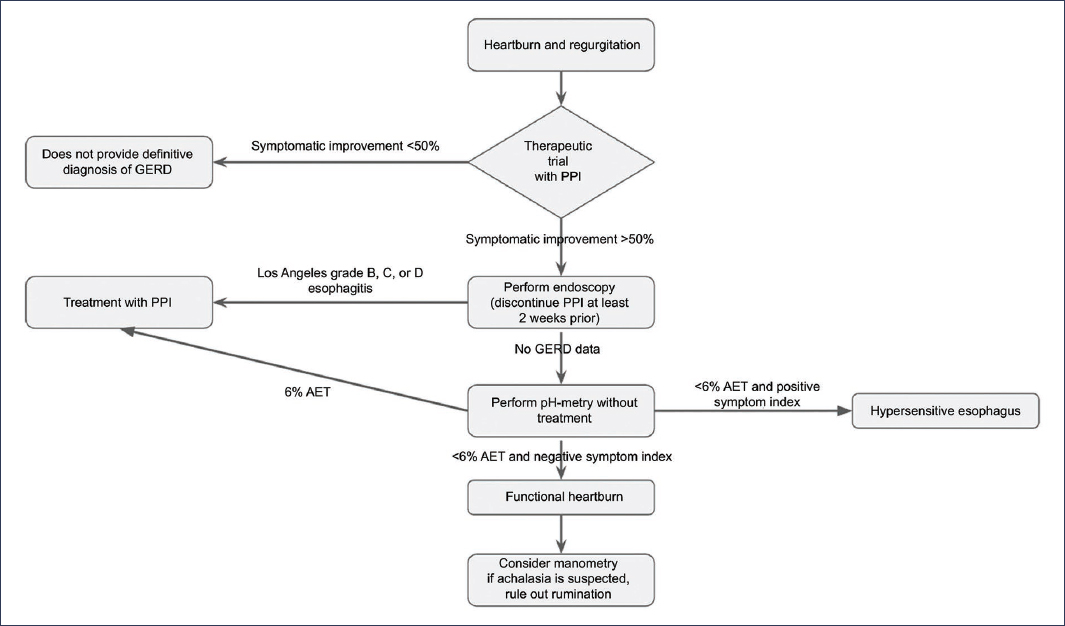
Figure 5. Flow diagram for the definitive diagnosis of gastroesophageal reflux disease (GERD). PPI: proton pump inhibitors; AET: acid exposure time.
Funding
The authors declare that they received no funding for this study.
Conflicts of interest
The lead author declares being a speaker and researcher for Laboratorios Carnot and Adium; speaker for AstraZeneca, Pfizer, Alfa Sigma, Ferrer, Medtronic, Takeda, Siegfried Rhein, Megalab, and Prometis Pharma. No fees or financial support directly related to the preparation of this manuscript were received.
Ethical considerations
Protection of people and animals. The authors declare that the procedures followed conformed to the ethical standards of the responsible human experimentation committee and in accordance with the World Medical Association and the Declaration of Helsinki. The procedures were authorized by the institution’s Ethics Committee.
Confidentiality, informed consent, and ethical approval. The authors have followed their institution’s confidentiality protocols, have obtained informed consent from patients, and have approval from the Ethics Committee. The recommendations of the SAGER guidelines have been followed, according to the nature of the study.
Declaration on the use of artificial intelligence. The authors declare that they did not use any type of generative artificial intelligence for the writing of this manuscript.
